Q: CH3 0 -H Br HB: CH, --C-CH-CH3 → (CH3)3 C-CE- -CH, A H20 CH3 Br (CH) ҫ - сн - сн, + (CH):С — сH…
A:
Q: 436 kJ/mol 431 kJ/mol 567 kJ/mol 413 kJ/mol 348 kJ/mol 293 kJ/mol C=C 614 kJ/mol H-H H-CI C-H C-C…
A:
Q: a) -СCH-CH-OH,-Br,-H, -CH-CHз b) -СООН, СООCH3,-CH-ОН,-ОН с) -СH-NHCH3,-CH-NHz-NHz,-CN
A: Answer:- This question is solved by using the simple concept of determination of priority order…
Q: Each of the following reactions has been reported in the chemical literature and proceeds in good…
A: Note: Since you have posted a question with multiple subparts, we will solve the first three sub…
Q: Name the following:
A: b. (2S,3S)-2-bromo-3-chloropentane d. (2R,3R)-2,3-dichloropentane
Q: CH;-CH,-CH, CH,-CH3 1. CH3-CH-CH,- сH - CH, CH3 CH3 CH3 2. CH,- с - сH,-CH - CH-CH, CH3
A: Write the IUPAC NAME of the given structure-
Q: HC=CCH,CH, CH,CH=CHCH, CH3CH2CH2CH3 С. d. HCI, H2O, H2S e. CH3CH2CH3, CICH2CH2OH, CH3CH2OH
A:
Q: Br HC-CH-CH-č-OH 3. H,C-C-CH-CHo 4. Hy C-CH-CH-OH CI 2.
A: 1.The 1st compound contian -o-,this is ether group 2.second compound contain both halogen and acid…
Q: 1. NaBH4 (е) CH,=CH-CH,--CH3 2. Но 1. (CH;),СHCH2-AlН-СH2CH(CH3)2, -78°C (f) -CH2-C-OCH3 2. H.О
A:
Q: + 2H2 H20 > B Na H3C-C: EC-CH3 + 1H2 A + Cl2 NH3(1) A H20 → B H2SO4 -c=c-CH3 + H2 Lindlar H,O H3C-C…
A: Hydrogenation is process of adding hydrogen across the unsaturated bond
Q: 6. 8. H3C-C=C-CH-CH-CH3 CH3 KM10, H;C-C=c-CH2-CH-CH3 2 mol Br2 ČH3 9.
A:
Q: Question attached
A: In the presence of strong acids, such as sulfuric acid and heat, alcohol dehydrates to an alkene.…
Q: HgSO4 H2SO4, H2O 1T (1) Br Вн, THF H,Oг, NaOH 10 NANH2 H =H 1S NH3 H20 HCI (1 equiv.) diethyl ether…
A: NaNH2 abstracts the acidic hydrogen from alkyne which reacts with alkyl halide to gives alkyne (1S).…
Q: Br Br- -CH-CH;, НО -H ОН CH2CH3 ŅH2 CH CH3 F- -CH; NH2 Cl キ, F НF Cl- -H H2N. Cl H2N- -CH3 НС Н H
A: Explanation- 1) Disteriomers- having different configuration around the more than one carbon atom .…
Q: a. H3C-CH2-NH-C-H IUPAC H3C-CH2-NH-ĉ-H Common H3C-CH-CH2-CH2-CH2-C-0-CH3 b. Br IUPAC…
A: (a) IUPAC name : N-Ethylmethanamide Common name : N-Ethyl formamide (b) IUPAC name :…
Q: How are the Fischer projection formulas in each pair related to each other? Are they identical or…
A: Enantiomers are those organic compounds which are non superimposable mirror images of each other.…
Q: Draw the organic products formed in each reaction.
A: Hello. Since the question consists of multiple sub-parts, the first sub-part shall be only solved.…
Q: 1. NaBH4 (е) CH2=CH-CH2-C-CH3 2. Нао 1. (CH3)2СHCH2-AIН-CH2CH(CHз)2, -78°C (f) -CH2-C-OCH3 2. Н,О
A: NaBH4 is mild reducing agent . It converts aldehyde and ketone compound to alcohol compound. DIBAL…
Q: CH,CH,OH :0: :0: || Но — С — CH,— СH, — С — ОН :0: - - CH,-S– CH, H,0 KCl Cl4
A:
Q: Name H H-C- H Name H H Н Н Н .С. С. -C-H H H H H H H H CH₂ H H H -H Name X -С H2C H -CH3 Name нннн н…
A: Since you asked multiple questions so as per Q&A guidelines of the portal I solved the first…
Q: What are the products of the reaction?
A: Step 1: The given reaction is known as dihydroxylation of alkene. Here, the osmium tetraoxide (OsO4)…
Q: Which of the following are a. hemiacetals? b. acetals? c. hydrates?
A: a. Hemiacetals are formed by reaction of 1 equivalent alcohol added to aldehyde or a ketone.
Q: CH3 f. H,C-CH2-NH-C-CH2-CH-CH3 Туре of functional group CH3 H,C-CH2-NH-ĉ-CH,-CH-CH3 IUPAC CH3…
A: Introduction: IUPAC nomenclature: IUPAC stands for International Union of Pure and Applied…
Q: Name the following:
A: Since the question has multiple sub-parts, we are entitled to answer only the first 3 parts.
Q: I нннн н | | | | | н-c-c=С-С-С-Н I | | н н Name Т. Name H-C-H c = c — с н / нннннн | | | | | |…
A:
Q: HC. N. NH aHC 'N' N. H-C-H H- OH H- -OH H- OH NH2 H- H. N 9- -0-CH2 но OH
A: Oxidoreductases - these are a class of enzymes that catalyze oxidoreduction or redox reactions .…
Q: CH3 H3C H30", A H;CH;CO OCH,CH3
A:
Q: Vhat is the molecular formula ÇH,OH Ç=O НО-ҫ н H-C-OH H-C-OH ČH,OH
A: Carbohydrates are sugars and one of the major nutrients found in foods and drinks.
Q: Name the following compounds: 1. 2. Br Hc-CH-CH-ö-OH 3. -CH-CH 4. H,C-CH-CH-OH CI 5. HC-C=C-CH,-CH,…
A: According to our policy we should solve one question (If any subparts present then we should solve…
Q: ОН HOH2C ОН ОН II CO2H CO2H Но- -H- H- -HO- HO HO- ČH2OH CH,OH II IV ÇO,H CO2H Но- H- H- -OH H- -OH…
A:
Q: Examine the following compounds, and answer questions 31-35. CH3-CH2-CH-CH, CH-CH-CH,-OH CH-CH2-OH…
A: The isomers which have same chemical formula but different structures are called as structural…
Q: OH PBR3 1. NaOCH3, CHзОН 2. Hао CI NaOCH,CH3, CH;CH2OH
A:
Q: Of the possible products shown for the following reaction, are there any that will not be formed
A: The first product in the above pic will not form in the reaction as we have change in the…
Q: CH3-o CH2-CH2-CH2.-Ct3 CH8 -CH-CH20-CH2-C H3 CH3-C Hz-0-CHz-CH 2 -Cto CH3-0-CH - CH 2-CH3 (F 0-CH3…
A:
Q: d. C,H-C-CH, H* e. C,Hs-C-CH f. C,Hs-C-CH; NaOH g. CH;-C=C-CH, HgSO., 4 h. C.Hs-C-C.H,
A:
Q: _____ H3PO4 + _____ KOH ----> _____ K3PO4 + _____ H2O
A: Complete the reaction--- _____ H3PO4 + _____ KOH ----> _____ K3PO4 +…
Q: C10H16B12 C C10H160 1. В1з, THF C10H180 F 2. Н2О2, НО E Br2 CH3CO0OH Br2 а-pinene C10H16 1. OsO4…
A: “Since you have posted a question with multiple sub-parts, we will solve first three subparts…
Q: 19 H3C- -CH, CI 20 Br Br HC Ec- -CH-CH-CH,-CH, 21 HC-C CH
A: The organic compounds are named according to the IUPAC nomenclature. It is done according to some…
Q: oper name for these compounds. Br OH OH Br a) b) CI c) CI CH3 Tevol (p H3C. CH3 H3C Br он
A: Given, Give the IUPAC names of the following compounds = ?
Q: CH3 Но CH3 CH3 CH3 -H- Но- - HO- H- OH -OH -H- CH3 H- OH H- -ОН Но- -H- CH3 CH3 CH3 I II III IV
A: Meso compounds contain chiral centers/stereogenic centre though they are optically inactive. Meso…
Q: (e) CH,CICHB1(CH2),C(CH,CH3);
A: IUPAC name : 2-Bromo-1-chloro-7,7-diethylnonane
Q: 1. 2eq CH3M9B 1.LIAIH4 F 2. H+ 2. НаО PBR3 CH30¯ Na + 1. NaBH4 B 2. H2O CH3-CH2-Č-OH C CH3CO,Na |1.…
A:
Q: : ОН :0:0 (а) CH3-CH2-CH2 CH3-CH2-CH2 I or II :0:0 :0:0 (b) CH3 CH2-CH2 CH3 CH=CH or II CH,O : NHO…
A: The bases given are,
Q: 0-CH,-CH3 H. 0-CH3 CH3-C-0-CH3 CH3-CH2-0-CH,-OH 0-CH3 OH 0-CH2 CH3 HO-CH, CH,…
A: Alcohols add reversibly to aldehydes and ketones to form hemiacetals. This reaction can continue by…
Q: H. OH CH3 1. H3C H3C CH3 and H. OH H3C. CH3 2. Hзс CH3 HO and H. H3C. CH3 CH3 HO HO. H. and 3.
A: As there occurs change in the stereocenter of the optically active structures, despite of the change…
Q: -CH2OH b. -CH,Br -CH(CH3)2 -CH3 -CH2CH,OH -CH¿OH -CH,CH,CH,Br а. -H -CH3 -CI -OH c. -CH,CH,Br
A: Priority is on the basis of atomic number of atoms as here in CH2OH ist atom is carbon next is 2…
Q: (c) CH₂-CH₂-CH₂-CH₂-CH₂-Br and CH₂-CH₂-CH-CH₂-CH₂ CH₂-CH-CH₂- T CH3 C CH₂-CH₂ CH₂ CH₁ i O−CH, and…
A: Please find the attachment.
Q: CH3-C=C-CH3 Li CHs-C=C-CH, H1 2mole H.O Hg Soy CHs -C ECH. h0
A: The question is based on the concept of organic reactions. it involves reactions of alkynes with…
Step by step
Solved in 2 steps with 1 images

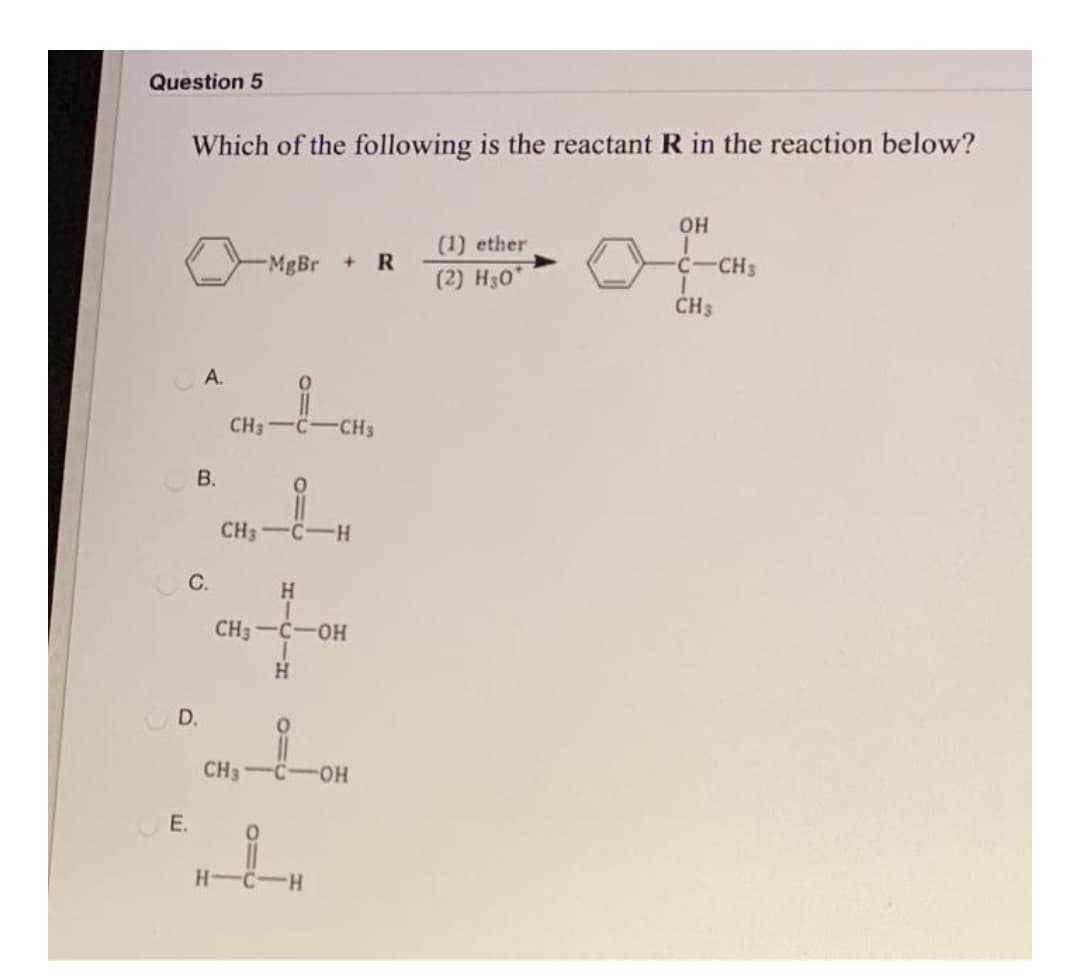
- full structural form is needed for complete reaction h.wis compound C 1-butanol?I also need help with compound A and BQUESTION 8 What reagent and condition are required to convert dicyclobutyl ketone to di-cyclobutyl methanol? A. KMnO4, acetone, H3O+ B. Propanoic acid, HCl, 30oC ? C. NaBH4, ethanol, H3O+ D. CH3CH2MgBr, ether, H3O+
- 1. Using Br2 in C2H4Br2 will result in HBr and ______. a. C2H3Cl3 b. C2H4Cl3 c. C2H2Cl3 d. none of the above 2. How many halogenation are posible in propane? a. 3 b. 8 c. 6 d. 10 3.Sulfonation of pentane will result in ________ and water. a. C5H11SO3H b. C5H12SO3H c. C5H14SO3H d. none of the above 4.Nitration of hexane will result in ________ and water. a. C6H13SO3H b. C6H15NO2 c. C6H13NO2 d. C6H14NO2 5.How many moles of O2 in heating a C12H26 (dodecane) a. 27 b. 37 c. 24 d. none of the aboveOxidation of Butanol -1 in KMnO4/H+ will produce-------------. a.Butanoic Acid b.Butanal c.Pentanoic acid d.Butene -2Assuming a 56.8% yield, how kany ml of alkene is required to produce 22.5 mL of 2,3-dimethyl-2-butanol? MW of alkene: 84.16, d: 0.653 MW of 2,3-dimethy-2-butanol: 102.17, d:0.823
- Which reagents are used for reaction 1? a NaN3, ethanol and NaBH4, ethanol, H3O+ b CH3CH2NH2, NaOH c NH3, NaOH d NH3, DCC Which reagent are used for reaction 2? a CH3COOH, DCC b CH3COCH3 and NaBH4, ethanol, H3O+ c CH3COH and NaBH4, ethanol, H3O+ d CH3CONH2 and CH3CH2NH2What is the final product formed in the reaction below?KMnO4, warm, conc'd reacts with hept-1-ene to yield __________. CO2, hex-1-ene CO2, hexanoic acid Formic acid, pentanoic acid Ethanoic acid, pentanal Formic acid, hexanone
- Illustrate the reaction of (CH3)3COH with HBr ?1. Which reaction conditions would be best for turning ethene into ethanol? a.H2O, HBr b. CH3OH, H2SO4 c.CH3OH, HBR d. H2O, H2SO4 2. Which alkene would be non-regioselective in a reaction with HBr? a. 2-ehyl-2-methyl-2-butene b.2-ethyl-3-methyl-2-butene c. 2,3-dimethyl-2-butene d.1,2-dimethyl-2-butene4. draw the structure of the organic product for each alcohol reacting with permanganate. Look on the internet for an explanation of why the tertiary alcohol in presence of KMnO4 might react. (hint may involve dehydration.) - methanol - ethanol - propanol - 2-methyl-2-propanol

