An idenl diatomic gas has a molar internal energy equal to E =RT which depends only on its absolute temperature T. A mole of this gas is taken quasi- statically first from state A to state B, and then from state B to state C along the straight line paths shown in the diagram of pressure p versus volume V. (a) What is the molar heat capacity at constant volume of this gas? (b) What is the work done by the gas in the process A → B→ C? (c) What is the heat absorbed by the gas in this process? (d) What is its change of entropy in this process? B 8 2 1 2 3 (10' cm
An idenl diatomic gas has a molar internal energy equal to E =RT which depends only on its absolute temperature T. A mole of this gas is taken quasi- statically first from state A to state B, and then from state B to state C along the straight line paths shown in the diagram of pressure p versus volume V. (a) What is the molar heat capacity at constant volume of this gas? (b) What is the work done by the gas in the process A → B→ C? (c) What is the heat absorbed by the gas in this process? (d) What is its change of entropy in this process? B 8 2 1 2 3 (10' cm
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter12: Thermodynamic Processes And Thermochemistry
Section: Chapter Questions
Problem 58P
Related questions
Question
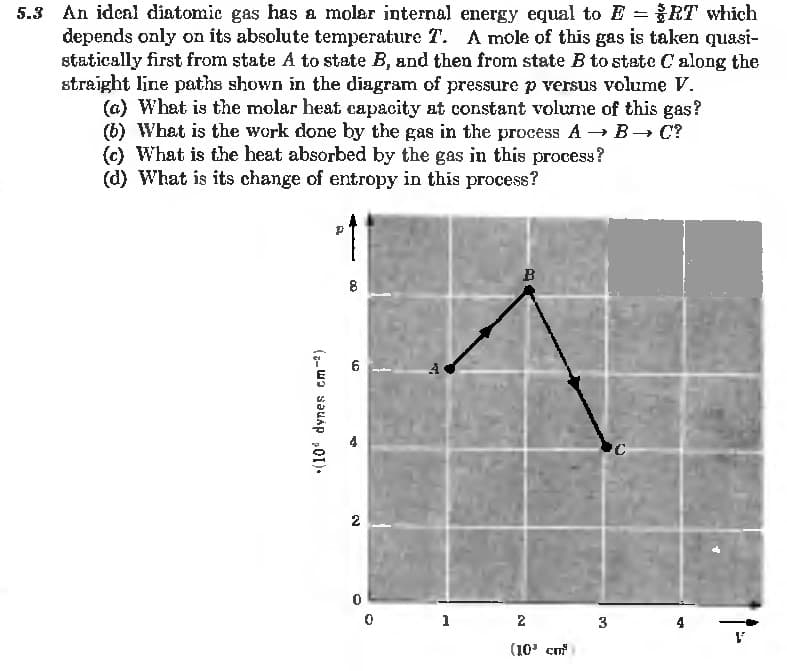
Transcribed Image Text:5.3 An ideal diatomic gas has a molar internal energy equal to E RT which
depends only on its absolute temperature T. A mole of this gas is taken quasi-
statically first from state A to state B, and then from state B to state C along the
straight line paths shown in the diagram of pressure p versus volume V.
(a) What is the molar heat capacity at constant volume of this gas?
(b) What is the work done by the gas in the process A → B→ C?
(c) What is the heat absorbed by the gas in this process?
(d) What is its change of entropy in this process?
B
6
2
1
2
3
4
(10' cm
(7- W) sauap 01).
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning