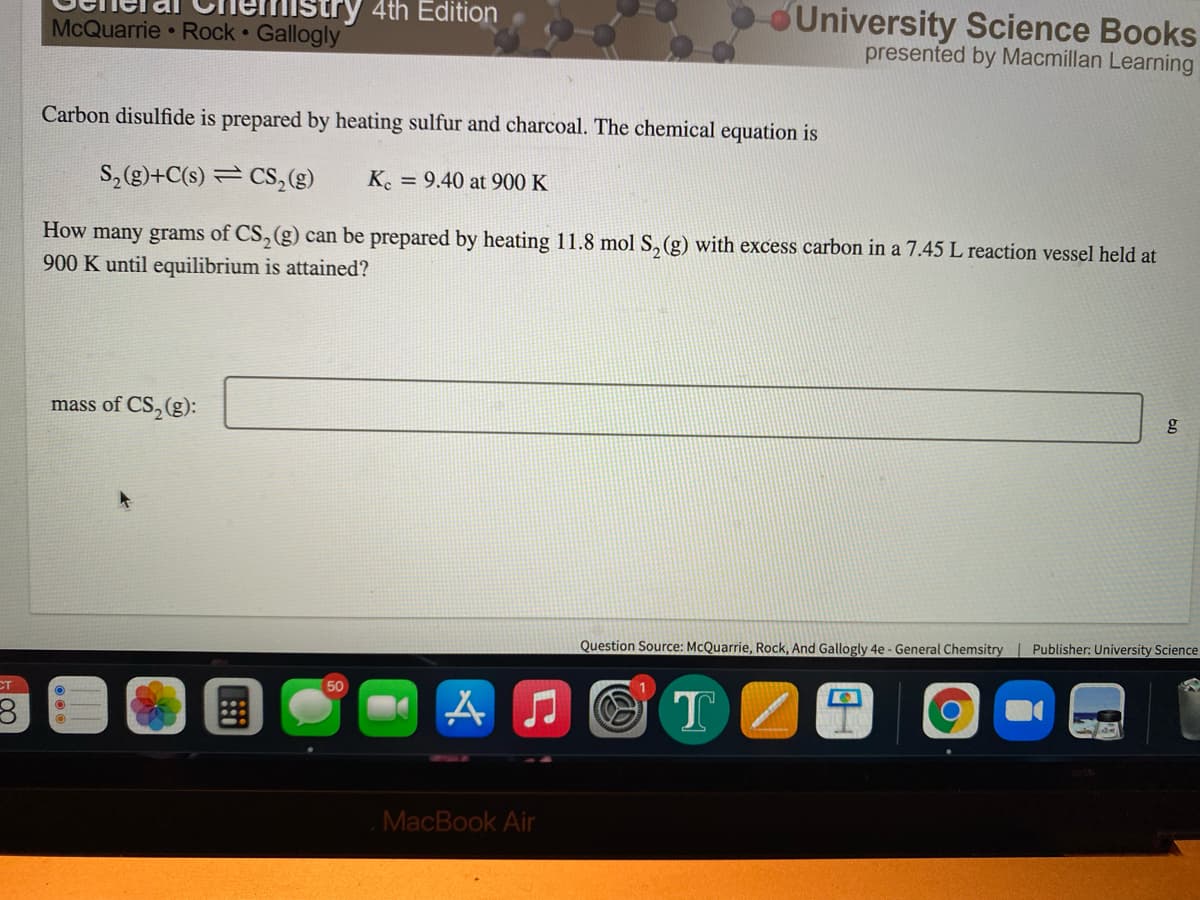
Carbon disulfide is prepared by heating sulfur and charcoal. The chemical equation is S, (g)+C(s) = CS, (g) K. = 9.40 at 900 K How many grams of CS, (g) can be prepared by heating 11.8 mol S,(g) with excess carbon in a 7.45 L reaction vessel held at 900 K until equilibrium is attained? mass of CS,(g):
Carbon disulfide is prepared by heating sulfur and charcoal. The chemical equation is S, (g)+C(s) = CS, (g) K. = 9.40 at 900 K How many grams of CS, (g) can be prepared by heating 11.8 mol S,(g) with excess carbon in a 7.45 L reaction vessel held at 900 K until equilibrium is attained? mass of CS,(g):
Chemistry by OpenStax (2015-05-04)
1st Edition
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Chapter10: Liquids And Solids
Section: Chapter Questions
Problem 59E: Consider a cylinder containing a mixture of liquid carbon dioxide in equilibrium with gaseous carbon...
Related questions
Question
Transcribed Image Text:University Science Books
presented by Macmillan Learning
istry 4th Edition
McQuarrie Rock Gallogly
Carbon disulfide is prepared by heating sulfur and charcoal. The chemical equation is
S, (g)+C(s) = CS, (g)
K. = 9.40 at 900 K
How many grams of CS, (g) can be prepared by heating 11.8 mol S,(g) with excess carbon in a 7.45 L reaction vessel held at
900 K until equilibrium is attained?
mass of CS,(g):
Question Source: McQuarrie, Rock, And Gallogly 4e - General Chemsitry
Publisher: University Science
50
T.
CT
MacBook Air
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning