Q: Compare the shape and volume of a solid and a gas.
A: The objective of this question is to compare the shape and volume of a solid and a gas, two of the…
Q: Determine the density of CO2 at 745 mmHg and 65 degrees C?
A: The objective of this question is to calculate the density of carbon dioxide (CO2) under specific…
Q: Correct each molecule in the drawing area below so that it has the skeletal ("line") structure it…
A: let us first tackle the amines issue while dissolving in strong bases like sodium hydroxide. Since…
Q: in the reaction 2SO(g) -- 2SO2(g), Kc= 4.0 x 10 -2 at 300 C. if the starting amount of SO2 was .5 M.…
A: The objective of the question is to find the equilibrium concentrations of all species in the given…
Q: Name the following molecule using the IUPAC system HO OH
A: The objective of the question is to name the given molecule using the International Union of Pure…
Q: CH, PPh Ph,P=CH(CH2CH₂) (CH3)₂CuLi
A: Step 1: Step 2: Step 3: Step 4:
Q: Calculate the pH of solution of the following weak acid. Use Table 14.2 and Appendix H in your…
A: Step 1:Amphiprotic ion is a specie that can accept as well as donate the H+ ion. It means it can act…
Q: For each of the substituted benzene molecules below, determine the inductive and resonance effects…
A: Step 1:Step 2:
Q: U U U CI Recall that the Friedel-Crafts alkylation reaction forms a new C-C bond between an aromatic…
A: The objective of the question is to identify which of the given groups (OH, Cl, +a, NO2, Cl as the…
Q: What type of reaction could be most easily used to accomplish the following? Br ○ SN1 Gilman O…
A: Step 1: Step 2: Step 3: Step 4:
Q: Which of the following would be a major product of the following reaction? Br Br Br hv ? Br2
A:
Q: Draw the product of the reaction shown below. Use a dash or wedge bond to indicate the…
A:
Q: A prepared buffer solution contains a mixture of H2PO4 and HPO ions. Which species will react with…
A: The objective of the question is to identify which species in a buffer solution will react with any…
Q: Describe how you could achieve a higher degree of separation for the bands of food dyes on a TLC…
A: The objective of the question is to understand how to achieve a higher degree of separation for the…
Q: Learning Learning ☑Online teaw... ot AA Learn Learning prod03-cnow-owl.cengagenow.com Learning…
A:
Q: None
A: Key points about Benzene:Hydrocarbon: It belongs to a class of molecules called hydrocarbons,…
Q: Select the nonprotein nitrogen compounds from the listed analytes. Question 57 options:…
A: The objective of the question is to identify the nonprotein nitrogen compounds from the given list…
Q: Draw the Orbital Overlap Bonding of Permaganate (MnO4-)
A: Resonance structures of the permanganate ion ( ) may be used to visualize the electron distribution…
Q: Give the currved arrow pushing mechanism for the following reaction, indicate Lewis acid/base and…
A: Step 1:• For the mechanism: • In the first step , protonation of alcohol is take place.• In the…
Q: Question 7 Please predict the products for each of the following reactions: ABCD 1. Na 2. PrBr Na 10…
A: Step 1:Step 2:Hence option B is correctStep 3: Step 4:
Q: 1. (5 pts.) Predict the product for the following reactions. O₂N- HCI 2 eqv. 1 eqv. Br2 CH2Cl2
A: Step 1: Step 2: Step 3: Step 4:
Q: Question 28 Predict the FINAL (?) product for each of the following reaction or synthetic chain: Mgl…
A: The sequence of reactions shown suggests the formation of a Grignard reagent followed by a reaction…
Q: None
A: Step 1: Balance the molecular equation with the correct number of coefficients per chemical species.…
Q: Draw Walsh diagram for a [IrCl 4]3-in Td symmetry undergoing Jahn - Teller distortion to [IrCl 4]3-…
A: The objective of the question is to understand the Jahn-Teller distortion of the [IrCl4]3- complex…
Q: Draw the starting epoxide that would form the product shown below. Use a dash or wedge bond to…
A: To help guide you through drawing the starting epoxide that would form a given product, we first…
Q: 5. How many grams of CuSO4 * 5 H₂O are needed to prepare 100 ml of a 0.10M solution? (This is like…
A: The target volume to prepare is 100 mL and the concentration is 0.10M CuSO4. So we have…
Q: question 3 help
A:
Q: An alcohol has the formula C4H9OH. How many isomers can exist for this alcohol?
A: The objective of the question is to determine the number of isomers that can exist for an alcohol…
Q: 2. Draw crystal-field splitting diagrams for PtCl 4 and [PtC14]² and calculate the CFSE. Be sure to…
A:
Q: 3. Identify the given drugs. Write its functional groups and heterocyclic rings present. Explain…
A: The IUPAC name of the given drug is sodium…
Q: question1 help
A: When all properties of a molecule can not be explained by a single structure, more than one possible…
Q: Determine the equilibrium constant for a reaction at 200.0 K if ∆G° =-11.70 kJ/mol. (R = 8.314 J/mol…
A:
Q: Please select the appropriate reagent to complete the following transformation. Ph3P-CH2 MgBrCH3…
A: Ketones react with phosphorus ylides in a reaction called the Wittig reaction. The Wittig reaction…
Q: It says that my answers are wrong and I can’t figure out what went wrong please help!
A:
Q: Please correct answer and don't use hend raiting
A: Step 1: Step 2: Step 3: Step 4:
Q: None
A: Part 2: Explanation:Step 1: Determine the moles of NaOH used in each trial:The moles of NaOH can be…
Q: A mixture of nitric and sulfuric acids generates the nitronium ion, + NO2 (mechanism provided, which…
A:
Q: Scanning electrochemical microscopy was used to study the lifetime of guanosine radical, which was…
A: The objective of the given question is to determine the percentage of tip-generated guanosine…
Q: 4. Which acid and alcohol must be reacted to generate the indicated esters? (a) (b) (c) ов ↑ (d) OH
A: Esters are formed when acid chlorides are reacted with alcohol nucleophiles. This is the more…
Q: dont provide handwriing solution ...
A: this helps
Q: dont want hand writting image i will dislike you in 10 accounts
A: Step 1: This is Corey House reaction. - Bromobenzene reacts with lithium in pentane to form aryl…
Q: 7. Balancing Equation Practice ections: Balance the following chemical equations. Write final…
A: Please see the attached image for the solution. If there are queries, please do not hesitate to ask.…
Q: Identify and provide an explanation of the differencesbetween homogeneous and heterogeneous sampling…
A: Approach to solving the question: Detailed explanation:The concepts of homogeneous and…
Q: The first enzyme to rise in acute pancreatitis is ________________, and the enzyme which stays…
A: The objective of the question is to identify the enzymes that are affected during acute…
Q: Iron nanoparticles were produced by the evaporation method and their average sizes are estimated 25,…
A: The peak width (β) can be calculated using the Scherrer equation which can be represented as…
Q: Identify whether each of the following molecules can exist as optical isomers: NH₂ Br-C-COOH H…
A: The objective of the question is to determine whether the given molecules can exist as optical…
Q: The chemical environment of carbons can be deduced form their chemical shifts. Deduce the relative…
A: For molecule 1:for molecule 2: Step 3: Step 4:
Q: What is the pH of the solution that results when the following solutions are combined? 0.020 L of…
A: Thank you,Please rate my response.
Q: Draw a resonance structure of the diene that will help determine the regiochemistryof the…
A:
Q: Current Attempt in Progress Propose an efficient synthesis for the following transformation: (c) →…
A: Step 1:Step 2: Step 3: Step 4:
i dont want hand writing image
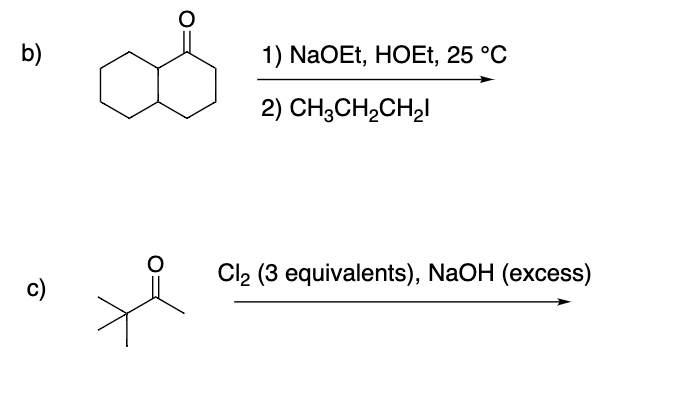
Step by step
Solved in 2 steps with 2 images

- Please provide a step-by-step explanation. Thanksorganic synthesis of Terephthalic acid from benzene, provide schemes/ figures of the synthesis structure4 Show how the compound thiodiazine~C21H26N2S2~decomposes anaerobically and how these end products in turn decompose aerobically to stabilized sulfur and nitrogen compounds.
- drug synthesis from corey-kim oxidationOrgonic Chemistry II: The answer is writtten as followed. But I Need Explanation. My question is that: Can I siwtch reagent 2 to reagent 1? for example reagent 1 is Cl2,Fecl3, can I changed it to reagent 2??? Why and why not???Give hand written explanation asap Thank you

