Q: Balance the following red ox reaction if it occurs in acidic solution. MnO4^-(aq) + H2C2O4(aq) →…
A: Steps in balancing redox reactions: 1) Divide the overall reaction into an oxidation half-reaction…
Q: Balance the following net ionic equation and write the sum of the coefficients of the reactants and…
A: Skeleton unbalanced reaction: For balancing this redox reaction, we 1st need to determine the…
Q: Using oxidation and reduction half-reactions, balance the skeletal equation Cl2O7(g) + H2O2(aq) →…
A: (1) Divide the reaction into half cell oxidation recation and half cellreduction reaction→Cl2O7(g)…
Q: In the following equation, which species is the reducing agent? SiO2(s) + 2C(s) → Si(s) + 2CO(g)…
A: We have to choose which species in the given reaction is the reducing agent SiO2(s) + 2C(s) → Si(s)…
Q: Complete and balance the following redox reaction in acidic solution H2O2(aq) + CrO7^2- (aq) ->…
A: Acidic medium is the medium in which solution is in acidic medium having pH value less than 7. To…
Q: Balance the following redox raction under acidic conditions. H2S(g) + NO3 (ag) → (3)ON In the final…
A: After balancing the electrons and atoms other than H and O , add water molecules to O deficit side…
Q: Consider the following balanced redox reaction CIO2°(aq) + H20(1) + 2Cu*(aq) → ClO"(aq) + 20H (aq) +…
A: Reducing agent can be defined as the species which undergo increase in oxidation state and itself…
Q: Balance the following reaction in a basic solution: CrO4- + HSnO2- → CrO2- +…
A:
Q: Balance the following chemical equation with the smallest whole numbercoefficients. What is the…
A: The given unbalanced equation is, C4H10 + O2 → CO2 + H2O
Q: Balance the following redox reaction under basic conditions. H2O2(aq) + CIO2(aq)→ CIO2(aq) + O2(g)
A: We have to Balance the Redox Reaction in the Basic medium.
Q: An acid reacts in water to transfer an H to water and form: O hydroxide ion O H;0* (aq) O NH," (aq)…
A: Given : Reaction of water and acid (a) OH- (b) H3O+(aq) (c) NH4+(aq) (d) hydrogen To…
Q: Balance the following in basic solution Te t NO3 Te O3
A:
Q: Complete and balance the following combustion reaction. C2H6O(l) + O2(g) ------>
A: The combustion reaction is given as,
Q: complete and balance the following reactions. If no reaction is possible, write NR. 4. 2Al + 6HCI…
A: The completed and balanced reaction as follows,
Q: For the reaction: Al + O2 ⟹⟹ Al2O3 1. Identify the type of reaction as acid-base,…
A:
Q: Predict the products and balance the following eqn. and identify the type of reaction FeCl 3(aq) +…
A:
Q: Balance the following chemical reaction under acidic conditions: Mn2+ + BiO3- → MnO4- + Bi3+
A:
Q: + IO3 (ag) I, (agi (Acidic)
A: The reaction given is, => I- (aq) + IO3- (aq) -------> I3- (aq) In the above reaction, I is in…
Q: Balance the following equation for acid conditions by the half-reaction method. Identify the…
A: Steps for Balancing the Redox Reaction (Oxidation-Reduction Reaction) in acidic Medium. Step 1 :…
Q: What type of reaction is the following: NaOH + HCI H20 + NACI O Single Displacement O Neutralization…
A: We have to predict the type of reaction.
Q: Q.) Balance the following reaction in acidic solution: ClO;+ SO2 --> SO,¯ + CI¯
A:
Q: Balance the following redox reactions. For each reaction, indicate the species that is oxidized and…
A:
Q: Please Balance the following equation: CH3COOH (aq) + NaHCO3 (s) = CO2 (g) + H2O(l) + NaCH3COO…
A: The chemical equation can be defined as a symbolic representation of the chemical reaction in the…
Q: Balance the following in both acidic and Basic solutions. N2O4(2) + Br(aq) + Br (aq) NO2 (aq) + BrO3…
A: The given redox reaction is: N2O4g+Braq-⇌NO2aq-+BrO3aq-
Q: C7H16 + 02. --> CO2 + H20
A: The given unbalanced equation is: C7H16+O2→CO2+H2O Step 1: Balance the carbon atoms Since, 7 carbon…
Q: Complete and Balance The following equation, in the case that the reaction doesnt occur write "NR"…
A: Given unbalanced and incomplete reaction is : MnO4- + OH- + SO32- --------------> ? Complete…
Q: 7. Write complete balanced equation for the following acid-base reaction: H;PO4(a9) + Ca(OH)2(aq) →
A: 2H3PO4 (aq) + 3Ca(OH)2 (aq) --> Ca3(PO4)2 (s)+ 3H2O
Q: Balance the following redox reactions. Show complete solutions. Indicate the reducing agent and the…
A:
Q: Which of the following metals will react with HCl to form hydrogen gas? A. Hg B. Au C. Cu D. Ag…
A: The following metals will react with HCl to form hydrogen gas has to be given,
Q: Balance the chemical equation below using the smallest possible whole number stoichiometric…
A: In a balanced chemical reaction the no of atoms present at reactant side is equal to the no of atoms…
Q: Balance the following equation by filling the gaps with appropriate numbers. ...........Mn…
A: To understand fully chemical equation reaction must be balanced. In any balanced chemical equation…
Q: (c) NO3 "(ag) + H2(g) NO(g) (d AUSK
A: For balancing an equation, one has to balance number of atoms in both sides of equation. lack of…
Q: 2/2a/kulate the equivalent weigh lezcrzoz) when reducing to (Erst) te=39 coalf
A: The equivalent weight is calculated by formula, equivalent mass=molar masscharge
Q: Complete and balance the following formula equation for the reaction below. Write the complete ionic…
A:
Q: Balance the following redox reaction: CuS(s) + NO3` (aq) à Cu2+(aq) + S(s) + NO (g) (in acid)
A: The balanced half-cell reactions are: S2- = S + 2e- ......(1) NO3- + 4 H+ + 3e- = NO + 2…
Q: The following skeletal oxidation-reduction reaction occurs under acidic conditions. Write the…
A: Oxidation half- cell exhibits the reaction of those species which undergoes oxidation. That is,…
Q: Balance the reaction of NaBr + CaO --> CaBr2 + Na2O
A: In a balanced chemical reaction, the number of atoms of each element is same on the reactant and…
Q: Balance the equation and give the coefficient of the question mark (?). KOH + H3PO, → K3PO, + H20…
A: We will first balanced the given chemical reaction equation. Unbalanced equation is : KOH + H3PO4…
Q: Predict the products and balance the following eqn. and identify the type of reaction HCl + AgNOae)…
A: Balanced chemical equation AgNO3 (aq) + HCl (aq) → AgCl (s) + HNO3 (aq) The reaction type is…
Q: e) OO0
A: Reaction of acid with base gives water and salt. Reaction is called as neutralization reaction. In…
Q: Identify the acid, base and conjugate acid-bases in the following: PO43- + HNO3 → NO3 + HPO42-…
A: a species is said to be acid if it donate positive charge species and its conjugate pair is known as…
Q: Calculate AHm for the following chemical equation, If necessary, balance the chemical equations.…
A: Given unbalanced reaction is : Pb (s) + PbO2 (s) + H2SO4 (l) --------> PbSO4 (s) + H2O (l)…
Q: Balance the following equation in acidic solution: Hg22+…
A: To balance a reaction no. of electrons on LHS and RHS must be same. The balanced reaction for above…
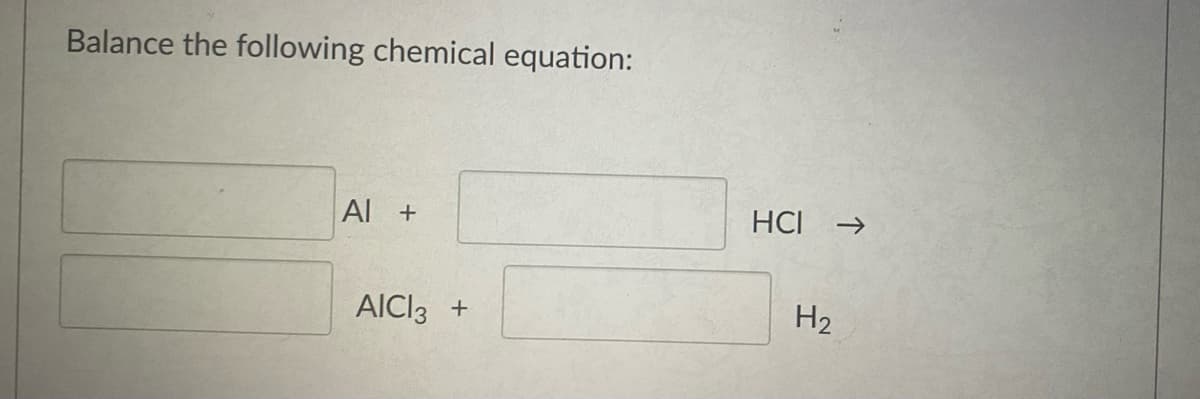
Q: Balance the following equations and identify the type c Al ---> 1. HCI + AICI3 + H2
A: While balancing the reaction, number of atoms in reactant should be equal to number of atoms in…
Q: Balance the given reactions by change in the ox. no. method. Identify the oxidizing agent and…
A: we need to balance the equation PbO2 + HI → PbI2 + I2 + H2O
Q: How do I balance the following equation? C+H2O→CO2+CO+H2
A: A balanced chemical equation is one in which all the atoms are in equal amounts on reactants and…
Q: Complete and balance the following double-displacement chemical equation: ZnCl2(aq) + AgC2H3O2(aq)
A:
Q: Balance the following equation in basic solution using the lowest possible integers and give the…
A:
Q: The following skeletal oxidation-reduction reaction occurs under basic conditions. Write the…
A: Ni(OH)2 + ClO4- à ClO3- + NiO2 Redox equation balancing guidelines Step 1: Construct an…
Q: Balance the following redox equation, for a reaction which takes place in basic solution. HS (aq) +…
A:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Balance the following equations: (a) Ag(s)+H2S(g)+O2(g)Ag2S(s)+H2O(l) (b) P4(s)+O2(g)P4O10(s) (c) Pb(s)+H2O(l)+O2(g)Pb(OH)2(s) (d) Fe(s)+H2O(l)Fe3O4(s)+H2(g) (e) Sc2O2(s)+SO3(l)Sc2( SO4)3(s) (f) Ca( PO4)2(aq)+H3PO4(aq)Ca(H2 PO4)2(aq) (g) Al(s)+H2SO4(aq)Al2( SO4)3(s)+H2(g) (h) TiCl4(s)+H2O(g)TiO2(s)+HCl(g)The carbon dioxide exhaled in the breath of astronauts is often removed from the spacecraft by reaction with lithium hydroxide 2LiOH(s)+CO2(g)Li2CO3(s)+H2O(l) Estimate the grams of lithium hydroxide required per astronaut per day. Assume that each astronaut requires 2.50 103 kcal of energy per day. Further assume that this energy can be equated to the heat of combustion of a quantity of glucose, C6H12O6, to CO2(g) and H2O(l). From the amount of glucose required to give 2.50 103 kcal of heat, calculate the amount of CO2 produced and hence the amount of LiOH required. The H for glucose(s) is 1273 kJ/mol.4-81 (Chemical Connections 4C) Balance the lithium iodine battery redox reaction described in this sec tion and identify the oxidizing and reducing agents present.
- Gold metal will dissolve only in aqua regia, a mixture of concentrated hydrochloric acid and concentrated nitric acid in a 3:1 volume ratio. The products of the reaction between gold and the concentrated acids are AuCl4-(aq), NO(g), and H2O. The equation for this reaction where HNO3 and HCl are strong acids is Au(s)+4Cl(aq)+4H+(aq)+NO3(aq)AuCl4(aq)+NO(g)+2H2O(a) What stoichiometric ratio of hydrochloric acid to nitric acid should be used? (b) What volumes of 12 M HCl and 16 M are HNO3 required to furnish the Cl- and NO3- ions to react with 25.0 g of gold?4-20 Calcium oxide is prepared by heating limestone (calcium carbonate, CaCO3) to a high temperature, at which point it decomposes to calcium oxide and carbon dioxide. Write a balanced equation for this preparation of calcium oxide.Ten mL of concentrated H3PO4 (91.7% by mass, d=1.69g/mL) was accidentally poured into a beaker containing 20.0 g of NaOH. Not all the NaOH was consumed. How many grams of NaOH were left unreacted? The equation for the reaction is H3PO4(aq)+3OH(aq)3H2O+PO43(aq)
- On Easter Sunday, April 3, 1983, nitric acid spilled from a tank car near downtown Denver, Colorado. The spill was neutralized with sodium carbonate: 2HNO3(aq)+Na2CO3(aq)2NaNO3(aq)+H2O(l)+CO2(g) a. Calculate H for this reaction. Approximately 2.0 104 gal nitric acid was spilled. Assume that the acid was an aqueous solution containing 70.0% HNO3 by mass with a density of 1.42 glcm3. What mass of sodium carbonate was required for complete neutralization of the spill, and what quantity of heat was evolved? (Hf for NaNO3(aq) = 467 kJ/mol) b. According to The Denver Post for April 4, 1983, authorities feared that dangerous air pollution might occur during the neutralization. Considering the magnitude of H, what was their major concern?4.19 How many metric tons of carbon are required to react with 7.83 metric tons of Fe2O3 according to the following reaction? 2Fe2O3+3C3CO2+4Fe How many metric tons of iron are produced?A power plant is driven by the combustion of a complex fossil fuel having the formula C11H7S. Assume the air supply is composed of only N2 and O2 with a molar ratio of 3.76:1.00, and the N2 remains unreacted. In addition to the water produced, the fuels C is completely combusted to CO2 and its sulfur content is converted to SO2. In order to evaluate gases emitted at the exhaust stacks for environmental regulation purposes, the nitrogen supplied with the air must also be included in the balanced reactions. a Including the N2 supplied m the air, write a balanced combustion equation for the complex fuel assuming 100% stoichiometric combustion (i.e., when there is no excess oxygen in the products and the only C-containing product is CO2). Except in the case of N2, use only integer coefficients. b Including N2 supplied in the air, write a balanced combustion equation for the complex fuel assuming 120% stoichiometric combustion (i.e., when excess oxygen is present in the products and the only C-containing product is CO2). Except in the case of use only integer coefficients c Calculate the minimum mass (in kg) of air required to completely combust 1700 kg of C11H7S. d Calculate the air/fuel mass ratio, assuming 100% stoichiometric combustion. e Calculate the air/fuel mass ratio, assuming 120% stoichiometric combustion.
- Magnesium metal (a component of alloys used in aircraft and a reducing agent used in the production of uranium, titanium, and other active metals) is isolated from sea water by the following sequence of reactions: Mg2+(aq)+Ca(OH)2(aq)Mg(OH)2(s)+Ca2+(aq)Mg(OH)2(s)+2HCl(aq)MgCl2(s)+2H2O(l)MgCl2(l)electrolysisMg(s)+Cl2+Cl2(g) Sea water has a density of 1.026 g/cm3 and contains 1272 parts per million of magnesium a5 Mg2+(aq) by mass. What mass, in kilograms, of Ca(OH)2; is required to precipitate 99.9% of the magnesium in 1.00103 L of sea water?Chlorine gas was first prepared in 1774 by C. W. Scheele by oxidizing sodium chloride with manganese(IV) oxide. The reaction is NaCl(aq)+H2SO4(aq)+MnO2(s)Na2SO4(aq)+MnCl2(aq)+H2O(l)+Cl2(g) Balance this equation.Balance the following equations: (a) for the reaction to produce "superphosphate" fertilizer Ca3(PO4)2(s) + H2SO4(aq) Ca(H2PO4)2(aq) + CaSO4(s) (b) for the reaction to produce diborane, B2H6 NaBH4(s) + H2SO4(aq) B2H6(g) + H2(g) + Na2SO4(aq) (c) for the reaction to produce tungsten metal from tungsten(VI) oxide WO3(s) + H2(g) W(s) + H2O() (d) for the decomposition of ammonium dichromate (NH4)2Cr2O7(s) N2(g) + H2O() + Cr2O3(s)











