Q: Carla and her friends were tasked with determining the heat of reaction for the neutralization of…
A: The reaction between HX and NaOH can be represented as: HX+NaOH→NaX+H2O Number of moles are…
Q: molar and normal concentration
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: 2MNO, + 5C,0²- + 16H* → 2Mn²+ + 10 CO, + 8H,0 4. Here, 20 mL of 0.1 M KMNO, is equivalent to
A: Given:- 2MnO4 _ +5C2O42- +16 H+ -------->2Mn+2 +10CO2 + 8H2O. To find:- 20 mL of 0.1 M KMnO4…
Q: prepare ( 0.3 M ) of HCl in 250 ml water, when you know Sp.Gr=1.18 and % = 36% . At.Wt / H=1 , Cl=…
A: Given = Specific gravity = 1.18g/mL Mass % of HCl = 36 % Molar Mass Of HCl= At Wt. of H + At. Wt. Of…
Q: Compute the weight (g) of solute needed to make the solutions listed below: a. 1250 mL of 0.85 M…
A: Hello. Since your question has multiple parts, we will solve the first question for you. If you want…
Q: Compute the weight (g) of solute needed to make the solutions listed below: a. 1250mL of 0.85M AlCl3…
A: We'll answer the first question since the exact one wasn't specified. Please submit the question…
Q: Ascorbic acid (MW = 176.12 g/mole) is readily oxidized by molecular iodine in the presence of acid…
A: Oxidation reactions are those reactions in which oxidation number increases whereas reduction…
Q: 7. The density of concentrated ammonia, which is 28.0% w/w NH3, is 0.899 g/m What volume of this…
A: Given, Mass percentage of NH3 in initial solution = 28.0% Density = 0.899 g/mL Final volume = V2…
Q: 2.1g of sample are dissolved in methylene chloride and the sample is extracted with aqueous…
A: Given, weight of the sample = 2.1g Isolated weight of compound, ethyl-p-aminobenzoate = 0.8g…
Q: 13-1. Describe the preparation of 2.000 L of 0.0500 M AGNO; (169.87 g/mol) from the primary…
A: Detail description is shown below
Q: PRE-LAB QUESTIONS 1. Calculate the molarity of a 6.15% (w/v%) sodium hypochlorite (NAOCI) solution.…
A: A question based on quantitative analysis, which is to be accomplished.
Q: Carla and her friends were tasked with determining the heat of reaction for the neutralization of…
A: Given: Concentration of NaOH = 0.05 M Volume of NaOH = 20.00 mL Concentration of HCl = 0.100 M…
Q: What is the molarity of an unknown HCl solution with a specific gravity of 1.10 and an assay…
A:
Q: Calculate the osmolarity (mOsm/L) of D51/2NS (dextrose 5%/0.45% NaCl). Round your answer to the…
A:
Q: Calculate the osmotic pressure associated with 150.0 g of enzyme (MW=45,000 g/mol) dissolved in…
A: The pressure created by the flow of solvent molecules from a less concentrated solution to a more…
Q: Lahen 9 5o1d is Impure, Its muting Point is higher and broader Tran the muhng unt fur pure Sampleã.…
A: When a pure substance is added with an impurities than the Properties like Melting point, boiling…
Q: What is the solubility of CdS (Ksp = 1.0 x 10-28) in pure water? Group of answer choices 1.0 x…
A: The solubility reaction is CdS (s) -----> Cd2+ (aq) + S2- (aq) Hence Ksp = [Cd2+][S2-] where…
Q: Barium sulfate is a contrast agent for X-ray scans that are most often associated with the…
A: Answer:- This question is answered by using the simple concept of calculation of molar solubility…
Q: 10 CCh and diluting to 150.0 mL. a Calculate moles of Na", Ca³" and Cl- in the solution b. Using…
A: a.) To calculate moles of each ion, we would first Calculate moles of NaCl and CaCl2 solution . Then…
Q: Calculate the solubility of CuCO3 in water at 25C. Ksp = 3.00x10-12. Round the answer to two…
A: The equilibrium between the soluble ions and insoluble salt can be described by the solubility…
Q: Suppose that a volume of 5.0 cm³ of acetic anhydride (density = 1.08 g·cm⁻³, molar mass = 102.087…
A:
Q: Please answer as soon as possible. Thank you! A 10.000-g cocoa powder sample was dissolved in…
A: First of all, we have to convert the concentration of Fe given in micromolar to the grams of Fe in…
Q: Indicate which of the two filtration techniques, gravity ( G ) or vacuum ( V ), is suitable for each…
A: Given operations are a) Hot filtration b) Removal of decolourising carbon c) Isolation of…
Q: 3. Calculate the molar concentration of a working solution produced by diluting 50.00mL of a 1.00 x…
A:
Q: 2- A solution of concentrated HCl has specific gravity of 1.185 and percentage is 36.5 % (w/w) HCl.…
A: Formality is concentration term, it is the number of formula mass in grams present in one liter of…
Q: if 361.3 mg of Li2(SO4) (FW=109.95) is added to 450 mL of 0.828 M NH4F, what is the Qip for lithium…
A: Given, Mass of Li2SO4 = 361.3 mg Formula weight of Li2SO4 = 109.95 g/mole Volume of NH4F = 450 mL…
Q: Dissolution 0.01 M HCI Distilled H20 Concentration (mg/ml) Total amount Concentration (mg/ml) Time A…
A: Dissolution is the process where a solute in gaseous, liquid, or solid phase dissolves in a solvent…
Q: Calculate serial dilution. If we pipette 1 mL of sample into tube of 9 mL of 0.1 peptone water.…
A: A stock solution made from 25 g of sample, for whom calculation for serial of dilution is to be…
Q: What mass of sodium acetate must be added to 435 mL of water to give a solution with pOH = 4.65? Kb…
A:
Q: Calculate the concentration of concentrated HCl in molarity. Concentrated HCl has 38% (w/w) HCl and…
A:
Q: 25- How to prepare 70% (v/v) Ethanol in water? (Ethanol is absolute, Mwt 46 g/mol) A) By Diluting…
A: The concentration in v/v of some solute means the volume of solute that is present in a certain…
Q: The standard concentration of a CdCl2 barium chloride solution containing 562 mg / liter is…
A: Note : CdCl2 is cadmium chloride but in question Barium chloride is written .Therefore name should…
Q: Convert the BOD concentration of 160 mg/L in the primary effluent into BOD loading rate in terms of…
A: The explanation is given below-
Q: A solution was prepared by dissolving 1210 mg of K3Fe(CN)6 (329.2 g/mol) in sufficient water to give…
A: Molarity is defined as number of moles of solute per liter of solution. M = n/V where, M = molarity…
Q: What is the final concentration if 100 mL of a 20% w/v solution is diluted to 250 mL with water?…
A: Correct answer is (A) 8% w/v. Amount of solute remains constant during dilution. So, mass of solute…
Q: You dissolve 14 g of gv03/2 in water and dilute to 750 mL. What is the molarity of this solution?…
A:
Q: Five white, 500-mg uncoated ascorbic acid (AA) tablets with an average weight of 0.6152-g were…
A:
Q: Preparation of HCl solution a. Measure 20.7 ml of concentrated Hcl (37% wt HCI) using the pipet and…
A: Solution - It is mixture of solute and solvent.
Q: alculate the molar concentration of a 20.0% solution (w/w) of KCl that has a specific gravity of…
A: Specific gravity of any subsstance can be calculated by multiplying density of that substance with…
Q: Isotonic saline solution is 0.154 M NaCl(aq). What is the solubility of AgCl (Ksp = 1.8 × 10−10) in…
A: Given data : Concentration of NaCl = 0.154 M Ksp of AgCl = 1.8 × 10-10
Q: 1-3: A stock solution of 70% v/v ethanol was diluted with distilled water to prepare 1L of 21% v/v…
A:
Q: In what proportion may the Manufacturing pharmacist mix 20%, 15%, 5% and 3% ZnO to produce 10% ZnO…
A: In pharmaceutical composition preparation, First, we have to pair an ointment of weaker strength…
Q: Balloon Volume of 1M Mass of lacetic acid colour NaHCOз (9) CH3COOH (mL) Pink 25.0 |4.20 Which…
A:
Q: Post-lab Question #7-2: Calculate the molalities (m) of some commercial reagents from the following…
A: Formula used:
Q: What is the function of 1% starch solution in the wijs method? a. Catalyst b. Indicator c.…
A:
Q: A solution was prepared by dissolving 3.50 g of KCI MgCl₂ 6H₂0 (277.85 g/mol) in sufficient water to…
A:
Q: 19. What is the molarity of a commercial phosphoric acid solution, H3PO4 (MW= 98.0 g/mol) that is…
A:
Q: 5- A chemist is given a nonelectrolyte white powder for analysis. 38.7 g of this substance is…
A: Given, The freezing point of the solution containing nonvolatile solute is: -4oC. Freezing point of…
Q: 1-3: A stock solution of 70% v/v ethanol was diluted with distilled water to prepare 1L of 21% v/v…
A: Since you have posted multiple questions, we will answer the very first question. kindly repost the…
Q: Determine the volume of glacial acetic acid to prepare 500 ml of a 0.020 M acetic acid solution…
A:
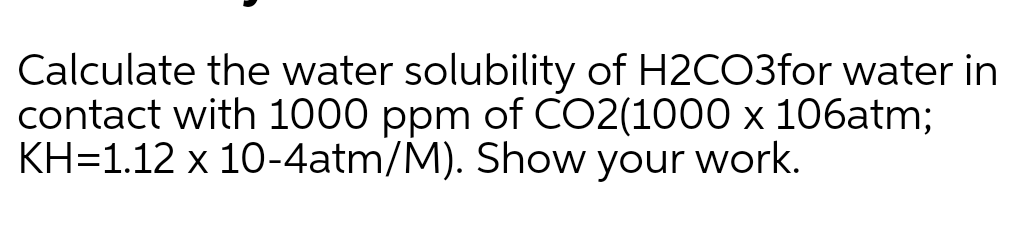
Step by step
Solved in 3 steps

- An unknown 36% v/v monohydroxide reagent has a density of 1.8 g/mL. Zeus wanted to determine the identity of this base, so he transferred a 90.916-mL aliquot of the base to a 1.0 L volumetric flask and diluted it up to mark. He then titrated the base solution and found out that its concentration is 1.0 M.a. What is the molarity of the base solution after dilution? b. If this 1.00M solution is used in the analysis of PLA (Polylactic Acid) plastics for lactic acid (H2Lactate) titration, what is its concentration in titer mg LA/mL titrant? (Lactic Acid: 90.08 g/mol)c. What is the molarity of the unknown monohydroxide reagent?d. What is the molar mass of the unknown monohydroxide reagent?(Density of H2O = 0.99707 g/mL)Hint: Mass of solution = mass of solvent + mass of solutee. What is the identity of the unknown monohydroxide reagent?What is the mass concentration (in g L–1 to two decimal places) of copper(II) hydroxide in a saturated aqueous solution (at 25 °C)? Ksp = 2.2 x 10^-20Calculate the solubility of solid Ca3(PO4)2 (Ksp=1.3 x 10^-32) in a .20 M Na3PO4 solution.
- 6. Give the molar concentration of hydrochloric acid with a specific gravity of 1.18 and has 37.0% (w/w) purity. MM=36.5 g/nWhat is the solubility of SrF2 in g/L if its Ksp is 1.5 x 10-9 ? (MM = 125.6 g/mol) 0.019 0.091 0.0091 0.0019How many mg of Ag2SO4 dissolve in 0.3 M, 500 mL of Al2 (SO4) 3 solution? Compare this solubility with the solubility in purified water and 0.1 M Al2 (SO4) 3 solution. (Ksp = 1.10-5)
- if 361.3 mg of Li2(SO4) (FW=109.95) is added to 450 mL of 0.828 M NH4F, what is the Qip for lithium fluoride. (Ksp of lithium fluoride= 3.8 x10^-3)How to prepare these solutions? 250.0 mL 0.125 M stock Na2S2O3 solution from Na2S2O3·5H2O crystals 250 mL 0.10 M NaOHNOTE: Use the 1.0 M NaOH prepared250.0 mL standard 2500 ppm Cu(II) stock solutiona. Weigh and dissolve appropriate amount of Cu(NO3)2·5H2O crystals in enough distilled water.84 mL of 0.060 M NaF is mixed with 28 mL of 0.15 M Sr(NO3)2. Calculate the concentration of Sr2+ in the final solution. Assume volumes can be added. (Ksp for SrF2 = 2.0 × 10-10) A. 0.038 M B. 0.00012 M C. 0.015 M D. 0.045 M E. 0.075 M



