A student must make a buffer solution with a pH of 7.00.7.00. Determine which weak acid is the best option to make a buffer at the specified pH. acetic acid, ?a=1.75×10−5,�a=1.75×10−5, 5.00 M propionic acid, ?a=1.34×10−5,�a=1.34×10−5, 3.00 M ammonium citrate, ?a=4.06×10−7,�a=4.06×10−7, 2.00 M sodium dihydrogen phosphate monohydrate, ?a=6.23×10−8,�a=6.23×10−8, 2.00 M Determine which conjugate base is the best option to make a buffer at the specified pH. sodium acetate trihydrate, CH3COONa⋅3H2OCH3COONa⋅3H2O disodium hydrogen phosphate heptahydrate, Na2HPO4⋅7H2ONa2HPO4⋅7H2O sodium propionate, CH3CH2COONaCH3CH2COONa sodium citrate dihydrate, C6H5O7Na3⋅2H2OC6H5O7Na3⋅2H2O The final volume of buffer solution must be 100.00 mL and the final concentration of the weak acid must be 0.100 M. Based on this information, what mass of solid conjugate base should the student weigh out to make the buffer solution with a pH of 7.00? mass = g Based on this information, what volume of acid should the student measure to make the buffer solution? volume = mL
A student must make a buffer solution with a pH of 7.00.7.00. Determine which weak acid is the best option to make a buffer at the specified pH. acetic acid, ?a=1.75×10−5,�a=1.75×10−5, 5.00 M propionic acid, ?a=1.34×10−5,�a=1.34×10−5, 3.00 M ammonium citrate, ?a=4.06×10−7,�a=4.06×10−7, 2.00 M sodium dihydrogen phosphate monohydrate, ?a=6.23×10−8,�a=6.23×10−8, 2.00 M Determine which conjugate base is the best option to make a buffer at the specified pH. sodium acetate trihydrate, CH3COONa⋅3H2OCH3COONa⋅3H2O disodium hydrogen phosphate heptahydrate, Na2HPO4⋅7H2ONa2HPO4⋅7H2O sodium propionate, CH3CH2COONaCH3CH2COONa sodium citrate dihydrate, C6H5O7Na3⋅2H2OC6H5O7Na3⋅2H2O The final volume of buffer solution must be 100.00 mL and the final concentration of the weak acid must be 0.100 M. Based on this information, what mass of solid conjugate base should the student weigh out to make the buffer solution with a pH of 7.00? mass = g Based on this information, what volume of acid should the student measure to make the buffer solution? volume = mL
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter14: Equilibria In Acid-base Solutions
Section: Chapter Questions
Problem 25QAP: A sodium hydrogen carbonate-sodium carbonate buffer is to be prepared with a pH of 9.40. (a) What...
Related questions
Question
A student must make a buffer solution with a pH of 7.00.7.00.
Determine which weak acid is the best option to make a buffer at the specified pH.
acetic acid, ?a=1.75×10−5,�a=1.75×10−5, 5.00 M
propionic acid, ?a=1.34×10−5,�a=1.34×10−5, 3.00 M
ammonium citrate, ?a=4.06×10−7,�a=4.06×10−7, 2.00 M
sodium dihydrogen phosphate monohydrate, ?a=6.23×10−8,�a=6.23×10−8, 2.00 M
Determine which conjugate base is the best option to make a buffer at the specified pH.
sodium acetate trihydrate, CH3COONa⋅3H2OCH3COONa⋅3H2O
disodium hydrogen phosphate heptahydrate, Na2HPO4⋅7H2ONa2HPO4⋅7H2O
sodium propionate, CH3CH2COONaCH3CH2COONa
sodium citrate dihydrate, C6H5O7Na3⋅2H2OC6H5O7Na3⋅2H2O
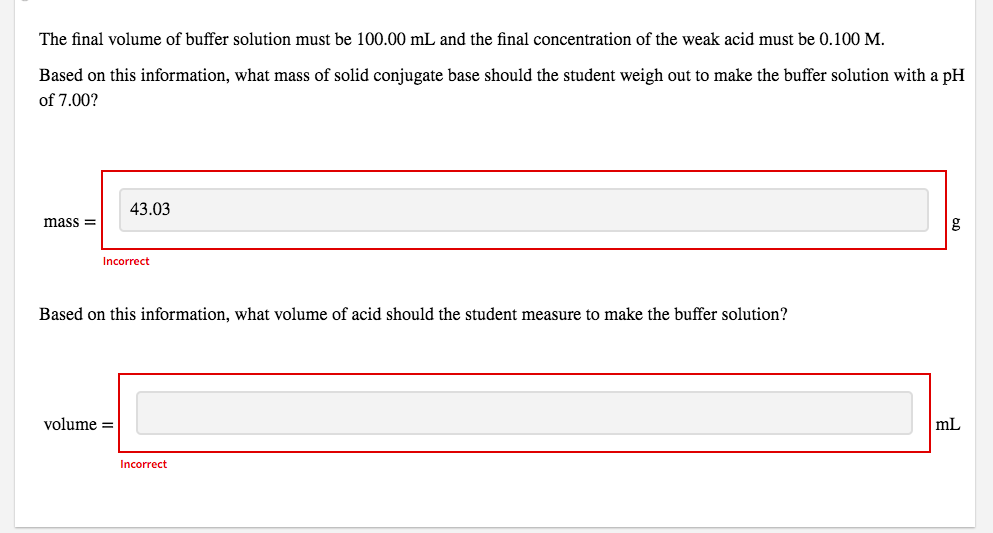
The final volume of buffer solution must be 100.00 mL and the final concentration of the weak acid must be 0.100 M.
Based on this information, what mass of solid conjugate base should the student weigh out to make the buffer solution with a pH of 7.00?
mass = g
Based on this information, what volume of acid should the student measure to make the buffer solution?
volume = mL

Transcribed Image Text:O Macmillan Learning
A student must make a buffer solution with a pH of 7.00.
Determine which weak acid is the best option to make a buffer at the specified pH.
O O
acetic acid, K₁ = 1.75 x 10-5, 5.00 M
O
propionic acid, K₁ = 1.34 x 10-5, 3.00 M
O ammonium citrate, K₁ = 4.06 x 10-7, 2.00 M
● sodium dihydrogen phosphate monohydrate, K₂ = 6.23 × 10-8, 2.00 M
Determine which conjugate base is the best option to make a buffer at the specified pH.
sodium acetate trihydrate, CH3COONa. 3 H₂O
● disodium hydrogen phosphate heptahydrate, Na₂HPO4.7H₂O
O sodium propionate, CH₂CH₂COONa
O sodium citrate dihydrate, C,H,O,Na, .2H₂O
The final volume of buffer solution must be 100.00 mL and the final concentration of the weak acid must be 0.100 M.
Based on this information, what mass of solid conjugate base should the student weigh out to make the buffer solution with a pH
of 7.00?
Transcribed Image Text:The final volume of buffer solution must be 100.00 mL and the final concentration of the weak acid must be 0.100
Based on this information, what mass of solid conjugate base should the student weigh out to make the buffer solution with a pH
of 7.00?
mass=
43.03
Incorrect
Based on this information, what volume of acid should the student measure to make the buffer solution?
volume =
Incorrect
g
mL
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning