Ammonia has been studied as an alternative "clean" fuel for internal combustion engines, since its reaction with oxygen produces only nitrogen and water vapor. The following reaction takes place (at a certain temperature) as follows: 4 NH3(g) + 302(g) = 2 N2(g) + 6 H,0(g) 2.1 In the reaction above, identify which reactant is oxidized and which is reduced NH3 O2 2.2 An industrial chemist studying this reaction fills a 500. mL flask with 1.3 atm of ammonia gas and 1.2 atm of oxygen gas, and when the mixture has come to equilibrium measures the partial pressure of nitrogen gas to be 0.33 atm. Calculate the pressure equilibrium constant for this reaction. You must fill out the ICEE table below for full credit. Round your answer to 2 significant digits. Make sure to box your final answer for K,. If you need more space to show your work, please use the backside of this page 4 NH3(g) + 302(g) 2 N2(g) + 6 H,0(g) Initial pressure Change Expression Equilibrium pressure K, =
Ammonia has been studied as an alternative "clean" fuel for internal combustion engines, since its reaction with oxygen produces only nitrogen and water vapor. The following reaction takes place (at a certain temperature) as follows: 4 NH3(g) + 302(g) = 2 N2(g) + 6 H,0(g) 2.1 In the reaction above, identify which reactant is oxidized and which is reduced NH3 O2 2.2 An industrial chemist studying this reaction fills a 500. mL flask with 1.3 atm of ammonia gas and 1.2 atm of oxygen gas, and when the mixture has come to equilibrium measures the partial pressure of nitrogen gas to be 0.33 atm. Calculate the pressure equilibrium constant for this reaction. You must fill out the ICEE table below for full credit. Round your answer to 2 significant digits. Make sure to box your final answer for K,. If you need more space to show your work, please use the backside of this page 4 NH3(g) + 302(g) 2 N2(g) + 6 H,0(g) Initial pressure Change Expression Equilibrium pressure K, =
Chemistry
10th Edition
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Chapter6: Thermochemistry
Section: Chapter Questions
Problem 114AE: Consider the two space shuttle fuel reactions in Exercises 81 and 82. Which reaction produces more...
Related questions
Question
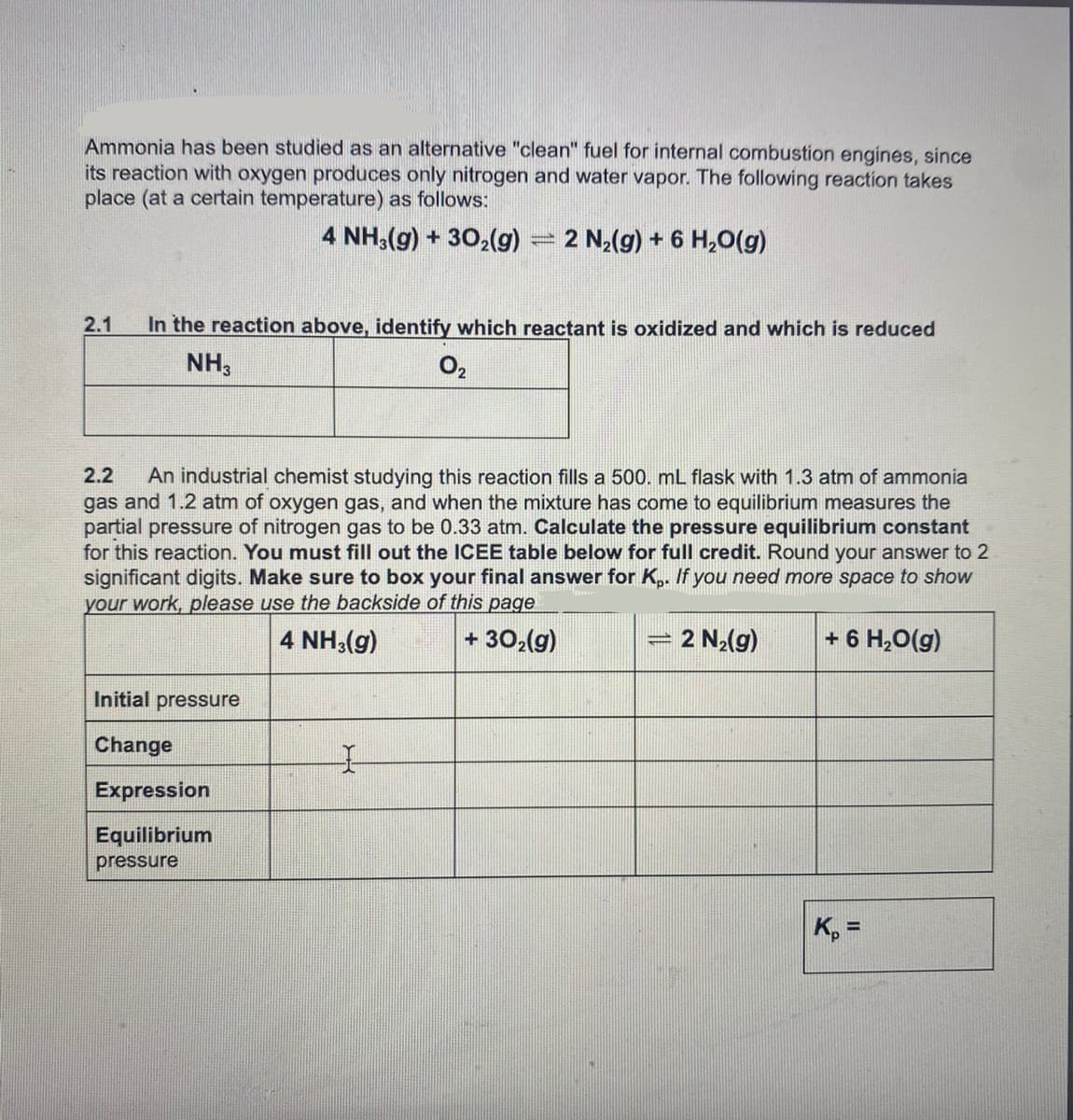
Transcribed Image Text:Ammonia has been studied as an alternative "clean" fuel for internal combustion engines, since
its reaction with oxygen produces only nitrogen and water vapor. The following reaction takes
place (at a certain temperature) as follows:
4 NH,(g) + 302(g)
- 2 N2(g) + 6 H,O(g)
2.1
In the reaction above, identify which reactant is oxidized and which is reduced
NH,
O2
2.2
An industrial chemist studying this reaction fills a 500. mL flask with 1.3 atm of ammonia
gas and 1.2 atm of oxygen gas, and when the mixture has come to equilibrium measures the
partial pressure of nitrogen gas to be 0.33 atm. Calculate the pressure equilibrium constant
for this reaction. You must fill out the ICEE table below for full credit. Round your answer to 2
significant digits. Make sure to box your final answer for K,. If you need more space to show
your work, please use the backside of this page
4 NH3(g)
+ 30,(g)
2 N2(g)
+ 6 H,0(g)
Initial pressure
Change
Expression
Equilibrium
pressure
K, =
%3D
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 5 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning