Q: 2. A chemical reaction proceeds in such a way that after the first second, the amount of a certain c...
A:
Q: 2) The dehydrogenation of benzyl alcohol to benzaldehyde is an equilibrium process described by the ...
A:
Q: ite Neat and Balanced Reactions from Word Equations You need to submit one sheet of paper (not this ...
A:
Q: N(CH3)2 HO,
A: The given compound is a secondary alcohol. Here we have to convert the given compound to the given a...
Q: The airbags that protect people in car crashes are inflated by the extremely rapid decomposition of ...
A: Decomposition reaction is a reaction in which a single reactant decomposes to form two or more produ...
Q: se. 'asam Ca + O2 -> CaO Ca = Ca = 0 = 0 =
A: Balanced the given reaction ---
Q: Please write the electron configuration of the following elements: Copper Sodium Nitrogen
A: Given elements: Copper Sodium Nitrogen The distribution of electrons or electronic configuration of...
Q: H2N- CH-C- NH- CH2-C-NH-CH2- NH-CH-C-NH-CH-C-OH CH2 CH2 CH2 CH Compound A (Leu-enkephalin) H3C CH3 O...
A: Since you have asked multiple questions we will answer the first one for you. To get the remaining q...
Q: The rate constant for a particular zero-order reaction is 0.075 M s1 If the initial concentration of...
A: Detail mathematical calculation to find out the value of time for given zero order reaction is shown...
Q: If a reaction will occur, give the product(s) otherwise write no reaction. Explain.
A: Here, the given reactant react with Br2 in presence of CCl4. We have to find the product.
Q: Calculate the pH of a 7.51 M solution of HBRO. The K, of HBRO is 2.50 x 10. Express your answer usin...
A: Given data : Initial concentration of HBrO = 7.51 M Ka = 2.50 × 10-9
Q: Calculate the pH that results when the following solutions are mixed: 35 mL of 0.20 formic acid, 55 ...
A:
Q: Why do we use a salt bridge during calomel measurement
A:
Q: What do you call the layer that balances out the charge of the layer directly covering the core of t...
A:
Q: What mass of Cu(NO 3) 2 can be prepared from the reaction of 5.00 grams of copper with 5.00 grams of...
A:
Q: Under certain conditions the rate of this reaction is zero order in ammonia with a rate constant of ...
A: Given, 2NH3(g) ➝ N2(g) + 3 H2(g) Reaction is a zero order reaction. Rate constant (k) = 0.0091 M.s-1...
Q: Problem no. 23-25 en the following data for the NH4* + NO2" – N2 + 2H20 reaction Trial Rate [NO2] 0....
A: K is rate constant
Q: You are called in as a consult for a patient who suffers from acidosis. Urine levels of both lactic ...
A: The pateint has a defect of phosphatase enzyme. Phosphatase enzyme has function to active pyruvate d...
Q: Here we have the molecular orbital energy diagram for this chlorophyll molecule. Match the absorptio...
A: Wavelength of absorption is inversely proportional to the energy gap between the two levels.
Q: If you added 5.0 mL of 2.0 M NAOH to 45.0 mL of pure water what would the resulting ApH of the solut...
A: 5.0 mL of 2.0 M of NaOH is added to 45.0 mL of pure water. We have to calculate the resulting ∆p...
Q: Evaluate A S for the reaction below at 25 °C. CH 4(g) + 2CI 2(g) CCI 4 (e) + 2H 2(g) -135.4 -74.81 A...
A: CH4(g) + 2Cl2(g) --> CCl4(l) + 2H2(g) ∆H°f (CH4) = -74.81 KJ/mole ∆H°f(CCl4) = -135.4KJ/mole ∆G°f...
Q: How many moles of C,H,O were dissolved into 91.340 g of ethylene glycol, if the freezing point of th...
A:
Q: Show all pertinent solutions neatly, orderly and completely. 1. NaMnO4 2. So32 3. KIO4 4. Nazs203 5....
A:
Q: EQUATION WRITING: Balance the following redox reactions. Use change in oxidation state method (oxida...
A: Let us step by step solve this:
Q: Estimate the charge of the following oligopeptides at pH 7 based on the pKa values of the constituen...
A:
Q: 1. I have problems understanding the results of this experiment, could you help me by explaining the...
A: In order to obtain the result of the experiment, we have to find the most suitable wavelength to mea...
Q: Which of the following bonds is the most polar bond? A) C –C B) C –O C) C –H D) C –Cl E) C –N Which...
A: Given:- Which of the following bonds is the most polar bond?A) C –C B) C –O C) C –H D) C –Cl E) ...
Q: What is the entropy change of the reaction below at 298 K and 1 atm pressure? 4AI(s) + 30 2(g) 2Al 2...
A:
Q: An buffer is prepared by combining 4 L of a 0.57 M X solution with 4 L of an 0.57 molar HXCI solutio...
A: Weak base are those base which does not dissociate completely in aqueous solution. Here X is weak ba...
Q: %23 H H CHs H-C-ć-H CH3 H-C-c-H HC=O НС—о н ОН Но H ČH,OH ČH,OH 2 CI CI CI H C = C C = C 6. нн H CI ...
A: Answer: When two molecules have same molecular formula but different structural formula or spatial o...
Q: For Problem no. 21-22 At elevated temperatures, dinitrogen pentoxide decomposes to nitrogen dioxide ...
A:
Q: The fumarate ion is a dibasic base with pKb values of 9.52 and 10.98. What is the concentration of t...
A: Given the pH of the solution = 5.21 Total concentration of both the buffer parts = 0.100 M
Q: Helium-filled balloons are used to carry scientific instruments high into the atmosphere. Suppose th...
A: Helium-filled balloons are used to carry scientific instruments high into the atmosphere. Suppose th...
Q: EQUATION WRITING: Balance the following redox reactions. Use change in oxidation state method (oxida...
A:
Q: For Problem no. 21-22 At elevated temperatures, dinitrogen pentoxide decomposes to nitrogen dioxide ...
A:
Q: 3. Describe the crystal structure of iron, which crystallizes with two equivalent metal atoms in a c...
A:
Q: Consider 3-methylpent-2-ene, if it undergoes hydroboration, which of the following final product is ...
A: Given - compound 3-methylpent-2-ene
Q: The following reaction sequence is described as an introductory organic chemistry laboratory experim...
A: In synthetic organic chemistry, the combination of appropriate reactions under suitable conditions o...
Q: Calculate the pH of a 7.51 M solution of HBrO. The K, of HBRO is 2.50 x 10.
A: pH = -log[H+] Ka for an acid is the ratio of product of concentration of products raised to their ...
Q: Use the paperclip button below to attach files. Student can enter max 3500 characters ckNav
A: Proteins are building blocks of body and can also serve as a fuel source. As a fuel, proteins provid...
Q: Relating Density to Crystal Structure 1. Show that the atomic packing factor for FCC is 0.74. 2. Sho...
A:
Q: 1. Show all pertinent solutions neatly, orderly and completely. Indicate the oxidation state of the ...
A: Oxidation state can be defined as the total number of electrons that an atom either lose or gain fo...
Q: Which among these objects should you use to filter the precipitate in precipitation gravimetry? D...
A: Please find your solution below : Gravimetry is a method of quantitative determination of analyte on...
Q: Determine the chirality of molecules 1, 2, and 3. Describe the relationship between them. HO SH HO,....
A: Since you have asked multiple questions, we will solve the first question for you as per our guideli...
Q: 2. Which of the following structural formulas represent identical compounds and which represent cons...
A: for the given structures, we need to identify the structures that are identical and the structures t...
Q: Calculate the standard energy change, A EO, for the reaction below. 12NH 3(g) + 210 2(g) 8HNO 3 (E) ...
A:
Q: Predict the effect of decreasing the container volume on the amounts of each reactant and product in...
A:
Q: A 25.00 mL sample of acetic acid (CH3COOH) is titrated to the equivalence using 31.08 mL of 0.1978 M...
A:
Q: 1. Cu HNO3 → NO2 + Cu(NO3)2 H2O
A: According to guidelines I am supposed to answer only first question
Q: 30) What is the percent yield for the reaction below? Only 28.16 g of CO2 are observed to have forme...
A: Actual yield of CO2=28.16g Theoretical yield of CO2=225.3g Moles of C8H18=4mol Moles of O2=8mol
Assign an IUPAC name to each of the following compounds:
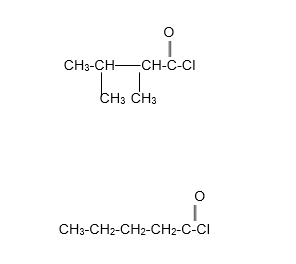
Step by step
Solved in 2 steps with 1 images

- What is [2 + 2] Cycloadditions ?Considering the following structures (I~V), answer the questions A-J. There may be more than one answer for a given question2. Give each of the following compounds an appropriate name.a. (C2H5)2Ob. CH3(CH2)2C(OH)(CH3)C2H5c. CH2OH(CH2)3CH(OH)CH3d. CH3CH2OCH2(CH3)C3H7 (n)e. CH3OC6H9 (neo
- Cyclopropane and cyclobutane have similar strain energy despite the fact that the C—C—C bond angles of cyclopropane are much smaller than those of cyclobutane. Suggest an explanation for this observation, considering all sources of strain discussed in Chapter 4.Rings + Unsaturation --- Hydrogenation If compound A C51H81BrN5O3P3 is hydrogenated to give compound B C51H101BrN5O3P3. How many rings does compound A have? Assume that P has a valency of 5. Would the answer be 4 rings? Formula -> unsat + rings = 1+C +N/2 - H/2 - X/2Assign priorities to these set of Substituents a) -CH2CH2OH,-Br,-H, -CH2CH3 b) -COOH,-COOCH3,-CH2OH,-OH c) –CH2NHCH3,-CH2NH2,-NH2,-CN d) –Br,-CH2Br,-Cl,-CH2Cl e) –OH, –CH2CH3,–CH=CH2,–OCH2CH3 f) –NH2 , –NO2 , –NHCH3 , g) –CH2CH3 , –CH2SH , –CH(CH3)2 , h)–CH(CH3)2,–C(CH3)3,–CH2CH(CH3)2, –CH2C(CH3)3 i) –OCl , –NHCl , –OBr , –NHBr
- (a) Draw in the curved arrows to show how A is converted to B in Step -1-. (b) Identify X, using the curved arrows drawn for Step -2-.Consider the molecule 1-bromo-2-methylbutane. C3 and C4 should be drawn as Et as in theexample. This group is called an ethyl group and can be considered a sphere about twice the sizeof a methyl group. Draw the following Newman projections sighting down the C1C2 bond... a. The lowest potential energy conformation. b. The highest potential energy staggered conformation.Draw the most stable conformation of pentane, using wedges and dashes to represent bonds coming out of the paper and going behind the paper, respectively.



