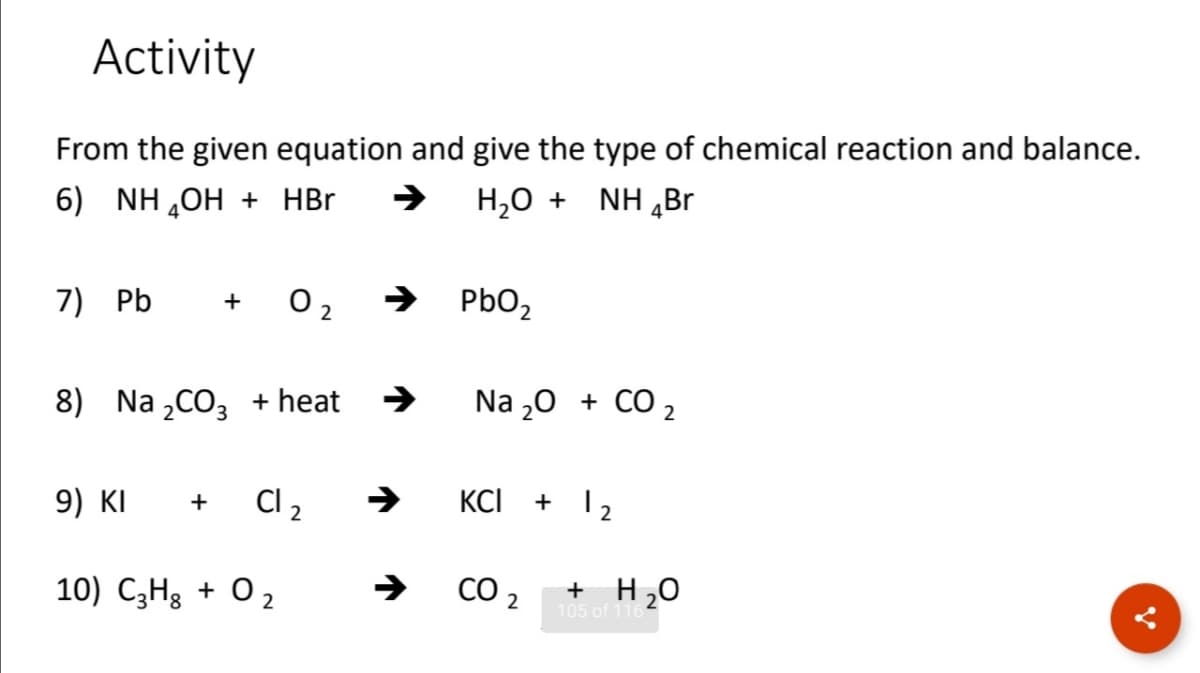
Activity From the given equation and give the type of chemical reaction and balance. 6) NH,OH + HBr H,0 + NH „Br 7) Pb O 2 PbO, + 8) Na „CO, + heat Na „0 + CO 2 9) KI C 2 KCI + 12 + 10) C3H3 + O 2 CO 2 + H20 105 of 116
Q: What is the indicator employed in Cerimetry?
A: Cerimetry : Principle: Cerric ammonium sulphate in acidic medium can function as a strong oxidising…
Q: What is the smallest whole-number coefficient for H₂O when the equation MnO₄⁻ + I⁻ + H₂O → MnO₂ +…
A: Introduction: The chemical equation shows the reaction between the two or chemical species and their…
Q: Consider thé following statement: Statement I: Matrix is equal to blank plus the analyte. Statement…
A: Matrix: Matrix refers to the components of a sample other than the analyte of interest. Analyte: a…
Q: Write all mass balance (MB) and charge balances (CB) equations for the following: (1) 0.2 M…
A: (1) 0.2 M K3PO4 + 0.3 M H3PO4 Solution -
Q: Name the products formed in a permanganate titration (FAS Vs KMnO4) and Give reason Why Burning of…
A: Redox reaction in which oxidation and reduction both happens simultaneously in the same reaction..…
Q: 1. (a) Balance the following reaction in acidic conditions: (do the work below and put a box around…
A: a.) To balance reaction in acidic medium , we would balance it by half reaction method. For this…
Q: one of the following is primary standard for iodine O a. potassium hydrogen phthalate b. sodium…
A: Standardization is the process of determining the exact concentration of the solution. Primary…
Q: You have measured 1.231 g of unknown acid on the balance. Final burette reading is 23.63 mL Initial…
A: Molarity is defined as number of moles of solute present in 1 Litre of the solution
Q: The iron (II) in an acidified solution is titrated with 0.0317 M solution of KMnO4: 5Fe2+ +MnO4-…
A: The balanced ionic Chemical equation given in the question is - 5Fe2++MnO4- +8H+ →5 Fe3+ +Mn2+…
Q: When a sample of the CH,CI, layer with a volume of 6.75 mL is collected, it requires 2- 19.53 mL of…
A: The reaction between I2 and S2O32- is as follows, I2+2S2O32-→2I-+S4O62- Therefore 2 mol S2O32- is…
Q: What is the Molarity of H2SO4 if a (4.2100x10^1) mL sample requires (2.760x10^1) mL of (1.32x10^-1)…
A:
Q: Ca2+ in a drinking water sample was analyzed by the following reactions below: Ca2+(aq) +…
A: Hey, since there are multiple questions posted, we will answer first question. If you want any…
Q: 1- Corrosion occurs at the anode by the oxidation of metals. Il-In wet corrosion, corrosion products…
A: A multiple choice question about corrosion, which is to be accomplished.
Q: When 38.0 mL of 0.1250 M H,SO, is added to 100. mL of a solution of PbI,, a precipitate of PbSO,…
A: We know that We have, Molar mass of PbSO4 = 303 g/mol Mass of PbSO4 = 0.0471g So Moles of PbSO4 =…
Q: Student A wanted to determine the IN of an unknown oil (density - 0.85 g/ml) using pyridinium…
A: Commonly, oil substances are triglycerides with unsaturated units (double bonds). The total number…
Q: Which one occurs? State your observations. • A metallic deposit appears • Bubbles appear • A…
A: Approximately 2 mL of Solution of NaOH is added to a sample of Solution Ni (II) Nitrate with a…
Q: on 29 of 31 > UZ JI Complete and balance the equation for this single displacement reaction. Phases…
A:
Q: Titration of the I2 produced from 0.1187 g of primary- standard KIO3 (Molar Mass= 214.0o g/mol)…
A: The reactions given are, Given: Mass of KIO3 reacting = 0.1187 g. And volume of Na2S2O3 required =…
Q: What is the precipitate formed from the reaction of permanent hard water with sodium carbonate?…
A:
Q: Vhat volume of 2.50 M HCl in liters is needed to react completely (with nothing left over) with…
A: For this problem equation used is M1 V1 = M2 V2 M1 = 2.50 V1=? M2 = 0.500 V2…
Q: The iron (II) in an acidified solution is titrated with 0.0317M solution of KMnO4:…
A: The question deals with the molar-ratios. It is the ratio of moles of reactant consumed and moles of…
Q: 2. Hydrazine (N,H) reacts with the bromate ion to produce nitrogen gas and the bromide ion. A 0.132…
A:
Q: 1.0 g of aluminum hydroxide reacts with 25.0 mL of 0.500 M carbonic acid according to the following…
A: Limiting reagant are those reactant which have less value of ratio of mol to stoichiometry…
Q: Calculate the food coloring concentration in a standard if 3.00 mL of 0.892 %(v/v) stock solution…
A: Given : Concentration of initial solution = 0.892 % (v/v) Volume of initial solution taken = 3.00 mL…
Q: How many liters of a 0.209 M KI solution needed to completely react with 2.43 g of Cu(NO,),…
A:
Q: After performing precipitation gra weighed the crucible with the wei of 23.2125 g, then 23.1136 g,…
A:
Q: Select the correct and balanced equation described by: An aqueous solution of lead(Il) sulfate is…
A: Lead sulfate reacts with sodium chloride to produce sodium sulfate and lead chloride.
Q: Calcium in a 2.00-g sample is determined by precipitating CaC2O4, dissolving this in acid, and…
A: Titration is general laboratory method of quantitative analysis of determining the concentration…
Q: The proton balance equation for a solution of NagPO4 is O (H3O'1+ [H2PO1+ 3[H3PO4] - (OH'] + 3[PO,1…
A:
Q: Titration of the I2 produced from o.1187 g of primary- standard KIO3 (Molar Mass= 214.0o g/mol)…
A: Please find your solution below : Molarity is a measure of concentration which is defined as the…
Q: When a 8.000 g of mineral ore sample containing Sn(CO3)2 (238.7 g/mol) and other inert material was…
A: Given that, 8.000 g of a sample containing Sn(CO3)2 and other inert material was is treated with…
Q: What is the product of the following reaction equation after balancing? If no reaction state that "…
A: Metals react with acid to produce metal salt and hydrogen gas.
Q: Student A weighed a 6.421-g sample known to contain ClO4- and Cl-. The sample wasdissolved in water…
A: Inorder calculate the amount of chlorine and hyperchlorate ion present, the concentration of…
Q: Most commercial samples of K MnO contain some manganese dioxide that must be femoved prie Submit…
A: Oxidation is said to be loss of electrons and reduction is gain of electrons. Oxidizing agent…
Q: Balance eaxch equation H2O2+ClO2 --> ClO2–+O2 (in basic solution)
A: 1. H2O2 + ClO2 --> ClO2– + O2 (in basic solution) To balance given reaction in basic medium;…
Q: 9) How many grams of AlCl3 (s) Should be dissolved, to prepare 1 liter of a solution in which the…
A:
Q: After reducing a 0.4080 g iron to Fe²⁺, it was titrated with 42.72 mL of 0.1211 N KMnO₄ solution.…
A: The chemical reaction taking place is: MnO4-+5Fe2++8H+→ Mn2++5Fe3++4H2O Mass of Fe3O4 in the sample…
Q: The iron (II) in an acidified solution is titrated with 0.0317M solution of KMnO4:…
A:
Q: As a technician in a large pharmaceutical research firm, you need to produce 250. mLmL of 1.00 MM…
A:
Q: calculate weight of AgCl when added excess of AgNOs to 0.75g of BaCl2 if M.wt AgCl=143.3 and M.wt…
A: Given that: mass of BaCl2 = 0.75 g M.wt of AgCl = 143.3 g/mol M.wt of BaCl2 = 208.3 g/mol
Q: As a food chemist for a major potato chip company, you are responsible for determining the salt…
A: Mohr method is the method to determine the chloride ion concentration of a solution. This is done by…
Q: Balance the equation in acidic conditions. Phases are optional. equation: Cr2+H2MoO4⟶Cr3+Mo
A:
Q: Ethyl Mercaptan (C2H5SH) is a colorless or yellowish liquid or a gas with a pungent, garlic or…
A: Given :- Mass of Ethyl Mercaptan, C2H5SH sample= 3.85 g Molarity of I2 = 0.01204 M = 0.01204 mol/L…
Q: 6. A 6.881-g sample containing magnesium chloride and sodium chloride was dissolved in sufficient…
A:
Q: A solid sample containing some Fe(2+) ion weighs 1.923 grams. It requires 36.44 mL 0.0244 M KMnO4 to…
A: The complete balanced redox reaction is given by
Q: b) Ammonium iron sulphate, (NH4)2 Fe (S04)2. 6H20], RMM = 392 may be used to standardize a solution…
A: Given data,Molarity of iron solution=0.1MVolume of iron solution=250cm3=0.250L
Step by step
Solved in 4 steps

- Potassium dichromate has several industrial applications. To determine the purity of the salt that will be used in different industrial processes, a sample mass equal to 2.660 g was dissolved and quantitatively transferred to a 500.00 mL flask. An aliquot of 25.00 mL of this solution was treated with excess KI and the released iodine was titrated with 0.1000 mol L-1 sodium thiosulfate, spending 27.00 mL. Calculate the purity of the analyzed salt. Data:K = 39.10 O = 16.00 Cr = 52.00 I = 126.9 S = 32.07Airbag experiment The simulation link is provided below if needed. It's probably best to use the simulation because the pictures attached won't show everything in full. Simulation link: https://interactives.ck12.org/simulations/chemistry/decomposition-reaction/app/index.html?screen=sandbox&lang=en&referrer=ck12Launcher&backUrl=https://interactives.ck12.org/simulations/chemistry.html Just select ammonium nitrate and then select 1 mole and press play. Record macroscopic observations of the bag and the crash test dummy as well as microscopic observations of the particles within the steering mechanism and the inflated/inflating airbag.If all other variables were kept constant, determine theeffect that the following errors would have on the calculatedpercent yield of the product. Would the yield be expected toincrease, decrease, or would there be no effect? Explainyour reasoning.– The product was insufficiently dried before weighing.– Some of the product was lost during the transfer fromthe Buchner funnel to the evaporating dish.– 7.5 mL of FeCl3 was added instead of 3.0 mL asoutlined in the procedure.– 4.587g of K2C2O4H2O was used instead of exactly4.000g .– The recrystallization step was skipped and theexperiment went straight to vacuum filtration.
- A first-stage recovery of magnesium from seawater is precipitationof Mg1OH22 with CaO:Mg2+(aq) + CaO(s) + H2O(l)---->Mg(OH)2(s) + Ca2+(aq) What mass of CaO, in grams, is needed to precipitate 1000lb of Mg(OH)2?Airbag experiment The simulation link is provided below if needed. It's probably best to use the simulation because the pictures attached won't show everything in full. Simulation link: https://interactives.ck12.org/simulations/chemistry/decomposition-reaction/app/index.html?screen=sandbox&lang=en&referrer=ck12Launcher&backUrl=https://interactives.ck12.org/simulations/chemistry.html Just select ammonium nitrate and then select 2 moles and press play. Record macroscopic observations of the bag and the crash test dummy as well as microscopic observations of the particles within the steering mechanism and the inflated/inflating airbag.Gravimetric analysis of Fe3O4 (MW = 232 g/mole) may be undertaken with the following reactions: Fe3O4 → Fe2O3 → Fe (OH)3. Weight of sample containing 8.00% Fe3O4 that must be taken to obtain a precipitate of Fe(OH)3 (MW = 107 g/mole) that weighs 150 mg is . a. 0.108 g b. 0.325 g c. 1.355 g d. 4.065 g Amount of Fe2O3 (MW = 160 g/mole) from which 150 mg of Fe(OH)3 (MW = 107 g/mole) may be obtained is . a. 0.112 g b. 0.224 g c. 0.448 g d. none of the other choices
- A fermenter was filled with 10L of 0.6 mol/L sodium sulfite solution containing 0.003M Cu2+ ion and the air sparger was turned on. After exactly 10 minutes, the airflow was stopped and a 5 mL sample was taken and titrated. The concentration of sodium sulfite in the sample was found to be 0.2 M. Calculate the oxygen uptakeA weight of 0.50 g was taken impure container containing sodium carbonate and bicarbonate. Dissolved in water and then crushed with hydrochloric acid (0.1 N), the burette reading game was at the endpoint of phenolphthalein of 10.5 ml and at the end point of the orange methylation point 30.1 ml. The percentage of sodium carbonate was in ................. knowing that the weights are: Na: 23, C: 12, O: 16To calculate the unknown concentration of a chemical in a solid sample, 4 different calibrationcurves were plot using 4 different methods. Which of the following method is considered asacceptable? a. Method 4, R? = 0.998b. Method 1, R2 = 0.650c. Method 2. R' = 0.890d. Method 3, R' = 0.169
- Which of the following are equivalent to 2,500 ppm Cu2+? (There may be more than one answer) MW: Cu (63.55) a) 2.5 ppb Cu2+ b) 2,500,000 ppb Cu c) 2.5 ppt Cu2+ d) 39.34 mM e) 0.03934 M f) 0.07868 N (in precipitation reaction) g) 0.07868 N (in redox into Cu+)The percentage protein content of chicken breasts are determined 5 times and the following results obtained: 28.5, 30.1, 30.2, 30.0 en 30.2%. The first value (28.5%) appears anomalous. Should it be retained or rejected at a 90% confidence level? Show all equations.1) Commercial fuming Sulphuric acid (Oleum-H2S2O6) is 99.9% solution. Please convert it into molarity.2) Find out the Volume (dm3) of product (gas) at RTP when 0.58 M, 150 mL NaOH (aq.) reacts with 350 mL, 0.25 NH4Cl.

