Q: At 25 °C, the vapor pressures of pure benzene (C6H6) and pure toluene (C7H8) are 95.1 and 28.4 mmHg,…
A:
Q: Are the following stabilised by resonance? A) C2H4 B) CHFO
A: Resonance structure are combination of Lewis structures that describe the delocalisation of…
Q: What is the pH of a 0.103 M aqueous solution of potassium hypochlorite, KCIO? This solution is pH =
A:
Q: Curved arrows are used to illustrate the flow of electrons. Using the provided starting and product…
A: While writing mechanism, curved electron pushing arrow to be shown from negatively charged…
Q: Silver nitrate reacts with Al to form aluminum nitrate and silver. The balanced chemical reaction…
A: A limiting reactant is the one which is completely used up in the reaction and which determines the…
Q: The following information is for questions 1 and 2. Consider the titration of 25.0 mL of 0.100 MHCI…
A: Please find your solution below : Titration is a quantitative method of volumetric analysis which is…
Q: The final ratio of the CH3COOH and CH3COOK
A: pKa of acetic acid solution = 6.38The final pH of the buffer solution = 5.79We have to calculate…
Q: 0.350 350 mol KBr L motKBr X 0.400 L 1 X 119 g KBr 1 mol KBr 0.400 1 = 16.7 g KBr What is the mass…
A:
Q: nt pressure, the volume of a gas is directly proportional to the urc of a gas.
A: True statement is 2,4,5
Q: b) Coal-fired power plants emit airborne particulate matter to the atmosphere known as fly ash.…
A: Fly Ash: When pulverised coal is burned, a finely divided residue known as fly ash is produced,…
Q: Calculate △Go for the following reaction N2O3(g) --> NO(g) + NO2(g)
A:
Q: 5. Two students attempt to etch their initials on a gold plate using hydrochloric acid.
A: I am supposed to do only first question. If you want the solution of 2nd please post it again. The…
Q: 3) A student places 120.0 mL of liquid water at 23.4 °C into a calorimeter and then dissolves 12.5 g…
A: It is an endothermic reaction. Detail solution is given in step 2.
Q: When 3.723 grams of copper are reacted with oxygen, the mass of the copper oxide that is formed is…
A:
Q: 8. A sample of nitrogen gas at 25.°C has a pressure of 740. torr and a volume of 0.400 L. What is…
A: Dear student, since you have posted multiple questions, we will solve only first question for you if…
Q: What quantity in moles of precipitate are formed when 35.0 mL of 0.350 M CaCl₂ is mixed with excess…
A:
Q: Consider these reactions: Reaction 1: H₂(g) + Cl₂ (g) → AH = AH-184.6 kJ Reaction 2: 20F2 (g) →…
A:
Q: C 다 12/pd-C DỊCH, L 2) H3O+ ?
A:
Q: A compound Y (150.0 g/mol) in a 1.00 cm quartz cell has an extinction coefficient (ε) of 23,150 at…
A: Answer = 509.12 Complete solution is given in next step.
Q: N
A:
Q: What is needed to understand Homotopic, Enantiotopic, Diasterotopic?
A: Here we have to predict the stereochemical realtionship between the two indicated protons in the…
Q: 3. In the following synthetic sequence, identify the compounds A, B, and C abla Ph Ph -OCH, Jo.…
A: Here we have to predict the major products formed in the following sequence of reactions. In strp-1…
Q: technetium-99 has a half-life of 6.0 hours. How many gramsin a 24.0 gram sample will remain…
A:
Q: Balance the following reaction (remember to show '1' when necessary) Li₂O + Al(OH)3 →→ •LIOH+A1203…
A:
Q: Which molecules have an octahedral molecular structure A) SbF6 - B)HCl C) BrF5 D) NaCl E) SnCl6 2-
A:
Q: At a pressure of 1 atm, a solution of sucrose (C12H22O11) in water has a boiling point of 101 °C. If…
A:
Q: What mass of precipitate (in g) is formed when 75.0 mL of 0.500 M AIBr; reacts with excess AgNO3 in…
A:
Q: what is a dimer in chemistry? and how are dimers useful in the pharmaceutical industry
A:
Q: VI. VII. During the reaction of benzene with n-propyl chloride and aluminum chloride as catalyst,…
A: If benzene is monosubstituted, the electrophilic substitution takes place depends on the nature of…
Q: V. Give the structures of the products expected from the reaction of 3-methyl-1-pentyne with each of…
A:
Q: A- B- P₁ 0.4atm 760 torr 0.6atm the following table: V₁ 2L 500mL 1500 mL T₁ 300K 150K C P₂ 2atm B…
A: I am supposed to solve only one question. But i am solving 2. Should repost the other questions to…
Q: A balloon contains 0.577 moles of helium gas at STP. What is the volume of the balloon in L?
A:
Q: How many mL of 0.200 M NaOH or 0.200 M HCl must be added to a 100.0 mL buffer solution containing…
A: Please find the attachment.
Q: . what is the volume of the ammonia, in mL needed to prepare the buffer solution? 2. whaw is the…
A:
Q: At a certain temperature the rate of this reaction is first order in NH4OH with a rate constant of…
A:
Q: Plot average pH vs. ([conj. base]/[conj. acid]) AND plot a second graph: average pH vs. log([conj.…
A:
Q: Which of the following would be expected to form hydrogen bonds with water? cyclopentane H H…
A:
Q: Which of the following contains a set of incorrectly paired words? * Chelatometry - Complexation /…
A: Here we have to choose the incorrectly paired words and Lewis base in a complexation reaction-
Q: A solution is prepared at 25 °C that is initially 0.094M in acetic acid (HCH3CO₂), a weak acid with…
A: According to Handerson- Hasselabch equation we have: pH = pKa + logSaltAcid From the given question…
Q: the molality of the following compounds is the same, which compound will have the highest boiling…
A:
Q: Write the balanced equation for the following situation. List the reaction type. Tell the amounts of…
A: A single displacement reaction is a chemical reaction in which one element is replaced by another in…
Q: 3. Do the following synthesis: صل حملام 000 005 مسلسل (a) (d) OH ? Ph (b) (e) OH OH OH
A: Please find the attachment.
Q: my 3800 3600 3400 3200 3000 2800 2600 2400 2200 2000 1800 1600 1400 1200 1000 800 Wavenumbers LOO…
A: Solution:
Q: 2. Starting from 1-propyne as your sole carbon source for the product, provide the steps required…
A: Here we have synthesize the following given compound from starting material 1-propyne by multistep…
Q: 1. Determine the Reaction Order for HCl using calculations described in the background section. Show…
A: Dear student I can do only first three parts according to bartleby guidelines. Please post other…
Q: At what pH does EDTA become a strong complexing agent? * 04 06 8 10 Which best describes the…
A: Ethylenediaminetetraacetic acid (EDTA) is a metal-chelating agent and binds metals such as chromium,…
Q: The reaction of nitric oxide with hydrogen is shown below: 2NO(g) + 2H₂(g) → N₂(g) + 2H₂O(g) The…
A:
Q: (b) A 1.20 g of antimony (Sb) reacts with 2.40 g of iodine (1₂) to form antimony(III) iodide (Sbl3)…
A: The limiting reagent is Iodine
Q: Which of the following would be expected to form hydrogen bonds with water? (Select all that apply.)…
A: The compounds that have slight negative charge on oxygen or nitrogen will form hydrogen bond with…
Q: НО. OH +ATP HO. -PP₁ R- 10x== -CO₂ O-AMP R R TR- - AMP O-AMP O-AMP
A:
Can you explain it and show the work.
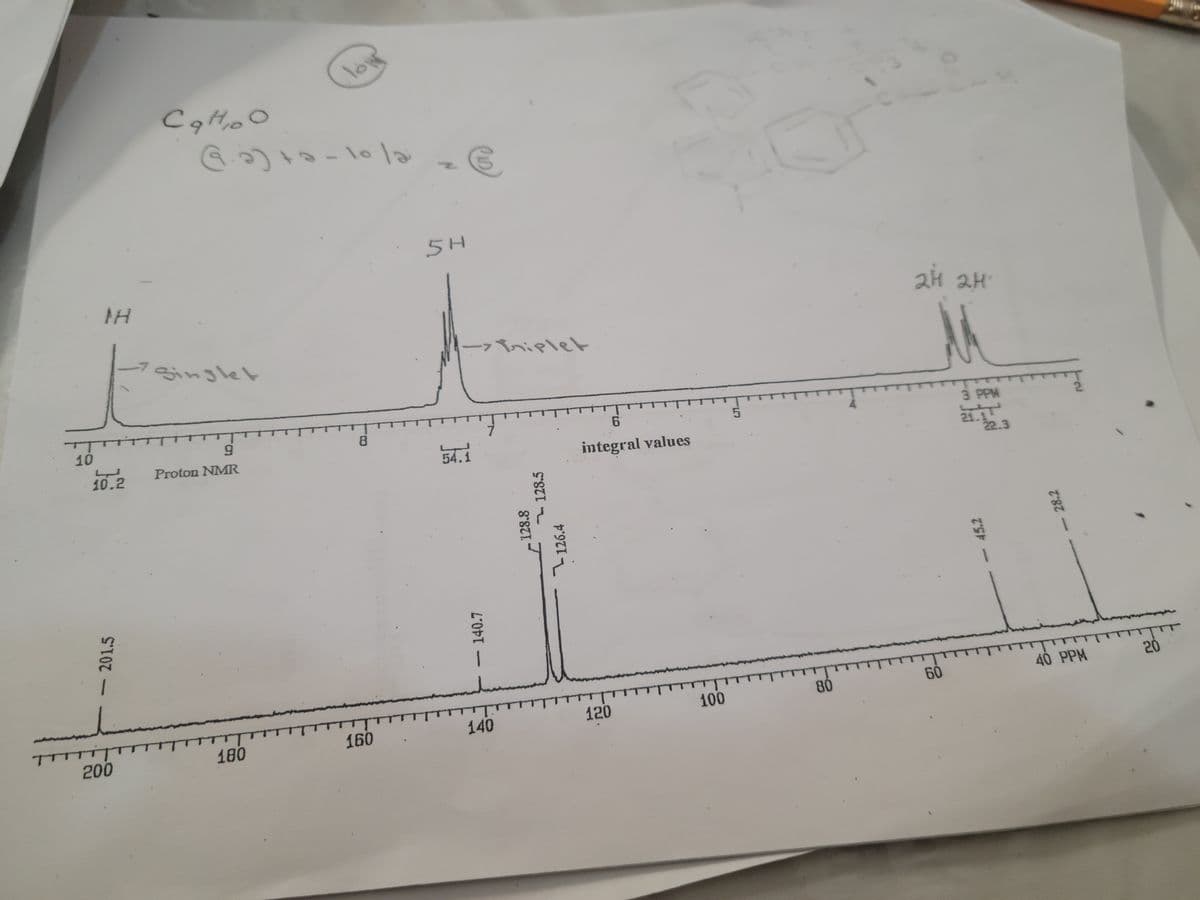
Step by step
Solved in 2 steps with 2 images

- 9. Calculate the Go in kJ/mol. 3Fe2+ (s) + 2Cr(aq) ⟶2Cr3+(aq) + 3Fe (s); Eocell=0.30 V Group of answer choices -170 +170 +87 +58 -195 10. Calculate Ecell in volts. Zn(s)|Zn2+(2.50x10-4M)||Sn2+(1.50M)|Sn(s) Eocell=+0.624 V Group of answer choices 0.736 0.635 0.512 0.848 no correct answerHow to convert 4.76pm to um? Showing the work. 4.76pm x___________m x_____________ um =________________um How to convert 25.0mL to cL? Showing the work. 25.0mL x______________ x ___________ = ______________cLUsing the Mg-Pb phase diagram below, if you start at point A andcool down very slowly to room temperature (point D), sketch the microstructure associated with each letter, determine what phases are present at each letter, determine the compositions for each phase, and determine the weight in each phase. The Mg-Pb mixture has 20 wt. % Pb in it. You start with 100 kg. A. 700ºC B. 600ºC C. 400ºC D. 25ºC
- A monolayer of CO molecules is adsorbed on the surface of 1.00 g of an Fe/AI2O3 catalyst at 77 K, the boiling point of liquid nitrogen. Upon warming, the carbon monoxide occupies 4.25 cm3 at O oC and 1.00 bar. What is the surface area of the catalyst?Determine the enthalpy of solution of MgCl2(s) in units of kJ/mol. The link provides the data needed. https://learn-us-east-1-prod-fleet01-xythos.s3.amazonaws.com/5df2d4e43336c/1279242?response-cache-control=private%2C%20max-age%3D21600&response-content-disposition=inline%3B%20filename%2A%3DUTF-8%27%27Hydration%2520enthalpies-%2520common%2520ions.png&response-content-type=image%2Fpng&X-Amz-Algorithm=AWS4-HMAC-SHA256&X-Amz-Date=20200916T180000Z&X-Amz-SignedHeaders=host&X-Amz-Expires=21600&X-Amz-Credential=AKIAZH6WM4PL5SJBSTP6%2F20200916%2Fus-east-1%2Fs3%2Faws4_request&X-Amz-Signature=a46787b52dc1cef15163e4c4ff519a111d03cbbb18f328c8c98afece27c86745Given below (c): Standard gibb's free energies (∆Gf0 kJ mol-1 ): UO2 = -962.7 UO22+ = -953.5 U4+ = -579.1 Fe2+ = -78.9 Fe(OH)3 ferrihydrite = -692.07 Mn2+ = -288.1 MnO2 pyrolusite = -465.1 HS- = 12.1 H+ = 0 H2O = -237.1 S0 = 0 Given: U(VI) as uraninite; UO2 (where Mn2+ = reductant; MnO2 pyrolusite = product): ∆ Gr0 = -21.3 KJ/mol E0 (emf) = 0.110 V n = 2 F = 96.42 QUESTION: Calculate Eh equation below to calculate at different pH: – Eh = E0 + (RT/nF) * lnK For U(VI) as uraninite; UO2 (where HS- = reductant; S0 = product): UO22+ + Hs- ---- > UO2 + S + H+ [A] pH 3 [B] pH 7
- What is the retardation factor of a pulse of dissolved benzene move through organic-rich wetland soil (organic carbon fraction of soil, foc=50%) if the effective porosity is 0.4 and the bulk density of the soil is 1.5 g/cm3. Follow these important hints: - Determine organic carbon partition coefficient (Koc) - Use this Equation: K oc = K d / f oc to calculate Kd. Select the range which includes your calculated answer. Group of answer choices 300 to 400 2.0 to 2.5 mg/l 2.0 to 2.5 2.6 to 4.8 0.5 to 1.0 2.6 to 4.8 mg/l None of these ranges include my answer.Substances A and B with capacity factors 6.3 and 11.4, respectively, are being carried by a mobile phaseflowing at a velocity of 0.53 mm/s. What is the length of a column required to separate these two substanceswith a resolution of 1? Assume that the sample zones for both A and B broaden based on the van Deemter equation H = 0.35 + 0.48/u+ 0.57u where Hand u are expressed in µm and mm/s, respectively.5 0.0030 0.0015 0.0005 6 0.0030 0.0057 0.0089 7 0.0086 0.0057 0.0089 Show the Calculations of Qc and Kc for Exp. 5-7 Please help me out. Thanks
- 10.11/ A 1.5-kg specimen of a 90 wt% Pb-10 wt% Sn alloy is heated to 250°C (480°F), at which temperature it is entirely an a-phase solid solution . The alloy is to be melted to the extent that 50% of the speci- men is liquid, the remainder being the a phase. This may be accomplished either by heating the alloy or changing its composi- tion while holding the temperature con- stant. (a) To what temperature must the speci- men be heated? (b) How much tin must be added to the 1.5-kg specimen at 250°C to achieve this state? 10.15) A magnesium-lead alloy of mass 5.5 ka consists of a solid a phase that has a compo- sition that is just slightly below the solubility limit at 200°C (390°F). (a) What mass of lead is in the alloy? (b) If the alloy is heated to 350°C (660°F). how much more lead may be dissolved in the a phase without exceeding the solubility limit of this phase? 10.27 A hypothetical A-B alloy of composition 55 wt% B-45 wt% A at some temperature is found to consist of mass…What is ΔSsys for a condensation phase transition at 26.5 °C for a compound that condenses at 26.5 °C and the ΔHsys = -40.1 kJ mol-1 for this process? Express your answer in J mol-1 K-1 to at least two significant figures.Solid Na2S is slowly added to a solution of 1.0 x 10-3 M Cr3+ (with no change in volume). At what [S2-] will Cr2S3 start to ppt? For Cr2S3, Ksp = 1.0 x 10-20

