Q: Name Lewis structure 3-dimensional drawing 1. n-hexane (C&H14) 2. 1-chloroethene (C:H;CI) 3. ethyne
A: Lewis structures are referred as, Lewis dot structures or electron dot structures represents the…
Q: Free Response Questions in this section are not weighted equally. Allocate your not (11) gepo to…
A:
Q: Nonbonding Electron Valence Shell Bonding Electron Аpprox. Bond Molecule or Lewis Electron VSEPR…
A: Lewis structures are representations of molecules that include all the electron pairs i.e. bonding…
Q: Arrange the highlighted bonds in the table below in decreasing order of polarity. That is, pick 1…
A:
Q: Arrange the highlighted bonds in the table below in decreasing order of polarity. That is, pick 1…
A:
Q: bond, show the direction of polarity by selecting the correct partial charges. I-Br F-I F-Br polar…
A:
Q: ange the highlighted bonds in the table below in decreasing order of polarity. That is, pick 1 for…
A:
Q: D. Structure of Molecules and Molecular lons On a separate sheet of paper, set up the following…
A: As per the bartleby guidlines , I can solve only first three questions of multipart or only first…
Q: Which of the highlighted chemical bonds in the molecules below is longest? Shortest? In between?…
A: Bond length is defined as the average distance between nuclei of two bonded atoms in which bond is…
Q: Which of the following Lewis structures best describes BF3 :F : :F -B- F :F : F-B- F .. :F: -B-F: :F…
A: The question is based on the concept of chemical bonding. We have to identify the correct Lewis…
Q: Name the highlighted chemical group in each molecule. Lewis structure name of highlighted group Н—…
A: Highlighted groups 1. NH2 2. OH 3. C=O
Q: Nonbonding Bonding Electron Valence Shell Approx. Bond Molecule or Lewis Electron Electron VSEPR…
A: The molecules given are PCl2F3, BrF3, XeF2.
Q: You will not turn in a formal write-up for this experiment. Turn in the completed Lewis dot…
A: Lewis structure is also known as electron dot structure. It represents the total electrons in the…
Q: 3. Draw structural formulas for the following covalently bonded molecules, representingeach covalent…
A:
Q: Match each of the Lewis structures (a to c) with the correct diagram (1 to 3) a. X-Y-X 1. 1 b. :Y-X…
A: We have to match Lewis structure with the diagram.
Q: Valence Shell Bonding Nonbonding Approx. Molecule or Lewis Electron Electron Electron VSEPR Bond…
A: Lewis structure is an electron-dot structure in which the electrons are represented by dots. The…
Q: For the molecules below use formal charge to identify the specific atom(s) which hold indicated…
A:
Q: Directions: Complete the data table below. It is not necessary to construct any models. Chemical…
A: We are authorized to answer three subparts at a time, since you have not mentioned which part you…
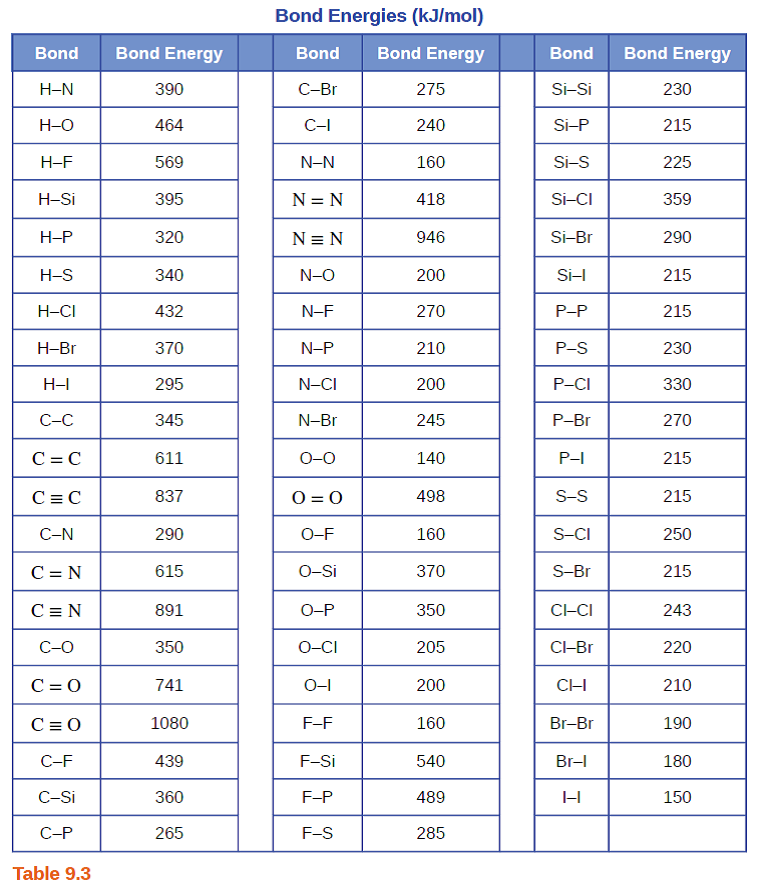
Q: Use bond energies listed in Table available on Canvas to find AHx for the reactions shown below: 1)…
A:
Q: 9.61 Use Figure 9.21 to indicate the polarity of each bond with partial charges: (а) Br—CI (d) Se-H…
A: We know, Greater is the electronegativity value more polar is the atom/molecule.
Q: Choose the correct ranking for the following bonds in terms of increasing bond length. F-H, N-N,…
A: 1- In this question, we will arrange bond length in increasing order. In F-H, O-H and N-H size of F…
Q: Rank the following bonds from least polar to most polar. Use numbers from 1 (least) to 4 (most) C-F…
A: We Know that, Electronegativity of C = 2.55 Electronegativity of F = 3.98 Electronegativity of Cl =…
Q: selum trome Choose the correct arrangement of the following bonds in terms of increasing ionic…
A: Choose the correct arrangement of the following bonds in terms of increasing ionic character. H-C,…
Q: Arrange the highlighted bonds in the table below in decreasing order of polarity. That is, pick 1…
A: the polarity of the bond depends upon the electronegativity of atoms The higher electronegativity…
Q: Draw a Lewis structure for cyanide ion, CN¯, adding charges and lone electron pairs to the…
A:
Q: С-Н -2800 – 3100 C=0 ~1700 – 1800 С-О ~1000 – 1200 1 a) What chemical bond is present in Unknown 1:…
A: 1 a) Carbonyl group is present in Unknown 1( a peak observed about 1700 cm-1 ). Double bond between…
Q: Rank the following in order of increasing ionic character of their bonds. i.e. the one with the…
A: The ionic character of the compound depends on Electronegativity difference between constituent…
Q: Calculate AEn for the following bond and classify it according to type: CsF O A. 3.3; ionic O B.…
A: We have to calculate the ∆En and classify the bond type.
Q: Which of the following is an example of an ionic bond? A) K -F B) C - H C) Kr - H D) C -F
A: Ionic bonds are the type of bond that is formed by the complete transfer of electrons from one atom…
Q: Arrange the highlighted bonds in the table below in decreasing order of polarity. That is, pick 1…
A:
Q: Which choice(s) correctly rank(s) the bonds in terms of increasing polarity? I. CI-F < Br-Cl < Cl-CI…
A: The question is based on the concept of polarity of the bonds. Higher the electronegativity…
Q: Cation Anion Bond length (pm) M2+ Q 207 M+ R- 196 M2+ 190
A:
Q: Be sure to answer all parts. Identify the correct Lewis electron-dot structures that would bond to…
A: The Lewis structure or molecular skeleton is also known as electron dot structure. It is the…
Q: Draw the Lewis structures for the following species: (a)CN-; (b) [N3]-; (c) [SCN]-
A:
Q: In assembling a Lewis Dot diagram of NO3−, there are ____ total electrons to use in the model
A:
Q: Arrange the highlighted bonds in the table below in decreasing order of polarity. That is, pick 1…
A: polarity is a separation of electric charge leading to a molecule with a negatively charged end and…
Q: Which of the following atom(s) below has/have five valence elec A) C B) N C) O D) F E) All of the…
A: The question is based on concept of electronic configuration. by writing the electronic…
Q: Complete the Lewis Structure of the molecule below. Then answer the following: CN a 5 3 2 1 H2C=ċ-N:…
A:
Q: Complete the chart below: Mol N,H, mol N,0, mol N, mol H,0 2 5.5 3 12.4 |
A: First of all calculate the number of moles of particular atom present no. of moles of N2O4 is…
Q: CH,(g) + 20,(g)→ CO,(g) + 2 H,0(g) Bond AH° (kJ/mol) 0–0 142 H-0 459 С-Н 411 C=0 799 O=0 498 С-О 358
A:
Q: Which one of the following bonds has the most ionic character? A) B - F B) Li - F C) H - F D) Be - F
A: Ionic character is the amount of electron sharing between two atoms; minimum electron sharing…
Q: Arrange the highlighted bonds in the table below in decreasing order of polarity. That is, pick 1…
A:
Q: Show Lewis Dot structures for the following compounds. a andb are ionic; c and d are molecular. For…
A: Since we only answer up to 3 sub-parts, we’ll answer the first 3. Please resubmit the question and…
Q: Identify the following bonds in the correct order in terms of decreasing polarity Select one: O a.…
A: Where, given that Cl is common to all of the bonds, the relative difference in electronegativity As,…
Q: A. Complete the table Bond formation according What happened to the Type of compound formed Given to…
A: Chemical bonds are generally three types ,which are: #1.Polar bond (a type of covalent bond)…
Q: Which of the following bonds is a nonpolar covalent bond? A) H - F B) N - F C) F - CI D) F -F
A: Polarity of bonds in terms of electronegativity of the atoms bonded together is given by…
Q: Calculate AEn for the following bond and classify it according to type: PB15 O A. -11.9; polar…
A: Note - Since you have asked multiple questions, we will solve the first question for you. If you…
Q: Use the following table to rank these bonds in order of decreasing bond length. Bond strengt…
A: As we know, Higher the bond energy, stronger is the bond. If bond is stronger, then shortest is…
Q: 0-H H-C-H H-N-C-C-0-H H a. Redraw the lewis structure given in 3a. of this assignment in 3…
A: Applying concept of polarity and hybridization by which we can determine polar molecules and bond…
Q: What would be the correct ratio between the following ions y1+ and x2- ?
A: At first we have to write the chemical formula from respective ions.Then we can find the ratio of…
Please answer the following three questions below with reasoning.. thank you

Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 3 images

- Give a clear handwritten answer with explanation....please give answer all sub points...pleasePlease fill the complete Answer Please answer very soon will give rating surelyThe previous answer I was given was incorrect. Please be sure of your information. Read the question thoroughly and explain. Thank you.

