A www-awn.aleks.com/alekscgi/x/lsl.exe/10_u-IgNslkr7j8P3jH-lijkPWvZoZLqKt1FLIq7wcPWKzBYGfE9IMFJTfOEia672gexleiXZ9Ke2X7cLj01purJIM7lvRgOUkyU.. O ATOMS, IONS AND MOLECULES Predicting ionic compounds formed by two elements Decide whether each pair of elements in the table below will form an ionic compound. If they will, write the empirical formula of the compound formed in the space provided. Forms ionic compound? empirical formula of ionic compound element #1 element #2 sulfur lithium O yes O no fluorine sulfur O yes O no magnesium sulfur O yes O no bromine lithium O yes O no II
A www-awn.aleks.com/alekscgi/x/lsl.exe/10_u-IgNslkr7j8P3jH-lijkPWvZoZLqKt1FLIq7wcPWKzBYGfE9IMFJTfOEia672gexleiXZ9Ke2X7cLj01purJIM7lvRgOUkyU.. O ATOMS, IONS AND MOLECULES Predicting ionic compounds formed by two elements Decide whether each pair of elements in the table below will form an ionic compound. If they will, write the empirical formula of the compound formed in the space provided. Forms ionic compound? empirical formula of ionic compound element #1 element #2 sulfur lithium O yes O no fluorine sulfur O yes O no magnesium sulfur O yes O no bromine lithium O yes O no II
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter6: Quantum Mechanics And Molecular Structure
Section: Chapter Questions
Problem 68AP: The Be2 molecule has been detected experimentally. It has a bond length of 2.45 Å and a bond...
Related questions
Question
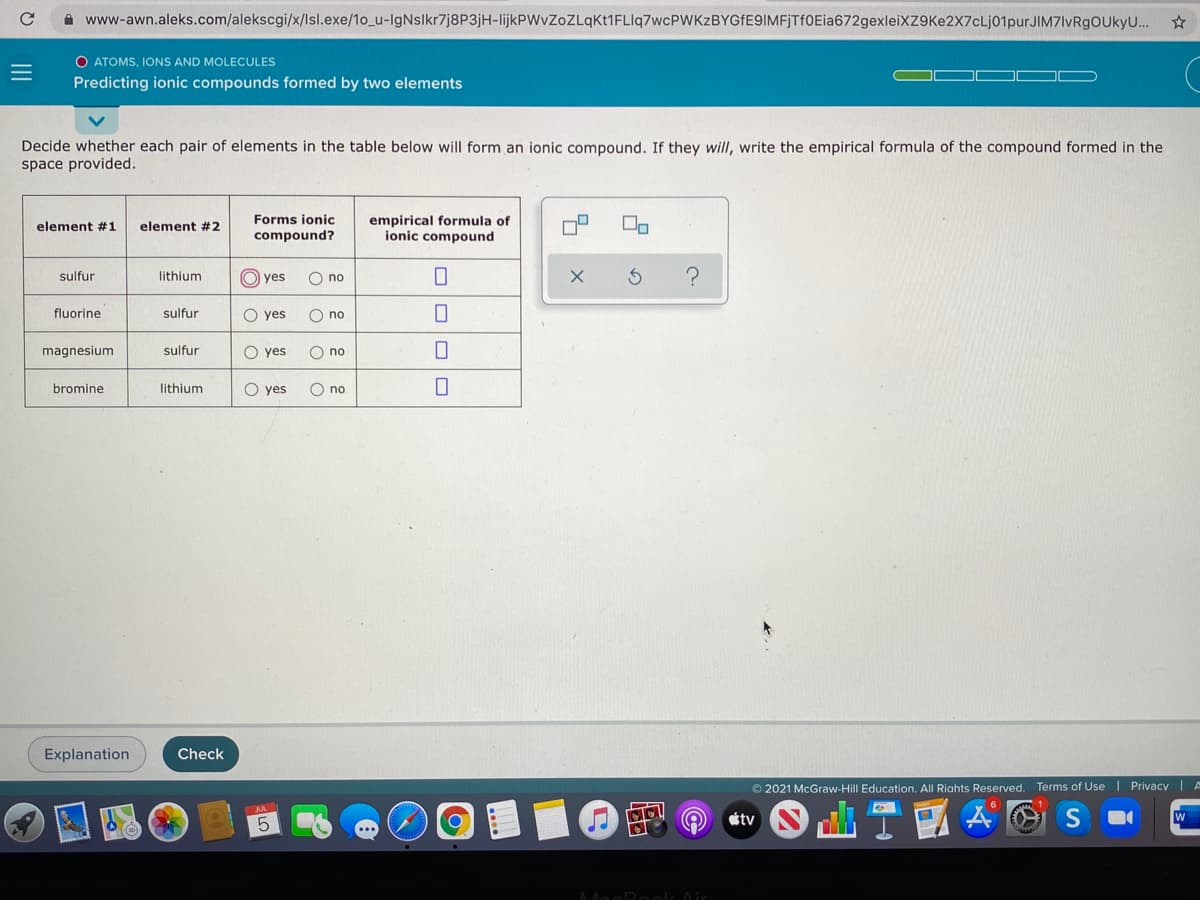
Transcribed Image Text:A www-awn.aleks.com/alekscgi/x/lsl.exe/10_u-IgNslkr7j8P3jH-lijkPWvZoZLqKt1FLIq7wcPWKzBYGfE9IMFjTf0Eia672gexleiXZ9Ke2X7cLj01purJIM7lvRgOUkyU.
O ATOMS, IONS AND MOLECULES
Predicting ionic compounds formed by two elements
Decide whether each pair of elements in the table below will form an ionic compound. If they will, write the empirical formula of the compound formed in the
space provided.
Forms ionic
empirical formula of
ionic compound
element #1
element #2
compound?
sulfur
lithium
O yes
O no
fluorine
sulfur
O yes
O no
magnesium
sulfur
O yes
O no
bromine
lithium
O yes
O no
Explanation
Check
© 2021 McGraw-Hill Education. All Riahts Reserved. Terms of Use I Privacy A
étv
S
MooRool Air
II
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 5 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,


Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,
