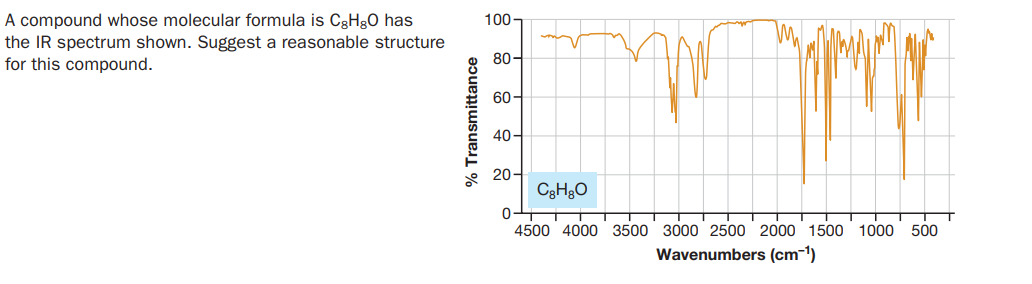
A compound whose molecular formula is C3H30 has the IR spectrum shown. Suggest a reasonable structure for this compound. 100- 80- 60- 40- * 20- 0수 4500 4000 3500 3000 2500 2000 1500 1000 500 Wavenumbers (cm-1) % Transmittance
Q: H3C CI C= H3C CI
A:
Q: Identify this compound with molecular formula C9H10O IR peak at 1742 cm-^1,H nmr data (ppm) at…
A:
Q: What is the structure of the compound with the formula C10H14O, if it shows a strong broad IR signal…
A: We have given data that match with the compound is 4-tert-butylphenol. The explanation is provided…
Q: Question is attached here
A: IR spectroscopy is the analysis of infra red light with the given organic molecule. It helps to…
Q: Click the "draw structure" button to launch the drawing utility. Draw the structure with the…
A: The molecular formula of the organic compound given is C8H11N. The degree of unsaturation =…
Q: The unknown compound that gives the following IR and NMR spectra has the formula CaH1,0. Based on…
A:
Q: The IR and TH NMR data of a compound with the formula C7H1402 are given below. Please choose the…
A:
Q: Deduce a possible structure for the compound with the IR absorptions below. (a) C5H8O: 2950, 1750…
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: 4000 3000 2000 1500 1000 500 Wavenumbers (cm) 3H 2H 2H 2H Exchanges with Dz0 11 10 9 8 4. ppm 170…
A:
Q: Examine the IR spectra below. Each arrow points to a significant absorption that indicates a…
A: IR spectra is one of the very important spectroscopic tool to identify the functional groups of…
Q: From the given values of ¹H-NMR data, predict the structure of the organic compound with the…
A: Firstly we find the double bond equivalent of the compound. And then we figure out the presence of…
Q: 3000 * TRANSMITTANCE 6CCC LS62 1248
A: Provided molecular formula of compound=C4H10O Broad ir peak at =3339 cm-1 Provided 6H doublet at…
Q: 100 80- 60- 40- 20. 0. 4000 3500 3000 2500 2000 1500 1000 Wavenumber (cm") What information may be…
A: Note: 3050 cm-1 sp2 C-H stretch, 2900 cm-1 sp3 C-H stretch. Therefore the compound is olefin.
Q: Give the structure for the compound with the empirical formula C3H3O2 and the following spectral…
A:
Q: Propose a structure for a compound of molecular formula C7H14O2 with an IR absorption at 1740 cm−1…
A: The given compound has the molecular formula C7H14O2 and it shows an absorption band in the IR…
Q: For the functional groups on the following molecule, what characteristics IR absorptions would be…
A:
Q: For the functional groups on the following molecule, what characteristics IR absorptions would be…
A: Correct option is first option i.e peaks around 1650 and 3300/cm Reference values C=C stretching…
Q: Identify the hydrocarbon that gives the IR spectrum shown in Figure 14.50 and has an m/z 102 in its…
A: Solution -
Q: A hydrocarbon has the following mass spectrum and IR spectrum. 100 55 80 60 40 84 20 20 40 60 80 100…
A: structure B is the correct IR DATA SHOW PEAK at 3000 it is peak of C-H bond at 1600 show C=C bond(…
Q: Consider the data below, Propose a structure for this compound. C₂H₂C1O; IR absorption at 810 cm-¹…
A:
Q: Please provide the IUPAC name (only) for the compound that best represents the spectroscopic data…
A:
Q: A chiral compound Y has a strong absorption at 2970−2840 cm−1 in its IR spectrum and gives the…
A: From the given mass spectrum it is clear that the compound contains m and m+2 peaks with the same…
Q: A chiral compound Y has a strong absorption at 2970–2840 cm−1 in its IR spectrum and gives the…
A: From the given mass spectrum it is clear that the compound contains m and m+2 peaks with the same…
Q: Thymol (molecular formula C10H14O) is the major component of the oil of thyme. Thymol shows IR…
A: Given information: Molecular formula for thymol = C10H14O Absorption peak at 3500-3200, 3150-2850,…
Q: Propose a structure for the compounds that display IR spectra with the following peaks : A)…
A: The spectrum which is used to identify the structure or study the chemical compounds are known as IR…
Q: What is the structure of the compound with the formula C6H12O2, if it has a strong IR signal near…
A:
Q: The following compound has the formula CaH,O2Br. Based on its IR and 'H NMR propose its structure.…
A:
Q: 200 180 160 140 120 100 80 60 40 CDS-06-420 ppm The Spectra shows an isomer of C3H60, which compound…
A: Here we are required to find the structure which matches with 13CNMR
Q: 4000 3000 2000 1500 1000 500 Wavenumbers (cm') 6H i 2H 1H 3 1 ppm 85 80 70 65 60 55 50 45 40 35 30…
A:
Q: Deduce the structure of the compound whose formula is C2H3NS from the IR spectrum below. 100 90 80…
A: Infrared spectroscopy deals with the infrared region of electromagnetic spectrum. It is divided into…
Q: Deduce the structures of compounds A and B, two of the major components of jasmine oil, from the…
A: IR absorbtion at 3091 - 2895 corresponds to aromatic double donds and 1743 corresponds to an ester…
Q: f. This compound has formula C3H60. The UV spectrum of this compound shows no maximum above 205 nm.…
A: Structure elucudation is done with the Help of IR , HNMR and Mass spectra
Q: A compound of molecular formula C4H8O2 lost its label. Based on the NMR and IR data given, determine…
A: The IR stretching frequencies stands for different functional groups and proton nmr stands for…
Q: Propose a structure for a compound of molecular formula C7H14O2 with an IR absorption at 1740 cm−1…
A: The given compound has the molecular formula C7H14O2 and it shows an absorption band in the IR…
Q: Determine the structure for the molecule with the formula C11H140 that most likely produced the 'H…
A:
Q: Which set of IR bands (in cm-1) would you expect to see for the following compound?
A: The correct answer about IR stretching frequencies is given below
Q: LTE lI. 9:.0 A docs.google.com Lläi 4 The IR spectrum of methyl salicylate shows absorption bands at…
A: Different types functional group has the different stretching and bending frequency . Hence the…
Q: The unknown compound that gives the following IR and NMR spectra has the formula CH1«0. Based on…
A:
Q: Click the "draw structure" button to launch the drawing utility. Identify the structure of compound…
A: C9 H100 D.B.E=No. of carbon+ 1 - No. of memovalent atem+ No 2 of trivalent atem2D.B.E =9+1-102…
Q: A compound with the molecular formula C6H11BrO2 shows an intense band at 1739 cm-1 in the IR…
A: Given: An intense IR spectrum at 1739 cm-1, then it should be a ester functional group. according to…
Q: For the functional groups on the following molecule, what characteristics IR absorptions would be…
A: we have to identify the absorbtion peak for this molecule.
Q: Which of the following compounds is represented by this spectral data? L00 4000 so00 1000 sdo…
A: A multiple choice question based on IR spectrum, which is to be accomplished.
Q: 4. Propose a structure for a compound that meet the following criteria: a. C5H3, with IR absorptions…
A:
Q: Deduce a possible structure for the compound with the IR absorptions below. C5H8O: 2950,…
A: IR spectra is the spectra that uses the IR radiation to excite the molecule where the IR spectrum is…
Q: Which of the following molecules is responsible for this IR spectrum? 0.8- 0.6 0.4 0.2 3000 2000…
A: IR(Infrared) Spectroscopy:- It is used to determine different types of functional groups present in…
Q: The following spectroscopic data were obtained for compound X (molecular formula C10H140), a natural…
A:
Q: For the functional groups on the following molecule, what characteristics IR absorptions would be…
A:
Q: 3. Draw a structure for a compound that meets the following description: A compound, C3H6O, with no…
A: Given, Compound C3H6O , with no IR absorption above 3000 cm-1
Q: For the functional groups on the following molecule, what characteristics IR absorptions would be…
A: Infrared spectroscopy is one of the useful techniques analyze the structure of an organic compound…
Q: Compound Y (molecular formula C6H10) gives four lines in its 13C NMR spectrum (27, 30, 67, and 93…
A: The molecular formula of the compound is C6H10. Calculate the double bond equivalence as follows DBE…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- What functional group is most likely present if a compound shows IR absorption at these frequencies? Q.) 2950 cm-1Which of the following IR absorbances do not correspons with the functional group in butanal? 2720 cm-1 3300 cm-1 1725 cm-1 2820 cm-11. Deduce a possible structure for the compound with the IR absorptions below. (a) C5H8O: 2950, 1750 cm-1 (b) C4H8O: 2950, 2820, 2715, 1715 cm-1 2. How could IR spectroscopy be used to distinguish between the following pair of compounds? (a) CH2=CHCH2CH(CH3)2 and CH3CH2CH2CH(CH3)2 (b) CH3OCH2CH3 and CH3CH2CH2OH 4. How does the O-H stretch in the IR spectrum of a carboxylic acid differ from the O-H stretch of an alcohol?
- Complete the following answer using the data provided here: Molecular formula: C4H8O2 Important IR data (cm-1): 3280 (broad); 2980-2730 (many); 1690; 1254 All 1H NMR data (ppm, splitting, integration): 11.0 ppm (s), 1; 2.57 ppm (m), 1; 1.07 ppm (d), 6 1. Sketch out a 1H NMR spectrum showing peak locations and peak splitting on a ppm scale for the data provided above. Include the data labels above in your spectrum, but do not show integration lines. 2. draw the most proper line bond structure for the data given aboveidentify the structure of molecular formula c9h10o with IR peak at 1688 Hnmr data ppm at 1.22(triplet,3H),2.98(quartet 2H) and 7.28-7.95(multiplet,5H)Propose a structure consistent with each set of data.a.) C9H10O2: IR absorption at 1718 cm−1 b.) C9H12: IR absorption at 2850–3150 cm−1
- For a mono-substituted phenyl ring, which of the following absorbances would one expect to observe? Group of answer choices 880-900 730-770, 690-710 3000-3100 735-7702. Use the spectroscopic data provided below to answer the questions.a. Determine the structure of C5H8O from the IR and NMR data given.IR: 3023 cm-1, 1685 cm-1, 1619 cm-1. 13C-NMR: 7.9 ppm, 32.9 ppm, 127.7 ppm, 136.5 ppm, 201.1 ppm1H-NMR: 1.1 ppm (t, 3H), 2.6 ppm (q, 2H), 5.8 ppm (d, 1H), 6.23 ppm (d, 1H), 6.34 (t, 1H)b. Identify all of the Lewis acid sites and the Lewis base sites on your molecule.What are the functional groups and peaks of this C10H22 IR Spectra?
- Propose a structure consistent with each set of data. C8H10 : IR absorptions at 3108–2875, 1606, and 1496 cm−1Calculate the IHD of C7H6XNO and identify the important peaks in the following MS spectral data and draw the structure of the important peaks in the following MS spectral data.compound with the molecular formula C7H9N exhibits IR bands at 3450 cm-1 (medium, doublet), and 855 cm-1 (strong) and shows the following major mass spectral signals (m/z): 106 (base); 107 (M+, about 70% of base), 91 (40% of base), and 77 (about 20% of base). Deduce a reasonable structure from this data.

