Binds here (1) and is changed to 2. Must bind here in order for reaction to occur Binds here (1) and prevents reaction from occurring Binds here and prevents reaction from occurring 1. 2. 5. 3. 4.
Binds here (1) and is changed to 2. Must bind here in order for reaction to occur Binds here (1) and prevents reaction from occurring Binds here and prevents reaction from occurring 1. 2. 5. 3. 4.
Chapter9: Parenteral Medication Labels And Dosage Calculation
Section: Chapter Questions
Problem 4.7P
Related questions
Question
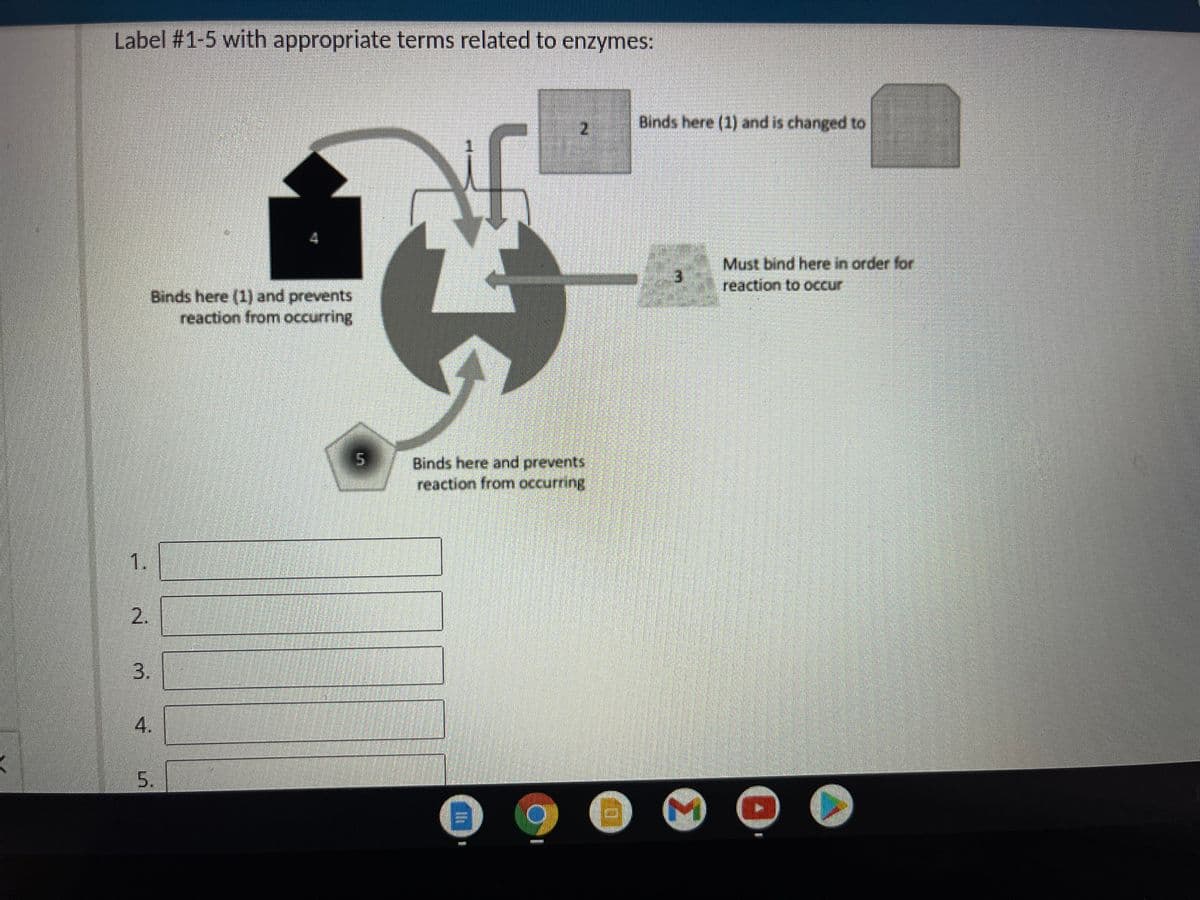
Transcribed Image Text:Label #1-5 with appropriate terms related to enzymes:
Binds here (1) and is changed to
Must bind here in order for
reaction to occur
3.
Binds here (1) and prevents
reaction from occurring
Binds here and prevents
reaction from occurring
1.
2.
3.
4.
5.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you





