A solution of oxalic acid dihydrate (H:C2O4-2H:O) with a known concentration of 0.400 M H:C2O4-2H20 is titrated with a 0.333 M NaOH solution. How many L NaOH are required to reach the second equivalence point with a starting volume of 65.0 mL H:C2O4-2H2O , according to the following balanced chemical equation: H:C:O4 2H:0 + 2 NaOH – Na:C:O4 + 4 H:O mol H.GO. ZH:O 65.0 mL H:C2O4-2H2O 2 mol H:C2O4 2H:0 0.156 L NAOH LH:C:04-2H:0 1 molH.GO. ZH:O 0.333 mol NaOH -O.-2H:0 65.0 mL H:C:0.-2H:0 2 mol H:C:O.-2H:0 = 0.156 L NaOH 2H:0 1 mol H:C:O.-2H:0 0.333 mol NaOH ADD FACTOR DELETE ANSWER RESET *( ) 0.400 0.156 2 0.001 156 7.81 x 10 0.156 0.0781 0.333 4 39.0 78.1 1.56 x 10 1000 0.0520 5.20 x 10° 65.0 mol H:C204-2H:O g NaOH mL NaOH MH:C2O+-2H2O g H:C2O+-2H2O mol NaOH L NaOH M NaOH L H:C:O.-2H:O mL H:C2O4-2H:0
A solution of oxalic acid dihydrate (H:C2O4-2H:O) with a known concentration of 0.400 M H:C2O4-2H20 is titrated with a 0.333 M NaOH solution. How many L NaOH are required to reach the second equivalence point with a starting volume of 65.0 mL H:C2O4-2H2O , according to the following balanced chemical equation: H:C:O4 2H:0 + 2 NaOH – Na:C:O4 + 4 H:O mol H.GO. ZH:O 65.0 mL H:C2O4-2H2O 2 mol H:C2O4 2H:0 0.156 L NAOH LH:C:04-2H:0 1 molH.GO. ZH:O 0.333 mol NaOH -O.-2H:0 65.0 mL H:C:0.-2H:0 2 mol H:C:O.-2H:0 = 0.156 L NaOH 2H:0 1 mol H:C:O.-2H:0 0.333 mol NaOH ADD FACTOR DELETE ANSWER RESET *( ) 0.400 0.156 2 0.001 156 7.81 x 10 0.156 0.0781 0.333 4 39.0 78.1 1.56 x 10 1000 0.0520 5.20 x 10° 65.0 mol H:C204-2H:O g NaOH mL NaOH MH:C2O+-2H2O g H:C2O+-2H2O mol NaOH L NaOH M NaOH L H:C:O.-2H:O mL H:C2O4-2H:0
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter14: Equilibria In Acid-base Solutions
Section: Chapter Questions
Problem 47QAP: A 0.4000 M solution of nitric acid is used to titrate 50.00 mL of 0.237 M barium hydroxide. (Assume...
Related questions
Question
I cant figure out what Im doing wrong
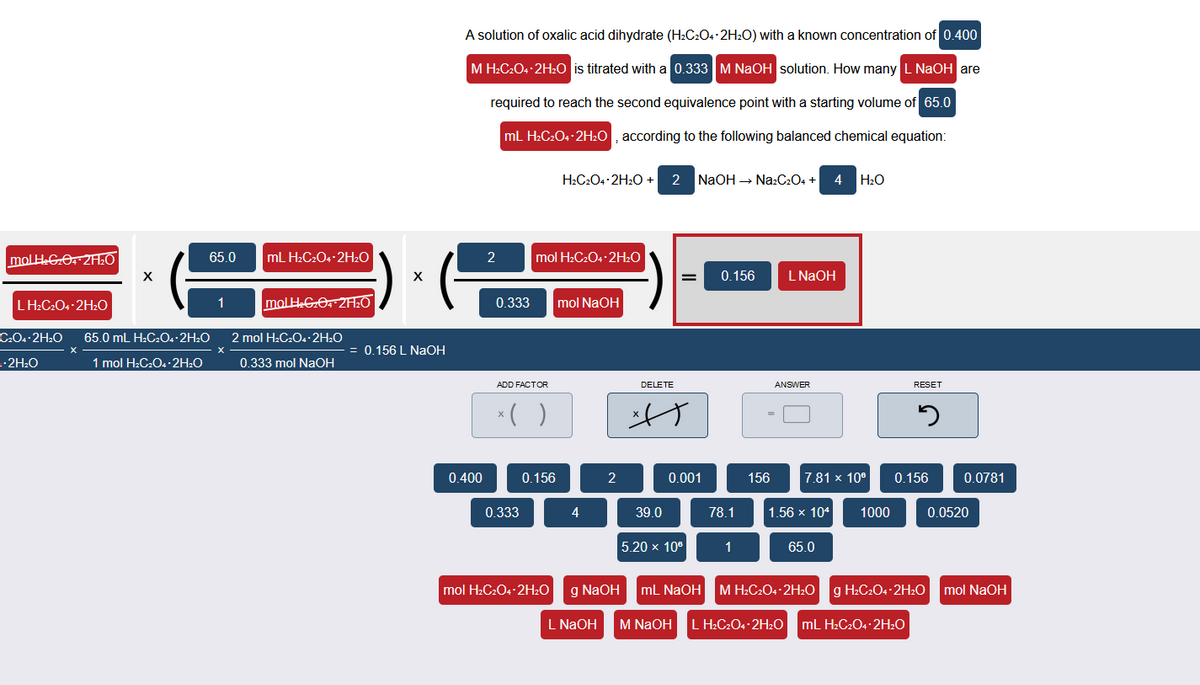
Transcribed Image Text:A solution of oxalic acid dihydrate (H2C2O4-2H2O) with a known concentration of 0.400
M H:C2O4 2H20 is titrated with a 0.333 M NAOH solution. How many L NaOH are
required to reach the second equivalence point with a starting volume of 65.0
mL H2C2O4-2H2O , according to the following balanced chemical equation:
H:C2O4 2H2O + 2 NaOH → Na:C204 +
4
H2O
molH GO 2A:0
65.0
mL H2C2O4-2H2O
2
mol H2C2O4·2H2O
0.156
L NAOH
LH:C204-2H2O
molHGO 2A:O
0.333
mol NaOH
C.Os 2H2O
65.0 mL H2C2O. 2H2O
2 mol H:C2O4·2H2O
= 0.156 L NAOH
-2H:O
1 mol H:C2O4 •2H:O
0.333 mol NaOH
ADD FACTOR
DELETE
ANSWER
RESET
*( )
げ
0.400
0.156
2
0.001
156
7.81 x 10°
0.156
0.0781
0.333
4
39.0
78.1
1.56 x 104
1000
0.0520
5.20 x 10°
1
65.0
mol H2C2O4-2H2O
g NaOH
mL NaOH
M H2C2O4 2H2O
g H2C2O4-2H2O mol NaOH
L NAOH
M NaOH
L H:C2O4-2H20
mL H2C2O4 2H2O
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemical Principles in the Laboratory
Chemistry
ISBN:
9781305264434
Author:
Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:
Brooks Cole

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning