Chemical formula & total valence e- Electron Group Geom. & Molec. Geom. Molecule Polarity Bond Bond Polarity (show calc) Lewis Structure Model Sketch Angle PCI, CH. SiOz CH,0
Chemical formula & total valence e- Electron Group Geom. & Molec. Geom. Molecule Polarity Bond Bond Polarity (show calc) Lewis Structure Model Sketch Angle PCI, CH. SiOz CH,0
Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Andrei Straumanis
Chapter2: Lewis Structures
Section: Chapter Questions
Problem 12CTQ
Related questions
Question
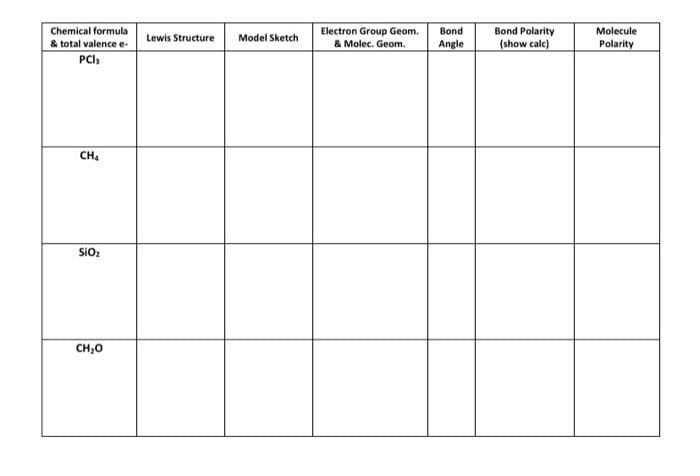
Transcribed Image Text:Chemical formula
& total valence e-
PCI
Electron Group Geom.
Bond
Bond Polarity
Molecule
Lewis Structure
Model Sketch
& Molec. Geom.
Angle
(show calc)
Polarity
CH4
SiO
CH,0
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning