Q: How many grams of H₂O are consumed to generate 13 g C₂H₂ via the process below? CaC₂ (s) + 2 H₂O (g)…
A:
Q: Given: 0.35g NaCl, 0.25 g NaHCO3, 0.15 g KCl & 2 g C6H12O6 are present in 100 mL ORS solution (MW:…
A: Given: 0.35g NaCl, 0.25 g NaHCO3, 0.15 g KCl & 2 g C6H12O6 are present in 100 mL ORS solution…
Q: reactant: 0.400g of p-amionphenol (MW=109.1) 0.450 ml of acetic anhyride (mw= 102.1 d= 1.08 ml)…
A: Hello. Since the question contains more than three subparts, the first three subparts shall be…
Q: [0] OH Consider the molecule above. If treated with chromate solution, {"version":"1.1","math":"…
A:
Q: 1.16 Ti(a) transforms to Ti(B) at 882°C (1155 K), with a heat of transforma- tion equal to 830…
A:
Q: What else is produced during the combustion of propane, C3H3? C3HB +502 3 CO2 +4-- O H20 O C3H8 O2 O…
A:
Q: A sample of processed meat scrap weighing 4.000 g is digested with concentrated H;SO, and…
A: Given: Weight of meat scrap sample = 4.000 g To calculate the percentage protein in the meat scrap.
Q: The ascorbic acid, C6H806 (176.124 g/mmol) content of a bottle of peach juice containing 2.50 L was…
A: I have attached a detailed solution for the question in the following steps:
Q: what is the coefficient in front of the O2 when the equation is balanced (using only whole numbers…
A: Given, Consider the reaction: C2H6O(g) +O2(g) ------>CO2(g) +H2O(g) The coefficient in front of…
Q: Calculate AG° for the below chemical reaction using the provided AG° values. 2 PCI3 (g) + O2 (g) → 2…
A: Given: ∆Go POCl3(g) = -502.5 kJ/mol ∆Go PCl3(g) = -269.6 kJ/mol ∆Go O2(g) = 0 kJ/mol
Q: Given: Ca2+(aq) + 2e → Cd(s) EO = -0.40 V %3! Co2+(aq) + 2e Co(s) EO = -0.28 V Calculate the…
A: Given information:
Q: Add Stoichmetric factors: ______Cr+ _______H20 yields ______Cr3O4+ +________H2
A:
Q: Voltammetry is limited to analysis of inorganic samples only True or false
A: Answer: False Voltammetry is an accurate electro-analysis method and combination with…
Q: Oa is required for complete Combustion
A: Number of moles = mass/molar mass Mass = moles × molar mass
Q: Complete and balance the following reactions. Don’t worry about state symbols for the second one.…
A: Introduction: The chemical equation shows the reaction between the two or chemical species and their…
Q: 6NACI+ Fe,(PO,), 1. 3FECI, + 2Na,PO, Single replacement or double replacement? -- Туре: CO, + 2H,0…
A:
Q: A www-awn.aleks.com Significant Figures Counter Chapter 5. The Nervous System-Neurology and…
A: Given, the mass of ammonium perchlorate = 9.30 g The balanced chemical equation is given - The…
Q: t was known to treat skin diseases. It was used for laundering and washing with much less effort. t…
A:
Q: Write balanced formation equations for the following, indicate proper states:…
A: The compounds given are N2O (g), Na2S2O7 (s), C2H6O (l) and B3N3H6 (s). Formation reactions are…
Q: Show the process of Balancing: 1. Zn ǀ 0.10M Zn(NO3)2 ; 0.10M CuSO4 | Cu
A:
Q: 1. Iron (Fe) nail + CuSO4 solution ? turned into the rusty Iron (Fe) nail turned into black…
A: We have to predict product of reaction, and name of salt.
Q: Al3 IЕО%3-1.66 V + Зе --> Al Tit2 + 2e -> Ti E° =-1.63 V
A: According to the given SRP values "Ti " metal has more reduction potential , hence it acts "Ti…
Q: How many miles of benzene should be completely combusted in order to produce .4000 mil of carbon…
A: The combustion reaction is, 2 C6H6 + 15 O2 -----> 12…
Q: Complete the combustion products of sucrose. Balance the equation. C12H22011 O2 Name of products:…
A: Combustion is the process of burning of a compound; in which the compound reacts with excess of…
Q: Calcium carbide reacts with water to produce acetylene (C2H2) according to the CaC2 equation below:…
A:
Q: A mixture contains Na2CO3, NaOH, and inert matter. A sample weighing 1.500 g requires 28.85 mL 0f…
A:
Q: True or false?
A: In this question we have to find correct and incorrect reaction also the real form of the incorrect…
Q: When the equation Fe2(C204)3 → FeC204 + CO2 is balanced with the smallest integer coefficients, the…
A: Since you have asked multiple type questions, we will solve only first questions for you. If you…
Q: Suppose you standardize a Na2S203 solution by dissolving 25.0 mg of KIO3 in acid and allow it to…
A:
Q: For each reaction, predict the product(s), balance the equation, and note the reaction type. 1.…
A: “Since you have posted a question with multiple sub-parts, we will solve the first three subparts…
Q: What is the coefficient in front of potassium hydroxide when the equation is balanced using smallest…
A: The reaction given is S (s) + O2 (g) + KOH (aq) → K2SO4 (s) + H2O (l) In the above reaction,…
Q: Which of these reactions does not occur? -) NICHS CH₂OH OCH₂ c) 808 / Ho a ca CHÍNH, 90
A: The reactivity of the acyl compound depends on the partial positive charge present on the carbonyl…
Q: Predict the product/s of the following reactions AND identify the type of reaction taking place.…
A: The various types of reactions are:- Combination Reaction Displacement Reaction Decomposition…
Q: The price of (a) CoCl•6H2O is $25.00 per 100 g and (b) NH,CI was $11.35 per 500 g. Calculate the…
A:
Q: Predict the products and balance the following eqn. and identify the type of reaction 7) Bacl +…
A: The given reaction are as follows, a) BaCl2 (aq) + H2SO4 (aq) → b) C6H12 (g) + O2 (g)→ c) Zn(s)+…
Q: An iron ore contains iron (II) carbonate, FeCO1. The percentage of iron (II) in the ore can with…
A: The question is based on quantitative estimation of iron carbonate present in the iron ore. it is…
Q: immediate precursor of several useful plastics. In the first step, calcium carbide (cac₂) and water…
A: Here we are required to find the mass of calcium carbide needed to make 6 kg of acrylic acid
Q: [1] LİAIH4 A HO, [2] H2O он NaBH4, HO, ethanol Ethanol, H2SO4 (aq) HO,
A: The first reaction is the reduction reaction as the acid group will be treated with a reducing agent…
Q: Part I -- Chemical Reactions: Record your observations below, and write a balanced equation for each…
A: Since you have asked a question with multiple sub-parts we will solve the first few subparts for you…
Q: 1. NaNH, 2 Br 3. H₂, Lindlar's catalyst
A:
Q: Complete the following equations to show the products that form in each of the reactions we observed…
A: Solution: The balanced chemical equation are as follows: (a) Ca(s) + 2 H2O(l) → Ca(OH)2 (aq) + H2…
Q: Which series is oxidized? 8NaI + 5H2 SO4 ---- 4I2 + H2S + 4Na2SO4 +4H2O
A: 8NaI+5H2SO4 → 4I2+H2S+4Na2SO4+4H2O Oxidized means oxidation number increases after the…
Q: Which reactant is limiting in a propane fire? O a) propane O b) charcoal O c) oxygen O d) water
A: When propane burns , it requires oxygen gas for combustion available in environment, to give Carbon…
Q: Write a balanced equation for each of the following reactions.(You may have to guess at one or more…
A: a. The reaction for the above process is as follows: Al2Se3(s) + 6H+(aq) --------------->…
Q: alcl3+ 3agno 3 --> al(no3)+3agcl combination? decompositiion? single replacement? or double…
A:
Q: It may be necessary to refer to the lecture textbook or the internet to answer some of reference if…
A: The unbalanced equation is C5H12 + O2 --------> CO2 + H2O Balance carbon: C5H12 + O2…
Q: Write the balanced combustion equation. Record fractions as ratios (e.g. 1/2) if needed. Include…
A: Since you have posted multiple questions, the answer for first question is given below. Kindly…
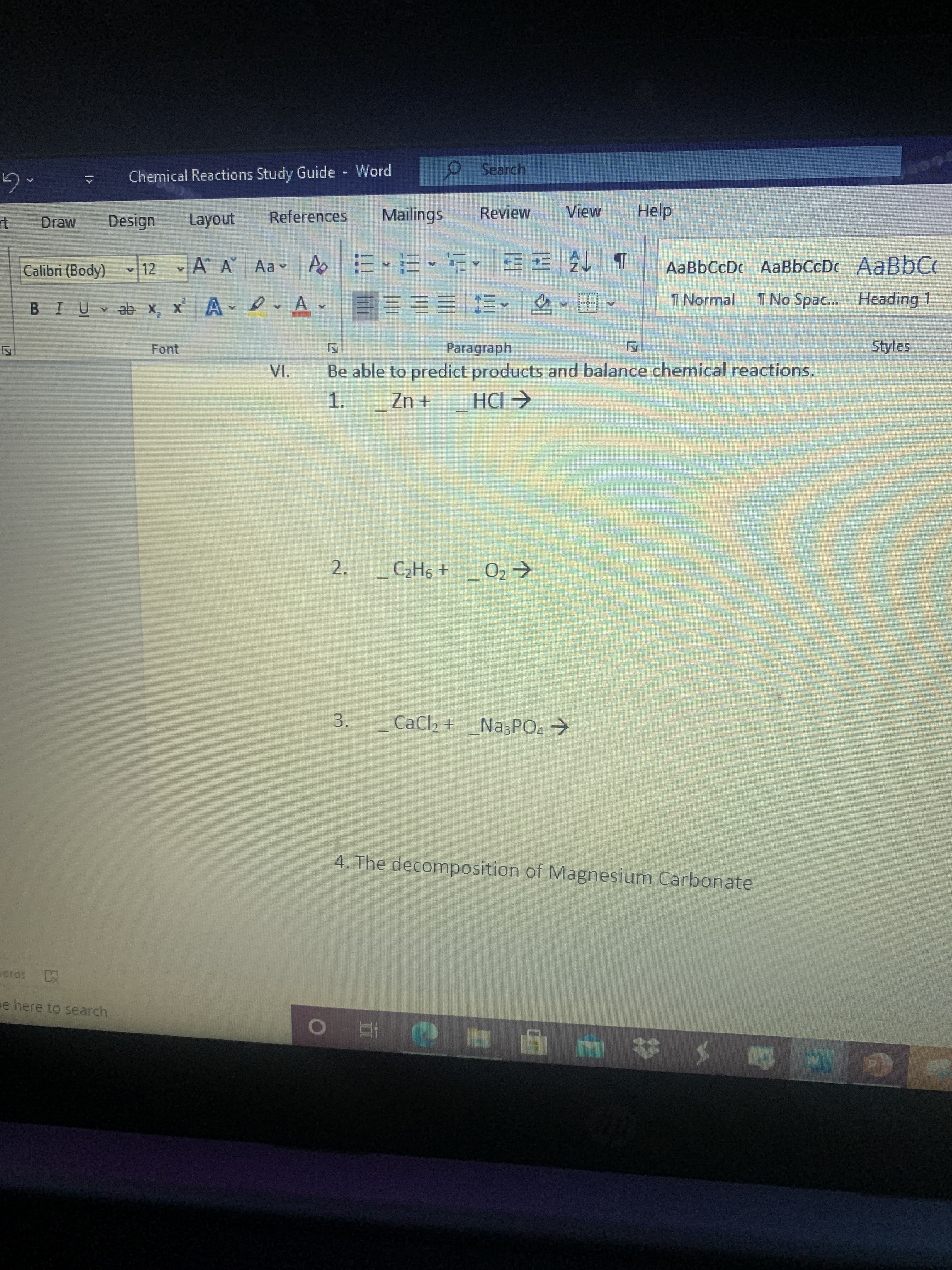
Need help on study guide
Step by step
Solved in 3 steps

- Quickly plezz!! The levigating agent role in Suppositories manufacturing ??Solution for 0.105g of the K3[Fe(C2O4)3] (MW= 437.2 g/ mol) were dissolved in sulfuric acid and got rated with 9 mL of 0.028M KMn04. What is the experimental weight percent of oxalate in the sample?Hello, I do not understand this quiestion or how to do this problem. Can you help me so I can follow along. 7. 5.00 mL of stock solution is diluted to 25.00 mL, peoducing solution ALPHA. 10.00 mL of solution ALPHA is diluted to 25.00 mL resulting in solutin BETA. 10.00 mL of solutionBETA is then diluted 25.00 mL producing solution Gamma. a. Determine the dilution factor in produciing solution Alpha from the stock solution. b. Determine the dilution factor in producing solution Beta from the solution Alpha c. Determie the dilution factor in producing solution Gamma from the stock solution Beta. d. Determine the overall serial dilution factor in producing solution Gamma form the stock solution.
- Balance these equations by providing the missingcoefficients:(a) __ Fe1s2 + __O21g2¡__Fe2O31s2(b) __ Al1s2 + __ HCl1aq2¡__ AlCl31aq2 + __ H21g2(c) __ CaCO31s2 + __ HCl1aq2¡__ CaCl21aq2 +__ CO21g2 + __ H2O1l2Give typed explanation of all partsSOLUBILITY – reactions with conc. Sulfuric acid Based from the video link --- https://www.youtube.com/watch?v=JmlDkC6IdKA&t=21s 1 mL of conc. H2SO4 was placed in a separate test tube. The test samples, cyclohexane and cyclohexene were added separately. Observe what happened after the reaction. Question: Based from your observation, write the results of the following samples: (a) Cyclohexane, (b) Cyclohexene, and (c) Toluene. Thank you very much!
- At constant pressure and 25°C, what is ΔrH° for the following reaction2C2H6(g) + 7O2(g) → 4CO2(g) + H2O(l)if the complete consumption of 14.5 g of C2H6 liberates 752.3 kJ of heat energy?a. Give the molar concentration of hydrochloric acid with a specific gravity of 1.18 andhas 37% (w/w) purity. MW=36.5 b. How would you prepare 1.2 L of 0.40 M hydrochloric acid starting from a 2.0 M solution?Including the state of reaction (aq),(l),(g),etc Thank you

