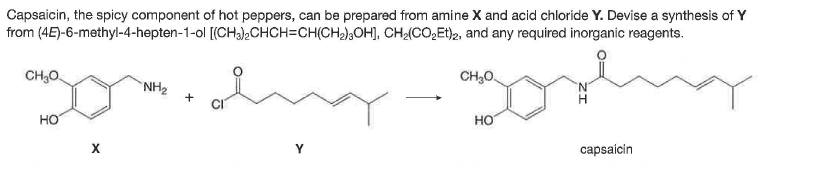
Capsaicin, the spicy component of hot peppers, can be prepared from amine X and acid chloride Y. Devise a synthesis of Y from (4E)-6-methyl-4-hepten-1-ol [(CH3).CHCH=CH(CH2),OH), CH(CO,Et)2, and any required inorganic reagents. CH,O. CH,0. NH2 но но capsaícin
Q: sed on the reaction scheme below, draw the structure for compounds D, E, F (i) LIAIH4 in ether (ii)…
A:
Q: Starting with benzene, which set of reagents A through D will yield the product? Click on a letter A…
A: To get ortho product we should have introduce a ortho directing group here it is acyl group which…
Q: CH3 1. TSCI, EtgN H3C 2. NaOCH3 DMF, 100 °C CH3 OH H2 (1 atm) cat. [Pd]
A:
Q: HC=CH The synthesis above can be performed with some combination of the reagents listed below. Give…
A: For deprotonation of acetylene we need strong base such as NaNH2.
Q: Which alkene that upon reaction with O3 followed by reaction with Zn, H* gives the indicated…
A:
Q: Treatment of but-1-ene with hot KMno4/OH- gives product D, and subsequent acidification of the…
A: Treatment of any alkene with hot KMnO4 in presence of base i.e OH- breaks the double bond and…
Q: Steroids are a class of organic compounds containing a specific tetracyclic skeleton (four rings),…
A: Given : We have to draw second step for the given reaction.
Q: PS#3 part 2- Synthesis: Given this group of simple compounds, devise a plan to synthesize each…
A: We have to snthesize the following given compounds from the given starting materials in multistep…
Q: Draw the structure(s) of the major organic product(s) of the following reaction. 1. NaOCH3 / CH3OH…
A: Here we are required to predict the product of the reaction.
Q: Choose the best reagents from the list provided below for carrying out the following conversion.…
A:
Q: The following reactions occur via SN1, E1, SN2, or E2. material paying attention to regio- and…
A:
Q: .CI 1. excess CEH5MGBR 2. H20
A:
Q: A mixture of 0.10 mol benzene and 0.10 mol p-xylene was allowed to react with 0.10 mol nitronium ion…
A: Given: A mixture of 0.10 mol benzene and 0.10 mol p-xylene was allowed to react with 0.10 mol…
Q: 1,4-Dioxane The transformation above can be performed with some combination of the reagents listed…
A: Given reaction,
Q: the compound can be synthesized by an SN2 reaction. Suggest a combination of haloalkane and…
A: The given compound can be prepared by the reaction of Benzyl Iodide (haloalkane) and Sodium benzoate…
Q: Draw the structure(s) of the major organic product(s) of the following reaction. 1. NaOCH3 / CH,OH…
A: The hydrogen present in the alpha carbon of the carbonyl group is acidic due to the resonance…
Q: the compound can be synthesized by an SN2 reaction. Suggest a combination of haloalkane and…
A: Given:
Q: 57. Which structure is expected to emerge as the product of the reaction between the given alkyl…
A: For an E2 elimination the stereo electronic requirement is the leaving group and the -H atom must be…
Q: OH 1. Hg(OAc)2, H2O 2. NABH4 CH3 .CH3 H2C° H3C d-catalyzed addition of water to an alkene yields an…
A:
Q: CH2l2 ? Zn/Cu The starting material necessary for this transformation is: A в D E
A: This reaction is known as Simmons Smith reaction. In this reaction alkene is get converted into…
Q: The following reaction schemes have flaws as presented. Which of the following statement is the best…
A: Option D is correct answer.
Q: Wittig Reaction 1. Why did you get the stereoisomer of the product that you did get rather than the…
A: 1. The Z-stereoselective Wittig reaction consists of a stereoselective first step forming the syn…
Q: Triethyloxonium tetrafluoroborate, (CH3CH2)3O+ BF4 −, is a solid with melting point 91–92 °C. Show…
A: For the chemical reaction between triethyl oxonium tetrafluoroborate and Nuc:-, the chemical…
Q: 2) Give the products of the following reactions. Be careful to show stereochemistry if relevant. 1.…
A: SN1 reaction is nucleophilic substitution, that involves nucleophile replacing leaving group. SN1…
Q: *5 carbon isoprene" - what is that? I had to find out more. According to this page in the LibreText,…
A: In this reaction 1st addition of hcl to diene and then substitution of Cl by SH and product.
Q: ОН to CF3CO,H
A:
Q: Predict the organic products of the reaction of 2 butene with each reagent. Be sure to indicate…
A:
Q: 1,2-Trans-cyclohexanediol has been shown to be an efficient support for the asymmetric…
A: PhLi can act as a nucleophile. It can undergo halogen exchange reaction to generate anion.
Q: What is the major organic product of the fallowing reaction? Show the stereochemistry of the final…
A: The major organic product for a given reaction has to be given.
Q: 'ou synthesized 4-penten-2-ol, and to confirm the identity of your produ ou are asked by your…
A:
Q: 18. AICI3 19. ОСНЗ 1. Br2, FeBr3 2. Ni, 100 atm H2, 150°C 20. +
A:
Q: The synthesis of carbohydrates can be particularly difficult because of the large number of chiral…
A: The Gilman reagent reacts with the epoxide, which is followed by the acid gives alcohols. Here, the…
Q: A student wanted to synthesize methyl1-butyl ether, CH3-0-C(CH)3. He attempted the synthesis by…
A:
Q: 2. Provide structure(s) for the product(s) of reaction of hexanal with each of the following…
A: According to guidelines we are supposed to answers only 3 subparts
Q: (vi) The final step in your reaction sequence is alkylation with benzylbromide. Draw the structure…
A: Given reaction: CC
Q: Give the major organic product of the following reaction, include stereochemistry as appropriate 1.…
A: Given incomplete reaction is : Give the major organic product of the following reaction, include…
Q: 4.35 Formulate the reaction of cyclohexene with (i) Br2 and (ii) meta-chloro- peroxybenzoic acid…
A: the reaction of cyclohexene with Br2 is followed by anti addition to give trans product having a…
Q: 2-Give the structures, including stereochemistry of compounds A and B in the following sequence of…
A:
Q: When anisole (methoxybenzene) is chlorinated the products are 65% 4-chloro anisole and 34.9% 2-…
A: D is the correct reason to explain this.
Q: NaBH4, EtOH + ОН H H ОН exo is MAJOR product. endo is MINOR product.
A:
Q: CH, -CH=CCH=CHCH3 The Wittig reaction can be used for the synthesis of conjugated dienes. Propose a…
A: The wittig reagent and the aldehydes used to synthesize the double bond denoted b is given below
Q: the compound can be synthesized by an SN2 reaction. Suggest a combination of haloalkane and…
A:
Q: When 3-methyl-1-butene is reacted with 9-borabicyclo[3.3.1]nonane, the "1-ol" product is formed.…
A:
Q: - Provide the structure of the major organic product for the reaction sequence shown below. 1.…
A: It is a 3 step reaction as shown in the following step.
Q: 1) Hg(O2CCH3)2, THF/H2O 2) NaBD4
A:
Q: HBr 40° C 21. Na, CH;OH NH3 22. 1. NBS, heat 2. NaOEt, EtOH 23.
A:
Q: OH 1. Hg(OAc)2, H2O 2. NaBH4 CH3 .CH3 H3C Acid-catalyzed addition of water to an alkene yields an…
A: Given reaction is oxymercuration- demercuration reaction of alkene.
Q: CH3 CH3 H2SO4 CH;CH2Ć-OCH3 CH3 CH;OH CH3CH2Ċ=CH2 Treating 2-methyl-1-butene with methanol in the…
A: The detailed mechanism given b
Q: 1. Provide a major product(s) [ignore stereochemistry] for each reaction: Cl2, MeOH H₂N Acid,…
A:
Step by step
Solved in 2 steps with 2 images

- KMnO4, warm, conc'd reacts with hept-1-ene to yield __________. CO2, hex-1-ene CO2, hexanoic acid Formic acid, pentanoic acid Ethanoic acid, pentanal Formic acid, hexanoneFor butenafine, develop a synthesis that starts with napthalene and benzene. (aromatic methyl groups can be converted to alkyl bromides using Br2/light, and aldehydes by heating the alkyl bromide in DMSO, eg. ArCH3 to ArCH2Br then to ArCHO).i. When But-1-ene (CH3CH2-CH=CH2) is treated with HBr in the presence of a peroxide, give the name and structure of the product. ii. Give the structure of the product from the reaction of propanal with 1M ethanol in dry acid. iii. What happens when Further 1M of ethanol is added to above (ii) ?
- A key step in the synthesis of naproxen, an NSAID more commonly known by its brand name, Aleve (Section 3.9), is a coupling reaction of 2-bromo-6-methoxynaphthalene to form 2-methoxy-6-vinylnaphthalene. Show three different coupling reactions, and the required reagents, that could be used tocarry out this step.d. Reaction with CH3MgI (excess), ether; then H+/H2O e. Reaction with LiAlH4, ether; then H+/H2O f. Reaction with DIBAL (diisobutylaluminum hydride), toluene, low temperature; then H+/H2OTunicates are marine animals that are called "sea squirts" because when they are taken out of water, they tend to contract and expel seawater. Lepadiformine is a cytotoxic agent (toxic to cells) isolated from a marine tunicate. During a recent synthesis of lepadiformine, the investigators observed the formation of an interesting by-product (3) while treating diol 1 with a reagent similar in function to PBr3 (J. Org. Chem. 2012, 77, 3390–3400):
- Triethyloxonium tetrafluoroborate, (CH3CH2)3O+ BF4 −, is a solid with melting point 91–92 °C. Show how this reagent can transfer an ethyl group to a nucleophile (Nuc:−) in an SN2 reaction. What is the leaving group? Why might this reagent be preferred to using an ethyl halide?A mixture of 0.10 mol benzene and 0.10 mol p-xylene was allowed to react with 0.10 mol nitronium ion until all the nitronium ion was gone. Two products were obtained: 0.002 mol of one and 0.098 mol of the other. a. What was the major product? b. Why was more of one product obtained than of the other?Predict the organic products of the reaction of 2 butene with each reagent. Be sure to indicate stereochemistry and regioselectivity where appropriate. a. Br2 in H2O b. Hg(OAc)2, H2O c. Product from (b) + NaBH4
- True or false? 2-bromo-2-methylpropane undergoes SN1 rather than SN2 because the nucleophile experiences steric hindrance, and a stable carbocation can be formed. When hydroxide ion reacts with 1-chloropropane the main product is 1-propanol.Benzene is one of the compounds used as octane enhancers in unleaded gasoline. It is manufactured by thecatalytic conversion of acetylene to benzene: 3C2 H2(g) ⇌ C6 H6(g). Which value of Kc would make this reactionmost useful commercially? Kc ≈ 0.01, Kc ≈ 1, or Kc ≈ 10. Explain your answerExplain the following result. Although alkenes are generally more reactive than alkynes towards electrophiles, the reaction of Cl2 with but-2-yne can be stopped after one equivalent of Cl2 has been added.

