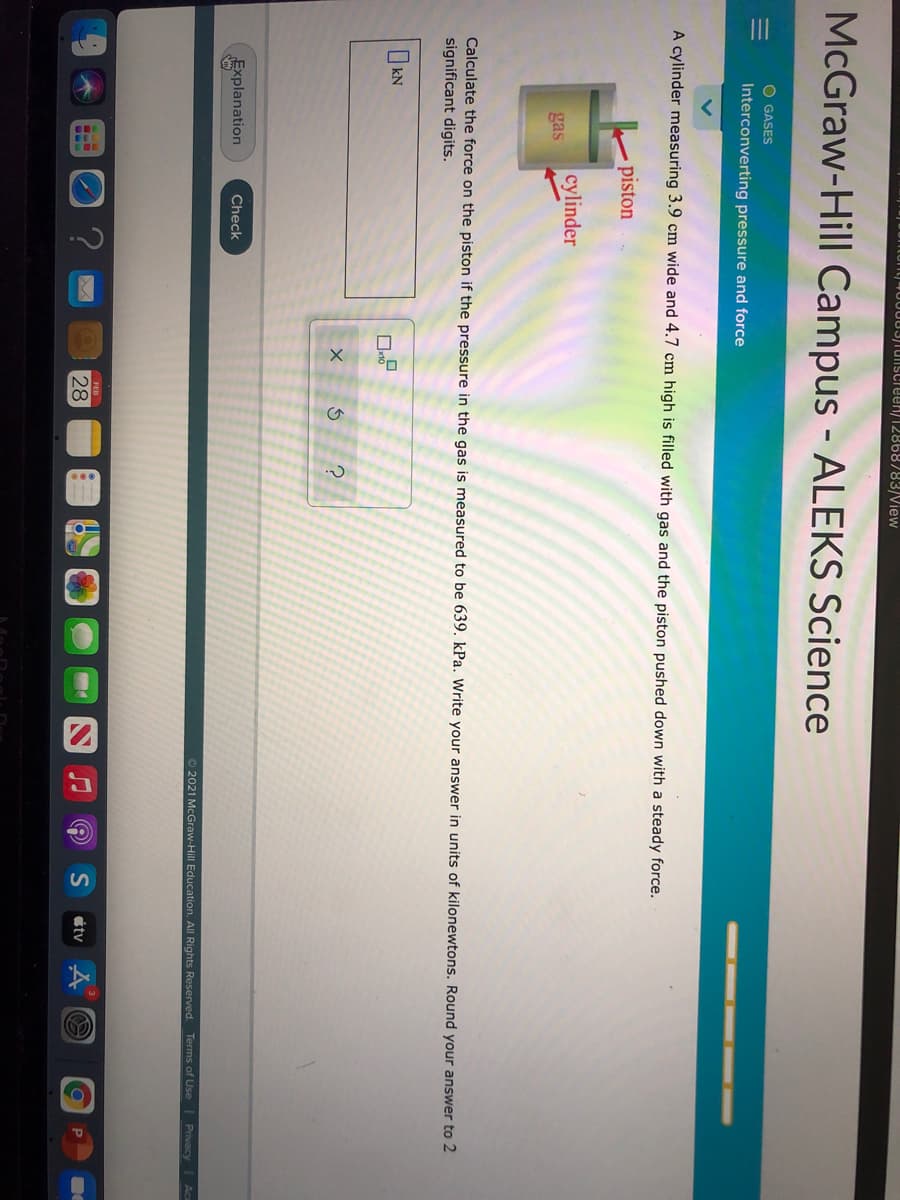
À cylinder measuring 3.9 cm wide and 4.7 cm high is filled with gas and the piston pushed down with a steady force. piston cylinder gas Calculate the force on the piston if the pressure in the gas is measured to be 639. kPa. Write your answer in units of kilonewtons. Round your answer to 2 significant digits.
À cylinder measuring 3.9 cm wide and 4.7 cm high is filled with gas and the piston pushed down with a steady force. piston cylinder gas Calculate the force on the piston if the pressure in the gas is measured to be 639. kPa. Write your answer in units of kilonewtons. Round your answer to 2 significant digits.
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter12: Thermodynamic Processes And Thermochemistry
Section: Chapter Questions
Problem 79AP
Related questions
Question
100%
Transcribed Image Text:II
uuntunyu00003/Tuliscreen/12868783/View
McGraw-Hill Campus - ALEKS Science
O GASES
Interconverting pressure and force
A cylinder measuring 3.9 cm wide and 4.7 cm high is filled with gas and the piston pushed down with a steady force.
piston
cylinder
gas
Calculate the force on the piston if the pressure in the gas is measured to be 639. kPa. Write your answer in units of kilonewtons. Round your answer to 2
significant digits.
Explanation
Check
2021 McGraw-Hill Education. All Rights Reserved. Terms of Use
Privacy Ac
28
étv
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,