A simple paper chromatography experiment involved the analysis of the food dyes in lollipop samples. A chocolate-flavored lollipop was dissolved using various solvents and was spotted in the chromatogram (E). Four food dye standards (blue, green, red, and orange) were tested alongside the chocolate-flavored lollipop. The mobile phase used for the analysis was 0.1% NaCl. After the analysis, the resulting chromatogram revealed the separation of three colors, which were attributed to the food dyes indigo carmine (blue), tartrazine (yellow), and erythrosine (red). 10 Ruler is in cm unit A B E A E BEFORE AFTER Calculate the rf for each dye. Express answers in 3SF. erythrosine: indigocarmine: tartrazine: Which dye is the most polar? Choose.. Which dye is the least polar? Choose...
A simple paper chromatography experiment involved the analysis of the food dyes in lollipop samples. A chocolate-flavored lollipop was dissolved using various solvents and was spotted in the chromatogram (E). Four food dye standards (blue, green, red, and orange) were tested alongside the chocolate-flavored lollipop. The mobile phase used for the analysis was 0.1% NaCl. After the analysis, the resulting chromatogram revealed the separation of three colors, which were attributed to the food dyes indigo carmine (blue), tartrazine (yellow), and erythrosine (red). 10 Ruler is in cm unit A B E A E BEFORE AFTER Calculate the rf for each dye. Express answers in 3SF. erythrosine: indigocarmine: tartrazine: Which dye is the most polar? Choose.. Which dye is the least polar? Choose...
Chapter14: Chromatography
Section: Chapter Questions
Problem 9P
Related questions
Question
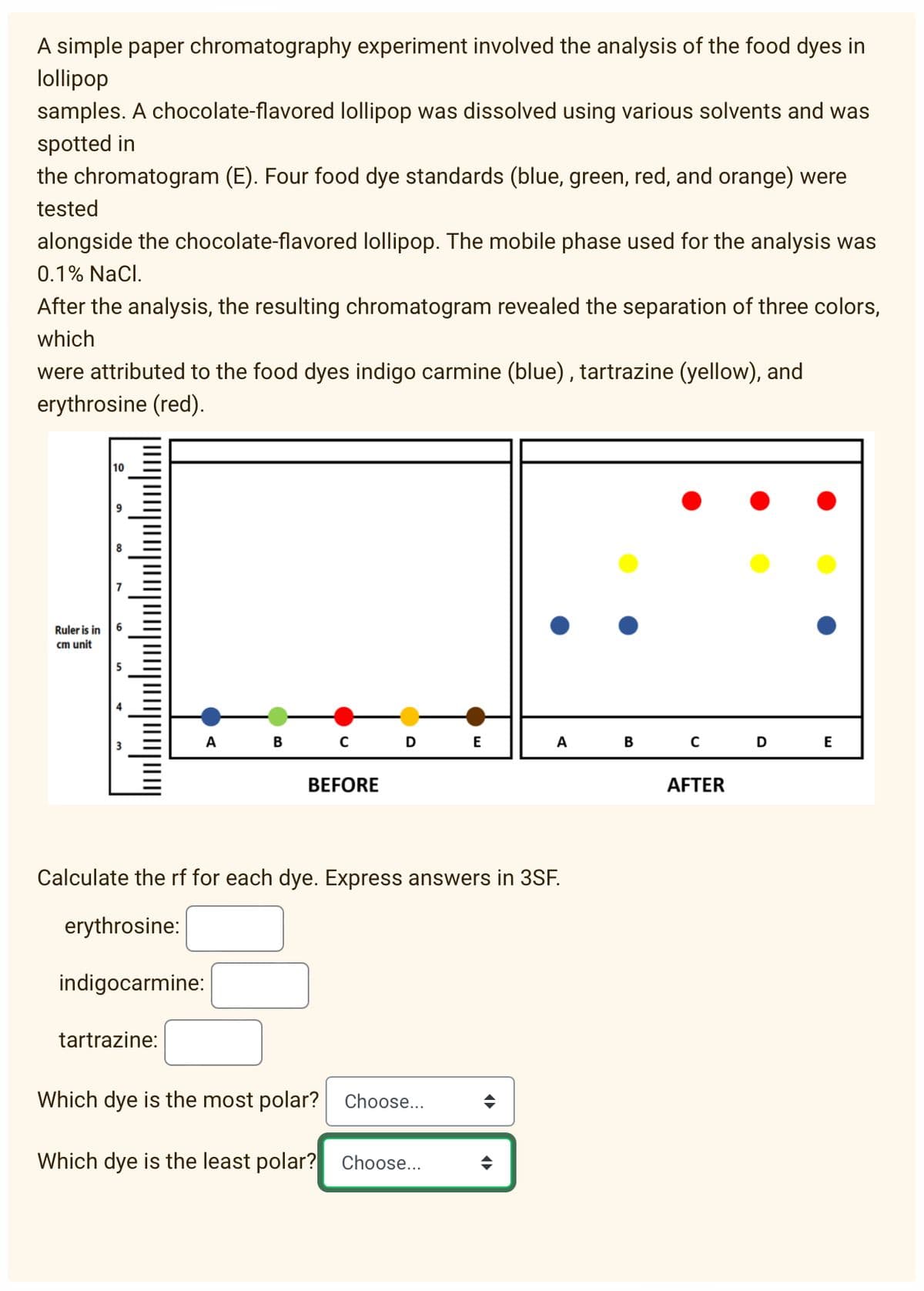
Transcribed Image Text:A simple paper chromatography experiment involved the analysis of the food dyes in
lollipop
samples. A chocolate-flavored lollipop was dissolved using various solvents and was
spotted in
the chromatogram (E). Four food dye standards (blue, green, red, and orange) were
tested
alongside the chocolate-flavored lollipop. The mobile phase used for the analysis was
0.1% NaCl.
After the analysis, the resulting chromatogram revealed the separation of three colors,
which
were attributed to the food dyes indigo carmine (blue), tartrazine (yellow), and
erythrosine (red).
10
Ruler is in
cm unit
в с
B C D
A
E
A
E
BEFORE
AFTER
Calculate the rf for each dye. Express answers in 3SF.
erythrosine:
indigocarmine:
tartrazine:
Which dye is the most polar? Choose..
Which dye is the least polar? Choose...
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you



Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning



Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole