A is soluble in water. Its water solution is neutral. When the solid substance is heated, brown caramel forms and water droplets are observed. A is SOLUBILITY RULES Solubilities of ionic substances in water: 1) All salts of Na*, K*, NH4* are soluble in water. 2) All salts of NO3, CH;CO0 (C2H3O2') are soluble in water. All salts of CI, Br, I' are soluble in water (except Ag*, Pb2*, Hgz?+) All salts of SO,2- are soluble in water ( except Ba2+, Pb2-, Sr²+) 3) 4) so,2- of Ca2+, Ag*, Hg22- are slightly soluble in water. 5) All salts of CO;², PO3- are insoluble in water (except Nat, K*, NH4*) 6) All salts of S2- are insoluble in water ( except Na*, K*, NH4“, Ba²+,Ca2+, Mg2+, Sr2*) 7) All salts of 02, OH" are insoluble in water ( except Na", K*, NH*, (Ba²-)) OH of Ca2+, Sr2*, (Ba²*) are slightly soluble in water OH of Mg?- is very very slightly soluble in water STRONG ACIDS STRONG BASES HCI LIOH HBr NaOH HI кон HCIO4 RbOH HNO: CSOH H2SO4 Mg(OH) 2 Ca(OH) 2 Sr(OH) 2 Ba(ОН) 2 О а. С12Н2011 (sugar) O b. NH,CI (ammoniumchloride) O . NazB407.10 H2O (borax) O d. NazSO4 (sodiumsulfate)
A is soluble in water. Its water solution is neutral. When the solid substance is heated, brown caramel forms and water droplets are observed. A is SOLUBILITY RULES Solubilities of ionic substances in water: 1) All salts of Na*, K*, NH4* are soluble in water. 2) All salts of NO3, CH;CO0 (C2H3O2') are soluble in water. All salts of CI, Br, I' are soluble in water (except Ag*, Pb2*, Hgz?+) All salts of SO,2- are soluble in water ( except Ba2+, Pb2-, Sr²+) 3) 4) so,2- of Ca2+, Ag*, Hg22- are slightly soluble in water. 5) All salts of CO;², PO3- are insoluble in water (except Nat, K*, NH4*) 6) All salts of S2- are insoluble in water ( except Na*, K*, NH4“, Ba²+,Ca2+, Mg2+, Sr2*) 7) All salts of 02, OH" are insoluble in water ( except Na", K*, NH*, (Ba²-)) OH of Ca2+, Sr2*, (Ba²*) are slightly soluble in water OH of Mg?- is very very slightly soluble in water STRONG ACIDS STRONG BASES HCI LIOH HBr NaOH HI кон HCIO4 RbOH HNO: CSOH H2SO4 Mg(OH) 2 Ca(OH) 2 Sr(OH) 2 Ba(ОН) 2 О а. С12Н2011 (sugar) O b. NH,CI (ammoniumchloride) O . NazB407.10 H2O (borax) O d. NazSO4 (sodiumsulfate)
Chemistry by OpenStax (2015-05-04)
1st Edition
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Chapter7: Chemical Bonding And Molecular Geometry
Section: Chapter Questions
Problem 75E: Element First Ionization Energy (kJImol) Second Ionization Energy (kiImol) I K 419 3050 Ca 590 1140...
Related questions
Question
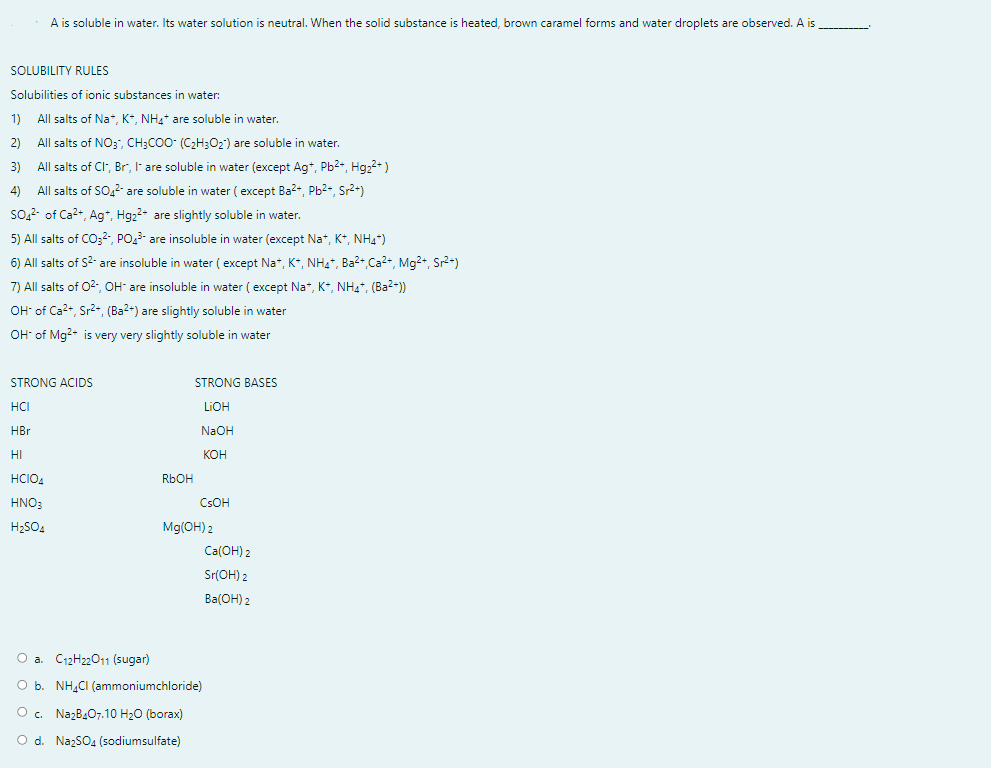
Transcribed Image Text:A is soluble in water. Its water solution is neutral. When the solid substance is heated, brown caramel forms and water droplets are observed. A is
SOLUBILITY RULES
Solubilities of ionic substances in water:
1)
All salts of Na*, K*, NH4+ are soluble in water.
2) All salts of NO3, CH3CO0- (C2H3O2) are soluble in water.
3) All salts of CI, Br, I are soluble in water (except Ag*, Pb2-, Hg2?+)
4) All salts of So,2- are soluble in water ( except Ba2-, Pb2-, Sr2+)
So.2- of Ca2+, Ag*, Hgz2- are slightly soluble in water.
5) All salts of CO;2, PO- are insoluble in water (except Na*, K*, NH4*)
6) All salts of S2- are insoluble in water ( except Na*, K*, NH, Ba2+ Ca2+, Mg2+, Sr2-)
7) All salts of 02-, OH- are insoluble in water ( except Na*, K*, NHa, (Ba2-))
OH- of Ca2+, Sr2+ (Ba2+) are slightly soluble in water
OH of Mg2* is very very slightly soluble in water
STRONG ACIDS
STRONG BASES
HCI
LIOH
HBr
NaOH
HI
КОН
HCIO4
R6OH
HNO:
CSOH
H2SO4
Mg(OH) 2
Ca(OH) 2
Sr(OH) 2
Ba(ОН) 2
O a. C12H22011 (sugar)
O b. NH,CI (ammoniumchloride)
O. NazB407.10 H20 (borax)
O d. NazSO4 (sodiumsulfate)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning


Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,