Boiling Surface Point Tension Viscosity (C) (1/m?) (kg/m-s) Water, H20 100 7.3 x 10-2 0.9 x 10-3 Ethanol, CH3CH2OH 78 2.3 x 10-2 1.1 x 10-3 Propanol, CH3CHLCH2OH 97 2.4 x 10-2 2.2 x 10-3 п-Butanol, CH,CH,СH,CH,ОH 117 2.6 x 10-2 2.6 x 10-3 Ethylene glycol, НОСH-CH,OH 197 4.8 x 10-2 26 x 10-3
Boiling Surface Point Tension Viscosity (C) (1/m?) (kg/m-s) Water, H20 100 7.3 x 10-2 0.9 x 10-3 Ethanol, CH3CH2OH 78 2.3 x 10-2 1.1 x 10-3 Propanol, CH3CHLCH2OH 97 2.4 x 10-2 2.2 x 10-3 п-Butanol, CH,CH,СH,CH,ОH 117 2.6 x 10-2 2.6 x 10-3 Ethylene glycol, НОСH-CH,OH 197 4.8 x 10-2 26 x 10-3
Chemistry: The Molecular Science
5th Edition
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:John W. Moore, Conrad L. Stanitski
Chapter9: Liquids, Solids, And Materials
Section: Chapter Questions
Problem 106QRT
Related questions
Question
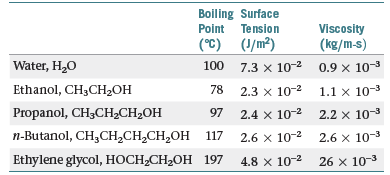
The boiling points, surface tensions, and viscosities of
water and several alcohols are as shown below:
(a) From ethanol to propanol to n-butanol the boiling
points, surface tensions, and viscosities all increase. What
is the reason for this increase? (b) How do you explain
the fact that propanol and ethylene glycol have similar
molecular weights (60 versus 62 amu), yet the viscosity of
ethylene glycol is more than 10 times larger than propanol?
(c) How do you explain the fact that water has the
highest surface tension but the lowest viscosity?
Transcribed Image Text:Boiling Surface
Point Tension
Viscosity
(C) (1/m?)
(kg/m-s)
Water, H20
100 7.3 x 10-2 0.9 x 10-3
Ethanol, CH3CH2OH
78
2.3 x 10-2 1.1 x 10-3
Propanol, CH3CHLCH2OH
97 2.4 x 10-2
2.2 x 10-3
п-Butanol, CH,CH,СH,CH,ОH 117
2.6 x 10-2 2.6 x 10-3
Ethylene glycol, НОСH-CH,OH 197
4.8 x 10-2 26 x 10-3
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning