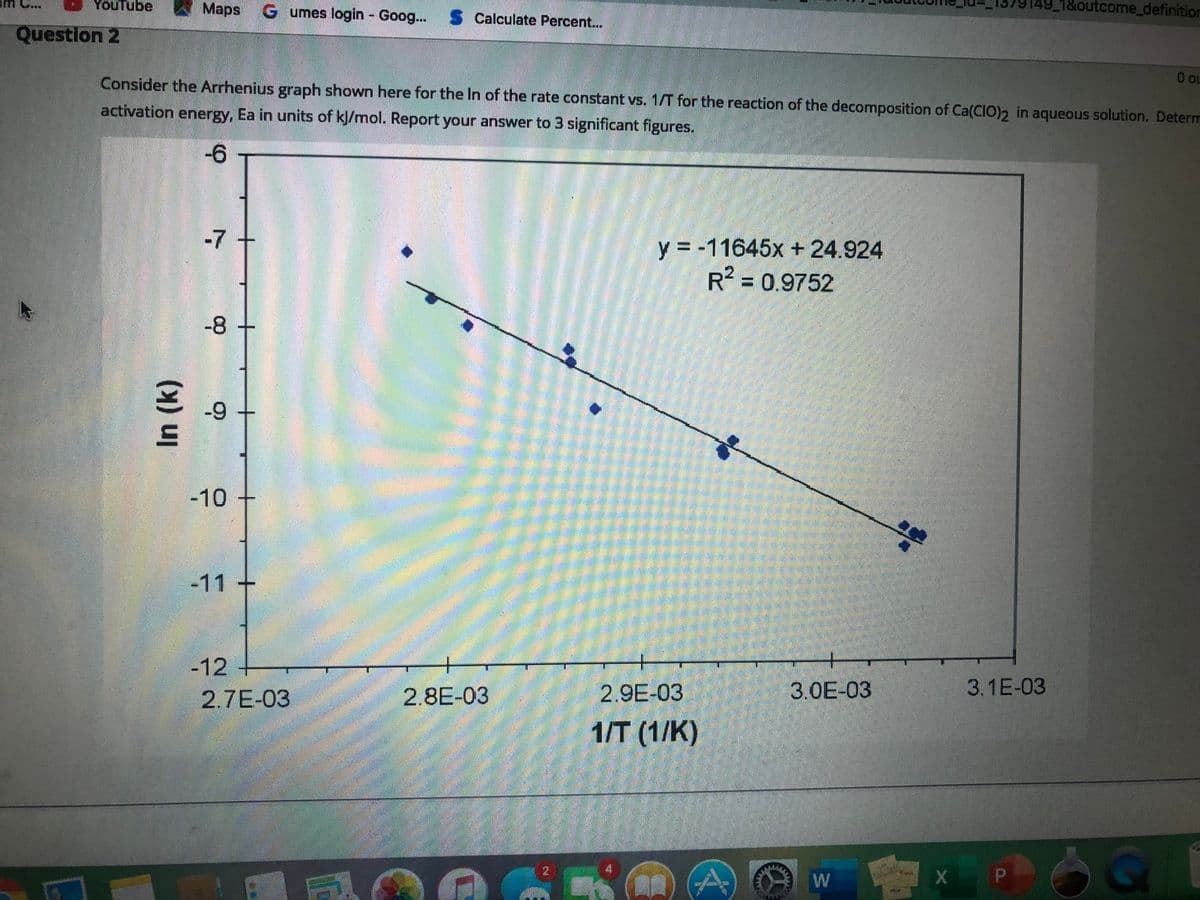
C.. YouTube Maps G umes login - Goog. 1&outcome_defi Calculate Percent.. Question 2 Consider the Arrhenius graph shown here for the In of the rate constant vs. 1/T for the reaction of the decomposition of Ca(CIO)2 in aqueous solution. De activation energy, Ea in units of kl/mol. Report your answer to 3 significant figures. -6 -7 y = -11645x + 24.924 R2 = 0.9752 -8 -9 -10 -11 + -12 2.7E-03 2.8E-03 2.9E-03 3.0E-03 3.1E-03 1T (1/К) In (k)
C.. YouTube Maps G umes login - Goog. 1&outcome_defi Calculate Percent.. Question 2 Consider the Arrhenius graph shown here for the In of the rate constant vs. 1/T for the reaction of the decomposition of Ca(CIO)2 in aqueous solution. De activation energy, Ea in units of kl/mol. Report your answer to 3 significant figures. -6 -7 y = -11645x + 24.924 R2 = 0.9752 -8 -9 -10 -11 + -12 2.7E-03 2.8E-03 2.9E-03 3.0E-03 3.1E-03 1T (1/К) In (k)
Chemistry by OpenStax (2015-05-04)
1st Edition
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Chapter12: Kinetics
Section: Chapter Questions
Problem 32E: Describe how graphical methods can be used to determine the order of a reaction and its rate...
Related questions
Question
Consider the Arrhenius graph shown here for the ln of the rate constant vs. 1/T for the reaction of the decomposition of Ca(ClO)2 in aqueous solution.
Determine the activation energy, Ea in units of kJ/mol.
Report your answer to 3 significant figures.
Transcribed Image Text:C.
149_1&outcome_definition
YouTube
Maps
G umes login - Goog..
S Calculate Percent...
Question 2
Consider the Arrhenius graph shown here for the In of the rate constant vs. 1/T for the reaction of the decomposition of Ca(CIO), in aqueous solution. Determ
activation energy, Ea in units of kJ/mol. Report your answer to 3 significant figures.
-6
-7
y = -11645x + 24.924
R2 = 0.9752
-8
-9
in
-10
-11+
-12
2.8E-03
2.9E-03
3.ОЕ-03
3.1E-03
2.7E-03
1/T (1/K)
is
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning