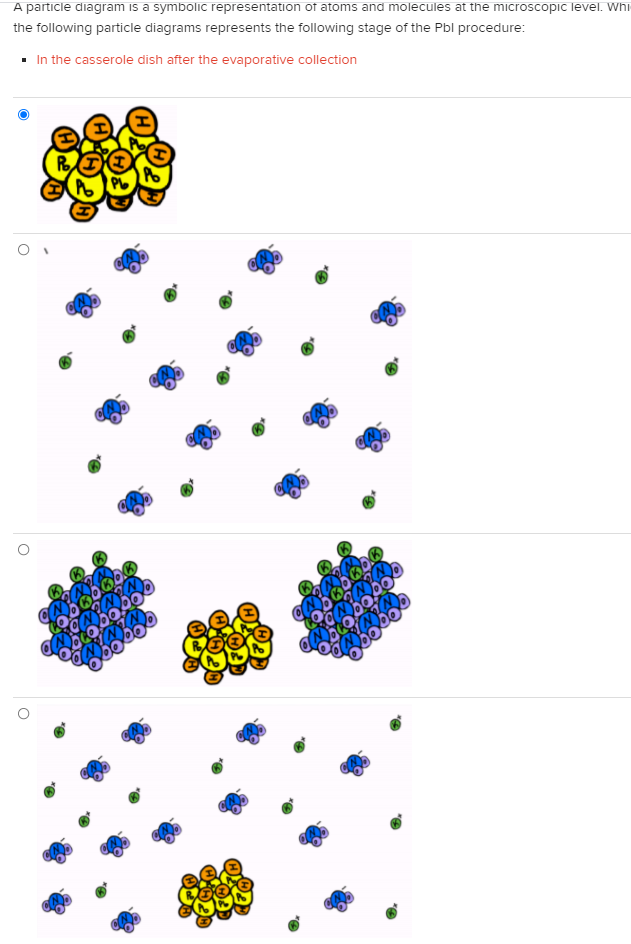
A particle diagram is a symbolic representation of atoms and molecules at the microscopic level. Whi the following particle diagrams represents the following stage of the Pbl procedure: • In the casserole dish after the evaporative collection
Q: The molar ratio of lead(II) hydroxide to water from the reaction below is 2:1 Pb(OH), + 2HCI - 2H,0…
A: • The given balanced chemical equation is, Pb(OH)2 + 2HCl → 2H2O + PbCl2 • The given…
Q: 5. Barium chloride is reacted with sodium sulfate to produce barium sulfate and sodium chloride. How…
A: Since you have asked multiple type questions, we will solve only first questions for you. If you…
Q: Preparation of HCI solution a. Measure 20.7 ml of concentrated Hcl (37% wt HCI) using the pipet and…
A:
Q: A. FERMENTATION OF CANE SUGAR 1. In a 500 mL Florence flask, place 50 gms of brown cane sugar and 1…
A: Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: Craw McGraw-Hill Education Campus Saved Ex. 1b - Mass/volume percent concentration - calculate using…
A: Volume of solution = 0.350 L Mass of KCl = 13.0 g We have to find m/v percent of solution.
Q: A tank initially contains 200 liters of fresh water. Brine containing 2.5 N/liter of dissolved salt…
A: Ans. Option (d) is right. 0.82N/L
Q: One solution states it contained a saturated 0.25302% dye by mass in ethanol solvent. By measuring…
A: % by mass refers to mass of solute present in 100 g of solution. Molarity is the moles of solute per…
Q: lution, and bubbling the resulting CO2(g) product in water that is at exactly 29 °C. After the…
A: The balanced chemical equation between Na2CO3 and HCl to produce NaCl, H2O and CO2 can be…
Q: When working with very low concentrations, such as 0.000245 M, it can be more convenient to…
A: Molarity: It is the number of moles of solute present per Litre of the solution. The units of…
Q: 500mM K2Cr2O7 solution (0.500 mol/L) About 36.775 g of K2Cr2O7 (MM = 294.20 g/mol) was weighed on an…
A: given, the weight of K2Cr2O7 taken = 36.775 g the molar mass of K2Cr2O7 = 294.20 gmol-1 molarity of…
Q: An inexperienced graduate student moved into a laboratory and began work. He needed some diethyl…
A: (a)
Q: A student performs a crystallization on an impure sample of biphenyl. The sample weighs 0.5 g and…
A: The boiling point of benzene is 80°C and melting point of biphenyl is 69.2°C. The solubility…
Q: KHP + NaOH—> KNaP +H2O Data of trial Mass of KHP : 0.8084g Initial buret containing NaOH reading:…
A:
Q: Lahen 9 5o1d is Impure, Its muting Point is higher and broader Tran the muhng unt fur pure Sampleã.…
A: When a pure substance is added with an impurities than the Properties like Melting point, boiling…
Q: For 0.250 L of aqueous solution with a density of 1.00 g/mL containing 13.7 mg of pesticide, express…
A: The concentration usually expressed in parts per million ppm and parts per billion ppb. It is…
Q: A 1.407 g sample of canned tuna was analyzed by the Kjeldahl method. The liberated NH3 required…
A:
Q: In the event of subsequent sedimentation, it must: 1- filtration quickly 2- disturbing the sediment…
A: In the process of sedimentation, the bigger particles settle down at the bottom of the container.
Q: 170 milliliters are required to prepare 900 mL of 2.00 M HCI (36.5g/mol) from the commercial reagent…
A: Concentration of commercial HCl = 37.2 % (w/w)
Q: By pipet, 15.00ml of the stock solution of potassium permanganate (KMn04) that was prepared by…
A: Mass of KMnO4 = 13.0 gm, Molar mass of KMnO4 = 158.034 gm/mol Number of moles of KMnO4 =
Q: How many kilograms (kg) of 20-20-20 soluble fertilizers should you add to a 50 gallon concentrated…
A: 20-20-20 fertilizer refers to a NPK fertilizer having N:P:K = 20:20:20.
Q: 50.00 cm3 of a 1.5784 mol.dm-3 solution of potassium hydroxide is transferred to an empty 700.00 cm3…
A: Given: The initial concentration, (C1)=1.5784 mol.dm-3 The initial volume, (V1)=50 cm3 The final…
Q: Q1/Prepare 0.1N of CH,COOH in 500 mL, Specific gravity 1.05 gm/cm and percentage 99.5% ?
A:
Q: Place the water treatment steps in the order they occur in a typical municipal water purification…
A:
Q: A 40 yo female suffering from anemia was prescribed ferrous sulfate (FESO, 7H20), to be taken one…
A: The required volume of syrup to be prepared = 8 fluid ounces. 1 fluid ounce = 6 teaspoon Total…
Q: 1 nickel (II) acetate is slowly added to 125 mL of 0.00846 M ammonium carbonate solution. What is…
A: The question is based on the concept of solubility product Principle. it states that a weak…
Q: A Hospital pharmacist wants to use three lots ZnO ointment containing repectively 50%, 20% and 5%…
A:
Q: Hydrogen peroxide solutions can easily be decomposed by dust, trace amounts of metals or light. It…
A: Given: Density of hydrogen peroxide solution = 1.11 g cm-3 = 1.11 g/mL Volume of H2O2 used for…
Q: A 0.4000g sample that assayed 96.4% Na2SO4 required 41.25 mL of a barium chloride solution.…
A:
Q: 25.0mL of a 0.515 M K2S solution is mixed with 30.0 mL of 0.833 M HNO3 acid solution to give the…
A: In this question, we have to find out the correct answer of given problem by the help of the…
Q: 1.Determine the mass of sodium chloride required to prepare 400 ml of a Mueller-Hinton broth…
A:
Q: In the Hoffman apparatus, the solution conducts electricity and water is decomposed to hydrogen at…
A: The Hoffman apparatus is used for the electrolysis of water. For electrolysis the water should be…
Q: 2. A tank contains 100 liters of brine holding 60kg of salt in solution. Water containing 1kg of…
A:
Q: A saturated solution of KNO3 was prepared at 25° Cby dissolving 12.050 g KNO3 in water and the total…
A:
Q: 2/50lved its volume (4L)His faus(oin) Nafl from sollunchloride i was related dding one liter of…
A: Given that, Initial concentration of NaCl solution = 0.02 M Volume of solution initially = 4 L One…
Q: The last calculation we need to do is find the %acetic acid (a.k.a the "acidity"). We can average…
A: The % m/v is a unit of concentration. Mass/volume percent of a solution is the ratio of the mass of…
Q: Masses of compounds used and observations NaCI CusO4-5H20 Bright blue crystaline solids…
A: Mass of the sodium Chloride used is eaqual to1. 2341
Q: 1. Volume of 0.10 M acetic acid needed for 25.0 mL of 0.020 M. mL 2. Molar mass of sodium acetate. g…
A: We’ll answer the first question since the exact one wasn’t specified. Please submit a new question…
Q: What is the concentration (in atm.) of TCE in the air at 25 C?
A:
Q: mass of eva dish+soap= 87.278 mass of evaporachingd=60.2055 moss of evadishE =67.206 soap Lab 4:…
A: After heating, the water content of the salt solution will evaporate leading to a smaller mass of…
Q: Prepared 1 M of NaCl solution in volume ?100 ml, Mwt is 58.5 0.585 gm. O 5.85 gm. O 58.5 gm. O
A: Given Data : 1. Concentration of NaCL = 1 M 2. Volume = 100 mL = 100 ÷1000 =…
Q: You need to prepare a solution of 0.123M CuSO4 by starting with solid CuSO4·5H2O. You would be able…
A: To prepare a certain concentrated solution from the solid substance, we must decide the amount of…
Q: Coming homo after the end of the winter semostor, you find that your family's swimming pool has not…
A: Solution - According to the question - Given - take whole swimming pool as a control volume. given…
Q: 19. What is the molarity of a commercial phosphoric acid solution, H3PO4 (MW= 98.0 g/mol) that is…
A:
Q: A 0.42 gram sample of acetylsalicylic acid (C₉H₈O₄, aspirin) is crushed and dissolved in water. The…
A:
Q: When a Vitamin C (ascorbic acid; MM = 176.12 g mol-1) tablet is crushed, dissolved and titrated with…
A: Given data,Molarity of KIO3=0.0340MVolume of KIO3=29.80mLMolar mass of ascorbic acid=176.12g/mol
Q: a 25.00 mL aliquot of vinegar was diluted to 250 mL in a volumetric flask. Titration of 50.00 mL…
A:
Q: How many grams of Ni(OH)2 are produced from the reaction of | 45.0 mL of a 1.60 M NAOH solution and…
A: Given, 45mL of 1.60M NaOH
Q: From the stock solutions of 1.00-mol L acetic acid and 1.00-molL of sodium acetate, six solutions…
A: Since you have asked a question with multiple subparts, we will answer only first 3 subparts for…
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

- A 19.51 ݃ sample of impure methylamine, which contains 72.58% (by mass) of CH3NH2 , isreacted with 30.81 ݃ of pure oxygen gas:4CHଷNHଶ(g) + 9Oଶ(g) ⟶ 4COଶ(g) + 10HଶO(ℓ) + 2Nଶ(g) In another experiment, this impure methylamine was used as follows: An unknown mass of the impure compound is dissolved in enough water to make 500.0 ݉ܮof solution. 20 ݉ܮ of this solution was transferred by pipette to a clean 250 mL volumetric flask andmade up to the mark. The molarity of the CH3NH2 in the final solution was determined to be 0.103 M.Determine the mass of CH3NH2 present in the original amount of impure compound used tomake this solution.25 ppm of CaCO3 was found in the water sample obtained on the lake in the nearby town.Calculate the moles of CaCO3 having a molar mass of 100.09 g/mol in 10 liters of water samplesolution having a density of 997 kg/m3. (Ans.: 24.95 moles CaCO3)In the standardization of HCl using pure anhydrous sodium carbonate as the primarystandard for methyl orange as an indicator, 1.0 mL HCl was found to be equivalent to 0.05gof sodium carbonate (MW =106). The normality of HCl is:
- A. FERMENTATION OF CANE SUGAR1. In a 500 mL Florence flask, place 50 gms of brown cane sugar and 1 gram ofdisodium phosphate.2. Dissolve 1 teaspoon of yeast in 15 mL of water. Add this to the sugar in the flasktogether with 150 mL water. Stir until the sugar is dissolved.3. Close the flask with a rubber stopper provided with a delivery tube so that theevolved gas will bubble through a solution of clear limewater.4. HAVE THE SET – UP CHECKED BY YOUR INSTRUCTOR.5. Keep the set – up inside the locker for 5 – 6 days at room temperature.Fermentation Set – up: ________________________________Write the equation for the hydrolysis of cane sugar _____________________________Write the equation for the fermentation of the simple sugars to alcohol __________6. At the end of the fermentation period, what is formed in the flask containinglimewater? _________________________________________________________________Write the equation for its formation ____________________________________________ C.…25.0mL of a 0.515 M K2S solution is mixed with 30.0 mL of 0.833 M HNO3 acid solution to give the following reaction: K2S(aq) + 2HNO3(aq) → 2KNO3(aq) + H2S(g) H2S is an unwanted by-product in a pulp and paper industry. To capture H2S gas, it is bubbled through a NaOH solution to produce Na2S with a yield of 94%. H2S(g) + 2NaOH (aq) → Na2S (aq) + 2H2O(l) The mass of H2S(g) that was processed (in kg) if 10.76 kg of Na2S was collected isA 25mL volumetric pipet is used to deliver a sample of the stock solution marked “0.6000M X2SO4” into a 100mL volumetric flask. Distilled water is added to the flask until it is about 3/4thfull, the solution is mixed well, then more water is added to fill it up to the calibration mark, then it is mixed again. Calculate the concentration of X+ion in the dilute solution made above.
- Solution and explanation for Percentage Yield and Theoritical Yield for this data (Synthesis of Aspirin) Salicylic acid - 1.950 g Labeled watch glass - 50.664 g Labeled watch glass & piece of filter paper together - 50.872 g filter paper only - 0.208 g Where the data is from: https://youtu.be/tLXljLxRZ0U Room temperature - filter paper, watch glass and aspirin - 53.088 ga 25.00 mL aliquot of vinegar was diluted to 250 mL in a volumetric flask. Titration of 50.00 mL aliquots of the diluted solution required an average of 34.88 mL of 0.09600 M NaOH. Express the acidity of the vinegar in terms of the percentage (w/v) of acetic acid (MM:60g/mol). Consider the correct significant figures of your answer and do not forget the unit.A researcher wants to determine the concentration of beta-carotene in carrots. She obtains a 0.677 M stock solution of beta-carotene and creates Dilution A by transferring 25.00 mL of stock solution into a new container and diluting it with solvent to a final volume of 94.00 mL. She creates Dilution B by transferring 50.00 mL of stock solution into a new container and diluting it with solvent to a final volume of 83.00 mL. She creates Dilution C by transferring 6.00 mL of Dilution B into a new container and diluting it with solvent to a final volume of 21.00 mL. She measures the absorbance of each solution and obtains the values below. Plot the data in a spreadsheet software and determine the best-fit line. What is the slope of the best-fit line? Report your answer with three places after the decimal. Remember to draw a dilution diagram! The best-fit line will likely look atrocious, since this is completely fictitious data. Solution Absorbance Stock solution 0.844 Dilution A…
- A researcher wants to determine the concentration of beta-carotene in carrots. She obtains a 0.677 M stock solution of beta-carotene and creates Dilution A by transferring 25.00 mL of stock solution into a new container and diluting it with solvent to a final volume of 94.00 mL. She creates Dilution B by transferring 50.00 mL of stock solution into a new container and diluting it with solvent to a final volume of 83.00 mL. She creates Dilution C by transferring 6.00 mL of Dilution B into a new container and diluting it with solvent to a final volume of 21.00 mL. She measures the absorbance of each solution and obtains the values below. Plot the data in a spreadsheet software and determine the best-fit line. What is the slope of the best-fit line? Report answer with three places after the decimal. Solution Absorbance Stock solution 0.844 Dilution A 0.140 Dilution B 0.463 Dilution C 0.223A researcher wants to determine the concentration of beta-carotene in carrots. She obtains a 0.609 M stock solution of beta-carotene and creates Dilution A by transferring 13.00 mL of stock solution into a new container and diluting it with solvent to a final volume of 96.00 mL. She creates Dilution B by transferring 52.00 mL of stock solution into a new container and diluting it with solvent to a final volume of 78.00 mL. She creates Dilution C by transferring 2.00 mL of Dilution B into a new container and diluting it with solvent to a final volume of 21.00 mL. What is the concentration in mM of Dilution C? Report your answer with one place after the decimal.A researcher wants to determine the concentration of beta-carotene in carrots. She obtains a 0.661 M stock solution of beta-carotene and creates Dilution A by transferring 26.00 mL of stock solution into a new container and diluting it with solvent to a final volume of 94.00 mL. She creates Dilution B by transferring 43.00 mL of stock solution into a new container and diluting it with solvent to a final volume of 75.00 mL. She creates Dilution C by transferring 6.00 mL of Dilution B into a new container and diluting it with solvent to a final volume of 15.00 mL. What is the concentration in mM of Dilution C?





