Organic Chemistry
8th Edition
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Chapter10: Alcohols
Section: Chapter Questions
Problem 10.50P
Related questions
Question
5d.
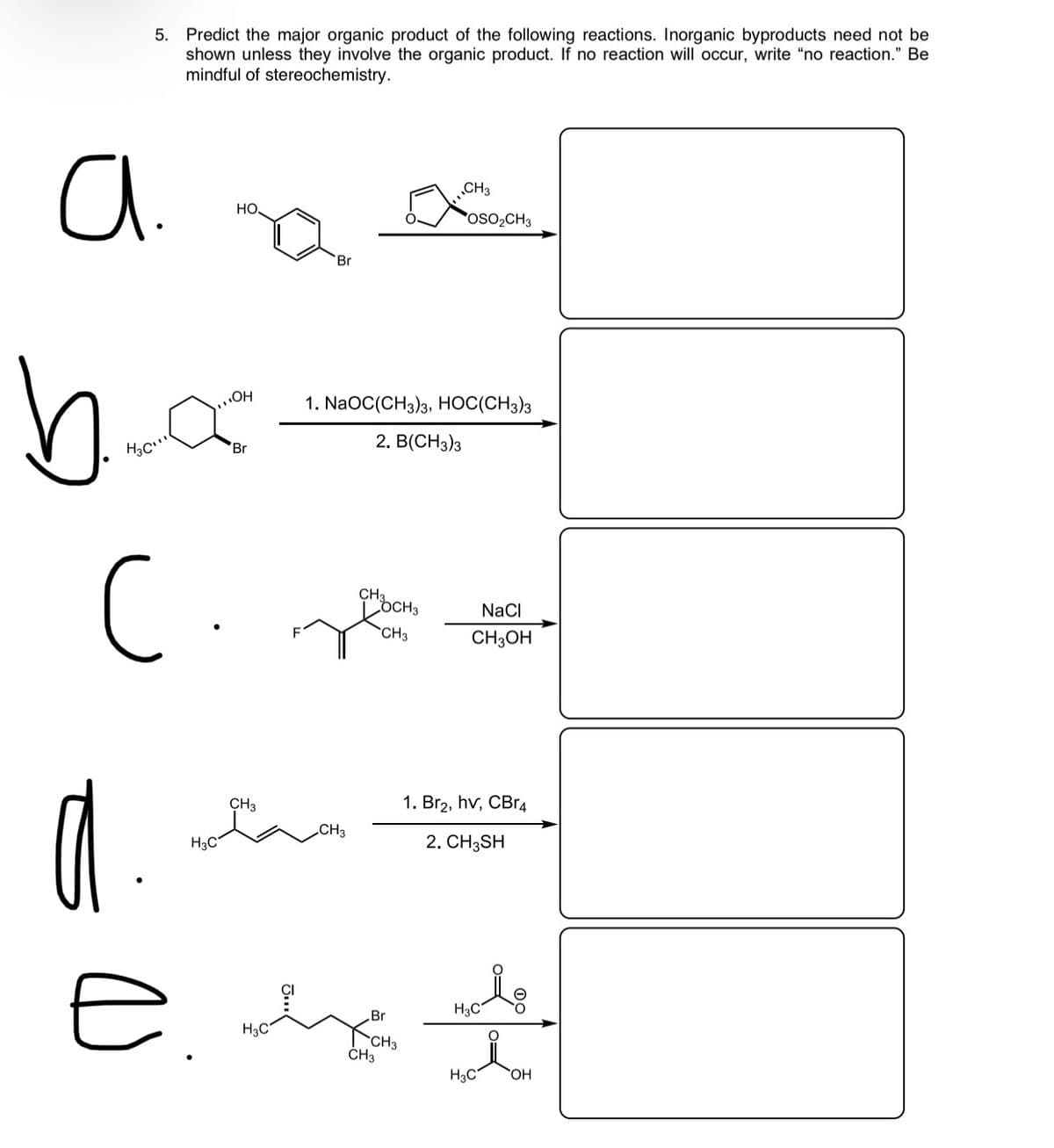
Transcribed Image Text:5. Predict the major organic product of the following reactions. Inorganic byproducts need not be
shown unless they involve the organic product. If no reaction will occur, write "no reaction." Be
mindful of stereochemistry.
a.
CH3
но.
OSO2CH3
Br
1. NaOC(CH3)3, HOC(CH3)3
Br
2. B(CH3)з
C
CH3
OCH3
CH3
NaCI
CH3OH
CH3
1. Br2, hv, CBr4
CH3
H3C
2. CH3SH
H3C
Br
H3C
ČH3
H3C
HO.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning