Q: fat undergoes: H₂C-O HC-0 H₂C-0 Refer to the fat structure below. Draw the possible products if the…
A:
Q: Above: FTIR of L-menthol. 3500 3000 2500 Wavenumber cm-1 2000 1500 1000 500 3246.67 2953.97 2925.63…
A: IR spectra provides the information of functional groups of unknown compound based on the…
Q: 2. On the basis of charge stability, rank species A-G from weakest base to strongest base. ia NO HO.…
A:
Q: Write the products and balance the following reac- tion for the complete combustion of butane,…
A: Since, Balanced reaction means that both side number of atom present in equal number. Thus,
Q: Provide IUPAC name for the structure shown:
A:
Q: Draw the major product and the mechanism. d. e. f. g. Br Hill میں -CF3 NaOH₂ Br H₂O heat Br Na H₂…
A:
Q: 140.0-g sample of water at 25.0°C is mixed with 126.1 g of a certain metal at 100.0°C. After thermal…
A:
Q: Give typed explanation not a single word hand written otherwise leave it Determine if the…
A: Salts that are formed from strong acid and strong base are neutral salts. Salts that are formed from…
Q: give the product of the reaction C- H₂C CH3 (H') xs ethanol
A: Ketone is protected by forming the ketal. Formation of ketal is the acid catalyzed reaction.
Q: 5. Calculate the concentration of Pb in a solution saturated with PbSO,. Ksp for PbSO, is 2 x 10%.…
A: Given Ksp = 2 x 10-7
Q: 1. Keeping in mind the definition of a buffer, prec Start with 50 mL of buffer at pH 9.00. Add b.…
A: Solutions- Buffer- It is defines as the buffer solution is a solution able to maintain its Hydrogen…
Q: 100 90- 80 70- 60 50- 40 30 20 12+ 4000 3500 3000 2843 96 2500 cm-1 1749.89cm-1 1680.33cm-1 2000…
A: The question is based on IR spectroscopy. we need to analyse and interpret the spectra.
Q: A solution is prepared by mixing 455 mL of 0.400 M NH 3 and 280 mL of 0.400 M HCl. Assuming that the…
A:
Q: 22-90. Calculate the equilibrium mercury(I) ion con- centration in a solution made by mixing 100.0…
A: Equilibrium concentration of Hg + ion is.
Q: QUESTION 5 Energy that is associated with the position or composition of an object is called…
A: Energy that is associated with position or composition of an object is called potential energy .…
Q: In a reaction where a carboxylic ester and water react to produce an alcohol and carboxylate (or…
A: In a reaction where a carboxylic ester and water react to produce an alcohol and carboxylate (or…
Q: Macmillan Learning Given the information A+B 2D D C calculate AG at 298 K for the reaction A+B 2C…
A: The relationship between Gibbs free energy, Enthalpy and entropy change is ΔG=ΔH−TΔS “Since you…
Q: The pH of 300 mL solution made of 0.58 M acetic acid and 0.66 M potassium acetate is (Ka = 1.8 x…
A:
Q: Hydrogen peroxide decomposes to water and oxygen at constant pressure by the following reaction:…
A: Given, decomposition reaction, The decomposition reaction of hydrogen peroxide is: 2H2O2(l)→2H2O…
Q: What is the hydroxide ion concentration of a solution prepared by dissolving 0.658 g of Mg(OH)2 in…
A: Given that - Mass of magnesium hydroxide Mg(OH)2 = 0.658 g Volume of solution (of water) = 333 mL…
Q: What is the pH of a 1.25M solution of nitrous acid if the Ka is 7.2 ×10^-4?
A:
Q: What is the coefficient for O2 when it is balanced? C6H5CH3(I) + O2(g) -> CO2(g) + H2O(g)
A:
Q: Draw a structural formula for the major product of the acid-base reaction shown. 4 H3C-N (1 mole) +…
A: An acid-base reaction is a chemical reaction that occurs between an acid and a base. In this…
Q: What is the major product of the following sequence of reactions? 1. NaOEt, EtOH 2. H₂O+ 3. 0= Br 4.…
A:
Q: Oxygen gas is bubbled through an acidified solution of Fe2+ No reaction H2O Fe3+ Cl2 gas…
A:
Q: Can you help me with how to do the states of the products (solid, aqueous, or gas) for the periodic…
A: Here are some guidelines to help you determine the states of the products in a chemical reaction:
Q: (h) H C=PPh3 +
A: Wittig reaction: It is the reaction between a Carbonyl compound and phosphorus ylide.
Q: If the reaction A + B + C --> Products, follows the rate law of rate= 5.05M-5s-2[A]²[C] what is the…
A:
Q: [References The mass spectrum of compound A shows the molecular lon at m/z 85, an M + 1 peak at m/z…
A: The question is based on the concept of organic spectroscopy. we need to analyse the spectral data…
Q: Decide whether the Lewis structure proposed for each molecule is reasonable or not. Note: If two or…
A: We have to check if the given structures are reasonable or not.
Q: Which of these has the most acidic proton?
A: Identify the most acidic proton in the given organic compounds.
Q: Draw the structure of the compound that is consistent with the ¹H NMR. (Assume that long-range…
A: Question is Based on the concept of organic spectroscopy. We need to analyse the spectre and…
Q: What is the conjugate base of [Fe (H2O)6]^3+ (aq
A: According to Bronsted and Lowry concept an Acid is a substance that has the tendency to donate a…
Q: Two sets of ionizations are shown in the tables below. Complete the tables by ordering each set of…
A: Ionization energy is the amount of energy required to remove an electron from an isolated gaseous…
Q: Determine the intermolecular forces for each of the following molecules: Compound N₂ CH4 CH3CI H₂O…
A: We have to fill the given table using Lewis structures and determine the intermolecular forces
Q: A solution contains 1.16x10-2 M potassium carbonate and 1.36x10-2 M potassium sulfide. Solid copper…
A: Answer: A sparingly soluble salt begins to precipitate when its ionic product becomes equal to its…
Q: g. h. Since the concentration of the H2O2 solution is 3% by mass, calculate the mass of H₂O2 in the…
A:
Q: 1 Consider a 1.0 M HNO2 solution. Part A: What is the pH of this solution? Hint: The K₂ of HNO2 is…
A: The question is based on the concept of chemical equilibrium. we need to calculate percentage degree…
Q: 1) Consider the following: H₂S (aq) + CO3² (aq) HCO3(aq) + HS (aq) a. Identify the acid, base,…
A: An acid is a species which donates Proton while a base accepts proton. an acid after donating…
Q: In a study of the decomposition of ammonia on a tungsten surface at 1100 °C NH3 1/2 N₂ + 3/2 H₂ the…
A:
Q: t takes 614. kJ/mol to break a carbon-carbon double bond. Calculate the maximum wavelength of light…
A: Answer: Relation between wavelength of light and energy of photon is shown below: E=hCλ Here:…
Q: The answer is 6.1 x 10^-2 but I'm not sure how to solve this problem. Please include every step.…
A: For any reaction: aA+bB→cC+dD The rate law will be: rate=kAxBy Here x and y are the orders with…
Q: Which of the following are NOT amphoteric? Select all that apply. PO43- H₂PO4 H₂O OH H3O+ HCO3-…
A:
Q: Macmillan Learning Use the van der Waals equation of state to calculate the pressure of 2.80 mol of…
A: The van der Waals equation of state for real gases is given by, P+an2V2V-nb=nRTwhere,P=Pressure…
Q: Identify the product for this reaction. excess H₂ /Rd.C 1 atm/25°C
A:
Q: A stock solution whose concentration is 25.00 mg/mL was provided. A primary standard solution was…
A: Given that, The initial concentration is C1 = 25.00 mg/mL. The initial volume is V1 = 5 mL. The…
Q: remain constant? FeNCS2+ comple Fe³+ ions SCN ions 2. If the KEQ for the formation of FeNCS²(aq) is…
A:
Q: Can you give 3 typical job functions of an analytical chemist?
A: An analytical chemist is a professional who specializes in the identification, separation, and…
Q: For the reaction below, Kc = 4.60 × 10⁻⁶. Note Kc is sometimes called K. What is the equilibrium…
A: Chemical equilibrium is defined as the stage at which both reactant and product are in equilibrium.…
Q: A 12.7 mL solution of 0.100 mol L-1 HOCI is titrated using 0.150 mol L-1 NaOH. What is the pH of the…
A:
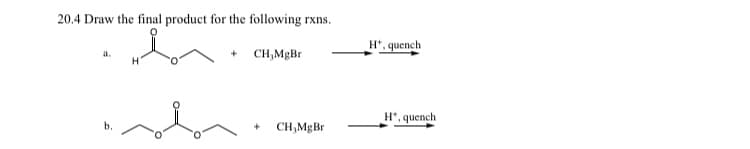
Draw final product
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 3 images

- What is the best reason for why nitriles do not undergo overaddition with Grignard reagents? A the nitriles are sp hybridized b the metalloimine intermediate is not a good electrophile c This isn't true, nitriles do undergo overaddition d Grignard reagents aren't nucleophilic enough to perform overaddition on any electrophileIs Methylamine polar protic or aprotic?Draw the Configuration of the D-Family of Aldoses in a short bond paper (fit in 1 page only) Label properly, thank you!
- Why is an alkylamine more basic than ammononia?Devise a synthesis of the attached compound from starting materials havingtwo or fewer carbons.Explain why the pKa of a hydrogen bonded to the sp3 carbon of propene is greater (pKa = 42) than that of any of the carbon acids but is less than the pKa of an alkane (pKa > 60).

