Using the Mg-Pb phase diagram below, if you start at point A and cool down very slowly to room temperature (point D), sketch the microstructure associated with each letter, determine
Q: Iron(II) chloride (melting point 677 °C) and potassium chloride (melting point 776 °C) form the…
A:
Q: A mixture of phenol and water, under certain conditions of temperature and composition, forms two…
A:
Q: 10 50 -Hot 30 20 0.2 0.8 MOLE FRACTI IEmiPER ÀTURE (oc)
A: The mole fraction represents the number of molecules of a particular component in a mixture divided…
Q: Based on the ternary phase diagram, a binary mixture of furfural and water will always split into…
A: To answer whether the binary mixture of furfural and water always split into two liquid phases:
Q: 4.0g of potassium hydrogen tartrate was added to 300mL distilled water. The temperature of the…
A: We are given initial and final pipette reading for 1.0 M NaOH and we have to find volume and moles…
Q: Sodium carbonate is recovered as decahydrate from 1000kg of 5% solution of sodium carbonate in…
A: 1.
Q: boiling point boiling point BPtof pure A vapour composition liquid composition Bpt of pure B O A 1.0…
A: Given data :-
Q: 200 mg of granular salt is briefly stirred into a glass container of 1/2 liter volume filled with…
A:
Q: boiling point boiling point BPtof pure A vapour composition liquid composition Bpt of pure B OA 1.0B…
A:
Q: The distribution coefficient, KD (C water/C ether), between ether and water for aspirin at room…
A:
Q: For the purpose of determination of agricultural pesticides in river water, 3.2 liters of river…
A: Distribution coefficient = Kd = Concentration of solute in organic phase / Concentration of solute…
Q: b) This sample is heated to T = 110 °C. Give the mass of the phases present at this temperature and…
A: In an ideal mixture of two liquids, both the liquids have different in boiling point. Therefore,…
Q: In an experimental study of membrane-like assemblies of synthetic materials, a phase diagram like…
A: First, consider At point a, the mixture is in liquid form. Suppose the liquid mixture reaches the…
Q: What mass of potable water (in kg) is extracted from 1000. kg of a 4.00% by mass solution of NaCl at…
A:
Q: The table below shows temperature/composition data collected for a mixture of methylbenzene (M) and…
A: Given: Boiling point of methylbenzene (M) = 110.60C Boiling point of octane (O) = 125.60C
Q: H Redew i Conatant MISSED THIST Watch KCVa2 Read Secnon e3 You can cick on the Review ink t accese…
A:
Q: Referring to the Al-Si Eutectic diagram discussed on the Phase Diagrams-Lever Rule handout,…
A:
Q: TIK |210 Compositon of one phase P.1 Vapour 292 290 P+2 Composton fsecond phase P2 Liquid 273 P.1…
A:
Q: 1) 1500 kg mixture contains 15% acetone, 75% water and 10% Acetic acid by weight. a) Calculate the…
A: Average molecular weight of a mixture of substances can be calculated as the total mass of all the…
Q: * Module 3: In his experiment, Mashiho wanted to determine the molar mass of his unknown sample.…
A:
Q: 4.0g of potassium hydrogen tartrate was added to 300mL distilled water. The temperature of the…
A: We are given initial and final pipette reading for 1.0 M NaOH and we have to find volume and moles…
Q: The process by which the supernatant liquid is poured out leaving the precipitate in the container…
A: Given process The process by which supernatant. Liquid is poured leaving the precipitate in the…
Q: A solution of common salt in water is prepared by adding 50kg of salt to 200kg of water to make a…
A: Weight fraction of a component is defined as the amount of component (salt) present in solution.
Q: A liquid mixture contains ethanol (C₂H5OH) with a volume fraction of 0.815 and the balance water…
A: According to the question, a liquid mixture contains 0.815 volume fraction ethanol C2H5OH and the…
Q: A liquid that is almost entirely water is reported to contain 125 ppb phenol on a mass basis. a)…
A:
Q: How to find final volume? what is the osmolarity and tonicity (hyper,iso,hypo) Tonicity Worksheet…
A:
Q: There are two separate phases, one with a composition of 12% phenol and the other with a composition…
A: Given : Total mass of mixture = 210 g Composition of phenol in mixture = 35 % Composition of phenol…
Q: Basis for recrystallization Nernst Distribution Law differences in solubilities of a solid and an…
A:
Q: Q3/The solubility of sodium chloride NaCl in water at 290 K is 35.8 kg / 100 kg of water. Express…
A: Given that: solubility of NaCl = 35.8 Kg/100 Kg Molecular weight of water = 18.016 g/mol Molecular…
Q: the following are wrong, give your correct answer: 1. Cloud point value of lubricating oil fraction…
A: According to Q&A guidelines of Bartleby, I am allowed to answer only the first question out of…
Q: Tetracycline produced in Streptomyces aureus fermentations is purified by crystallisation. One…
A: Tetracyclines are oral antibiotics originally derived from stremptomyces aureofaciens .Their method…
Q: Water boils at 100 oC at 1 atm (101325 Pa). Tom dissolve 574.4 g of sugar (MW = 342.3 g/mol) in…
A: Raoult's law: According to this law pressure of solution is equal to the product of mole fraction of…
Q: Given the following data, prove that the distribution of benzoic acid in water and benzene follow…
A: The total number of moles/mass of benzoic acid is not given to calculate the extracted number of…
Q: The solvent for an organic reaction is prepared by mixing 70.0 mL of acetone (C3H6OC3H6O) with 56.0…
A: Vapor pressure of the mixture is calculated using the Roulte's law, solvent's partial vapor pressure…
Q: Consider the remperagture composition diagram showe bolow for bexane and heptane For he mituRe of…
A:
Q: Two mixtures of methanol (CH3OH) and water (H2O) are contained in separate flasks. In the first…
A: Solution -
Q: answer the following questions? 1-Paraffins are obtained by process from petroleum. | Filtration |…
A: here you uploaded 10 questions but we are solving the first three questions only if you want the…
Q: The phase diagram for the nearly ideal mixture of benzene and toluene is shown below. Consider a…
A: Given that , The mole fraction of toluene=0.6 The phase diagram of Benzene and Toluene mixture is…
Q: A liquefied mixture of n-butane, n-pentane, and n-hexane has the following composi- tion in percent:…
A: The fraction of a particular entity in a mixture containing that entity amongst other chemical…
Q: 6. Using the figure below, what is the composition of the vapor from a solution containing 20 grams…
A: Given the mass of toluene = 20 g Molar mass of toluene = 92.14 g/mol => moles of toluene = 20 g *…
Q: There are two separate phases, one with a composition of 12% phenol and the other with a composition…
A: Given : Total mass of mixture = 210 g Composition of phenol in mixture = 35 % Composition of phenol…
Q: 10. if a 75%hexane /25% ethyl acetate mixture is used as a solvent system and the sample of interest…
A: In a 75% hexane/25% ethyl acetate mixture, the concentration of non-polar solvent is high and the…
Q: 1) 1500 kg mixture contains 15% acetone, 75% water and 10% Acetic acid by weight. a) Calculate the…
A:
Q: Using the phase diagram for mixtures of cyclohexane and toluene, estimate: A) Boiling point of pure…
A: In any particular boiling point diagram, there are two curves. First curve (upper curve) is called…
Q: 3. In the liquid-liquid extraction, there are two funnels with solutions of total volume 250 mL and…
A: It is given that, for every 1 part of aqueous solution, 3 parts of ethyl acetate is required for…
Q: Q3 / The solubility of sodium chloride NaCl in water at 290 Kis 35.8 kg / 100 kg of water. Express…
A: Molar mass of NaCl is 58.45 g/mol It is not 135.8
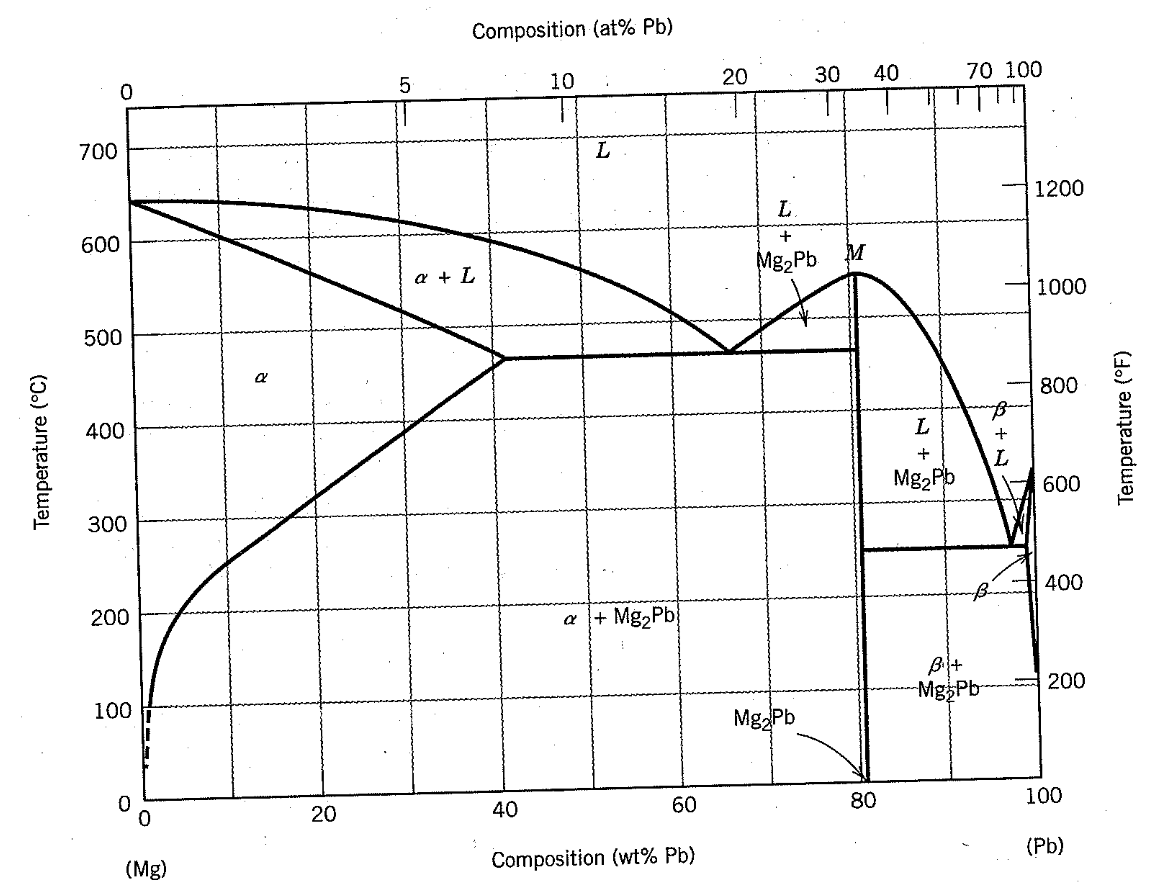
Using the Mg-Pb phase diagram below, if you start at point A and
cool down very slowly to room temperature (point D), sketch the microstructure associated with each letter, determine what phases are present at each letter, determine the compositions for each phase, and determine the weight in each phase. The Mg-Pb mixture has 20 wt. % Pb in it. You start with 100 kg.
A. 700ºC
B. 600ºC
C. 400ºC
D. 25ºC
Step by step
Solved in 2 steps

- What is the molarity of a concentrated solution of sulfuric acid with a specific gravity of 1.84 and 95.0% by weight sulfuric acid? specific gravity = density of substance at certain temp/density of water at 4 degrees C the density of water is 0.999973 g/mL at 4 degrees celsius. Describe how to prepare 3.0 L of 0.100M. I will pipet ______ and add this to 200mL water. The solution will be stirred and diluted to 3L with water.Ethyl alcohol (C2H5OH) may be prepared by the fermentation of glucose (C6H12O6) as indicated by the equation: yeastC6H12O6 ----> C2H5OH + CO2 74.12 mL of ethyl alcohol (specific gravity = 0.790) was collected by this fermentation pro- cess. What mass of glucose was used?Ethyl alcohol (C2H5OH) may be prepared by the fermentation of glucose (C6H12O6) as indicated by the equation: yeastC6H12O6 ----> C2H5OH + CO2 74.12 mL of ethyl alcohol (specific gravity = 0.790) was collected by this fermentation pro- cess. What mass of glucose was used? SET-UP: Answer:
- Stock iron(II) solution (200Ug mL-1 Fe) ferrous ammonium sulfate hexahydrate mass= 0.1437g, transfer it to a 100 ml beaker. add 15 ml approx of water and 15m1 'approx of dilute sulphuric acid (2M H2SO.). then transfer FeII to 100 ml flask makeup to the mark with water. calculate the moles of ferrous ammonium sulfate hexahydrate solution in unit ug/mL.A mixture of ethanol (ethyl alcohol) and water contains 40.0% water by mass.(a) Assuming volume additivity of the components, estimate the specific gravity of themixture at 20°C. What volume (in liters) of this mixture is required to provide 150 mol ofethanol?(b) Repeat Part (a) with the additional information that the specific gravity of the mixture at20°C is 0.89045 (making it unnecessary to assume volume additivity). What percentage errorresults from the volume-additivity assumption?125.1 mg of streptomycin sulphate are dissolved in 10 ml of water. A GC headspace analysis is carried out in order to determine the methanol content of the drug. A peak for methanol is produced which has 73.2% of the area of a peak for a methanol standard containing 0.532 mg/100 ml of methanol in water analysed under exactly the same conditions, What is the methanol content of the streptomycin sulphate in ppm and %w/w? Answer: 311.3 ppm, 0.3113 % w/w.how??
- The gravimetric factor used to express CoCBr6·H20 in a sample that is finally weighed as PbClBr is choose below: FW PbClBr / 6 x FW CoCBr6·H20 FW CoCBr6·H20 / FW PbClBr FW CoCBr6·H20 / 6 x FW PbClBr 6 x FW PbClBr / FW CoCBr6·H20Calculate the Constant Weight (in grams) of the Empty Crucibles. Show your solution. Crucible No. 1 2 3 Weighing 1 22.6035 22.0223 24.1535 Weighing 2 22.6017 22.0204 24.1533 Weighing 3 22.5994 22.0199 - Weighing 4 22.5992 - - Constant Weight, g ??? ??? ???Using the percent purity calculations, determine the percent yield of synthesis of aspirin. Part I Synthesis of Aspirin Mass of salicylic acid used (g) 2.029g Volume of acetic anhydride used (mL) 5ml Mass of acetic anhydride used (vol. × 1.08 g/mL) 5.4g Mass of aspirin synthesized (g) 3.256g Part II Melting Temperature Data Melting temperature (°C) 133°C Part III Salicylic Acid Standard Stock Solution Initial mass of salicylic acid (g) 0.210g Moles of salicylic acid (mol) 0.0147 mol Initial molarity of salicylic acid (M) 0.724 M Part III Beer’s Law Data for Salicylic Acid Standard Solutions Trial Concentration (M) Absorbance Water (mL) 1 10 0.301 0 2 7.5 0.219 2.5 3 5.0 0.163 5.0 4 2.5 0.074 7.5 Best-fit line equation for the salicylic acid standards Test of the Purity of the Synthesized Aspirin Initial mass of aliquot of product (g)…
- Produce Water mass “X” is known to be a mixture of water masses “1”, “2”, and “3”. Using the data plotted below, calculate the composition of “X” (i.e. determine the fraction from each of the 3 water masses that makes up water mass X). Watermass#1– SF6 =25 S=33.5 Watermass#2– SF6 =75 S=32 Watermass#3– SF6 =50 S=36.5 Water mass X – SF6 =60 S= 34150 kmol of an aqueous phosphoric acid solution contains S mol percent H3P04. The solution is concentrated by adding pure phosphoric acid at a rate of 20L per minute. Write a differential mole balance on phosphoric acid and provide an initial condition. Solve the balance to abtain an expression vdor np(t) . Use the result to derive an expression far xp(t), the mole fraction of phosphoric acid in the solution. How long will it take to concentrate the solution to IS percent H3P04 ?How would you prepare 75 mL of an H2SO4 solution at 20% in weight (density=1.1394 g/mL, richness=227.9 g/L) starting from the commercial H2SO4 solution (98%, density=1.8361 g/mL, richness=1799 g/L)? Please choose among the most appropriate procedure which better describes the preparation of the previous diluted H2SO4 solution: Select one or more: a. Take the required volume of commercial H2SO4 solution with an adequate pipette, pour it into a graduated flask and add water up to the final volume (75 mL in this case). Homogenize the flask by stirring it gently. b. Take a beaker and put about 40 mL of distilled water. Then take the required volume of commercial H2SO4 solution with an adequate pipette and pour them over the beaker. Let the solution be warm and pour the mixture into a graduated flask and add water up to the final volume (75 mL in this case). Homogenize the flask by stirring it gently. c. Take a beaker and put about 40 mL of distilled water. Then take the required volume…

