Chemistry First, identify whether these compounds are ionic or covalent. Then, use the correct formula writing rules to write the correct chemical formulas for each compound. Type of Compound: lonic or Covalent Compound Name Chemical Formula 1) copper (II) chlorite Ionic 2) sodium hydroxide Ionic NaOH 3) nitrogen dioxide Covalent NO2 4) cobalt (III) oxalate 5) ammonium sulfide 6) aluminum cyanide 7) carbon disulfide 8) tetraphosphorous pentoxide 9) potassium permanganate 10) manganese (III) chloride Compound Name Type of Compound: lonic or Covalent Chemical Formula 11) calcium bromate 12) carbon monoxide 13) potassium oxide 14) antimony tribromide 15) zinc phosphate 16) copper (II) bicarbonate 17) dinitrogen tetroxide 18) manganese (IV) carbonate 19) lead (IV) nitride 20) pentacarbon decahydride
Chemistry First, identify whether these compounds are ionic or covalent. Then, use the correct formula writing rules to write the correct chemical formulas for each compound. Type of Compound: lonic or Covalent Compound Name Chemical Formula 1) copper (II) chlorite Ionic 2) sodium hydroxide Ionic NaOH 3) nitrogen dioxide Covalent NO2 4) cobalt (III) oxalate 5) ammonium sulfide 6) aluminum cyanide 7) carbon disulfide 8) tetraphosphorous pentoxide 9) potassium permanganate 10) manganese (III) chloride Compound Name Type of Compound: lonic or Covalent Chemical Formula 11) calcium bromate 12) carbon monoxide 13) potassium oxide 14) antimony tribromide 15) zinc phosphate 16) copper (II) bicarbonate 17) dinitrogen tetroxide 18) manganese (IV) carbonate 19) lead (IV) nitride 20) pentacarbon decahydride
Chemistry by OpenStax (2015-05-04)
1st Edition
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Chapter7: Chemical Bonding And Molecular Geometry
Section: Chapter Questions
Problem 13E: Predict which of the following compounds are ionic and which are covalent, based on the location of...
Related questions
Question
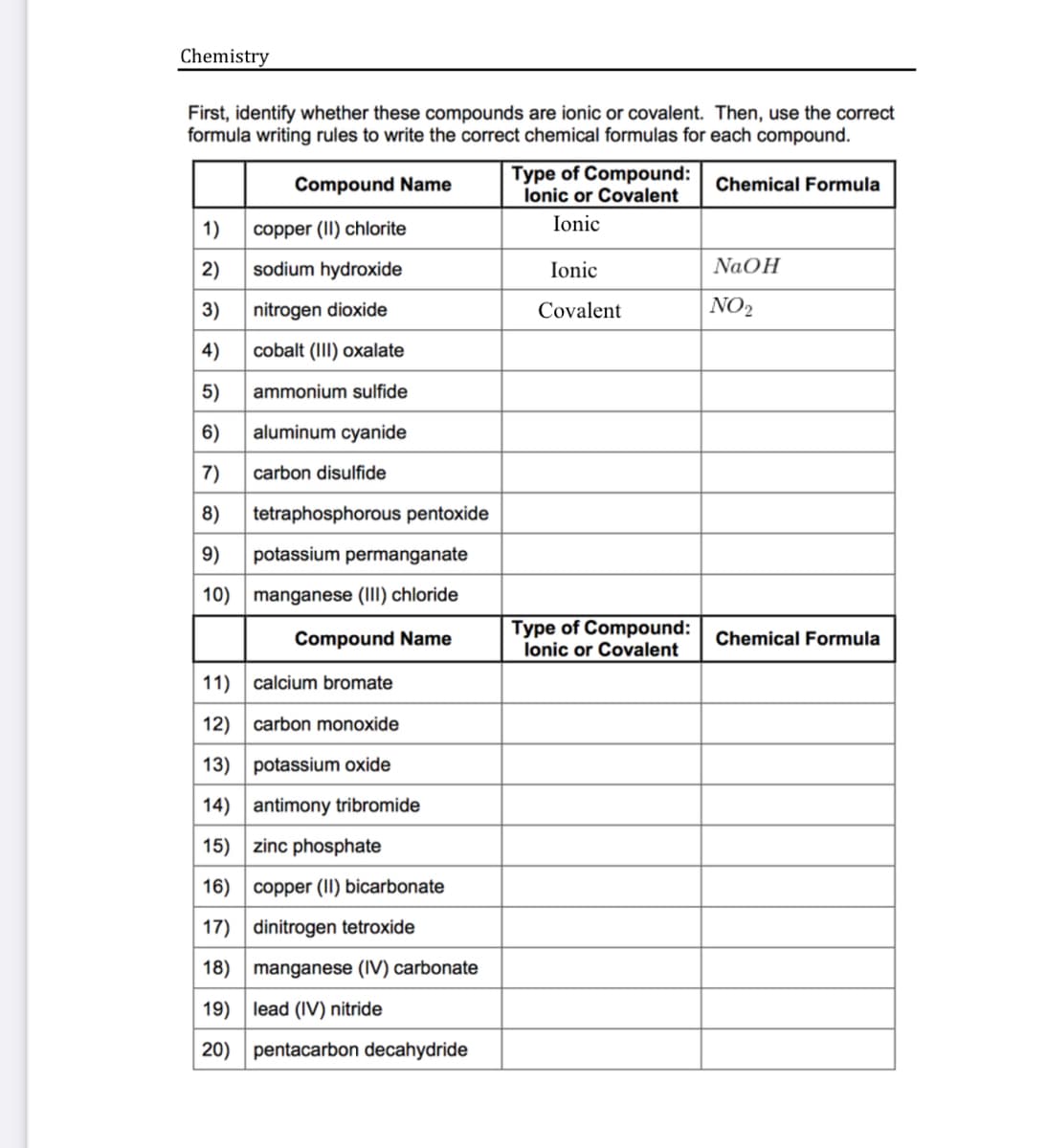
Transcribed Image Text:Chemistry
First, identify whether these compounds are ionic or covalent. Then, use the correct
formula writing rules to write the correct chemical formulas for each compound.
Compound Name
Type of Compound:
lonic or Covalent
Chemical Formula
1)
copper (II) chlorite
Ionic
2)
sodium hydroxide
Ionic
NaOH
3)
nitrogen dioxide
Covalent
NO2
4)
cobalt (III) oxalate
5)
ammonium sulfide
6)
aluminum cyanide
7)
carbon disulfide
8)
tetraphosphorous pentoxide
9)
potassium permanganate
10) manganese (III) chloride
Compound Name
Type of Compound:
lonic or Covalent
Chemical Formula
11) calcium bromate
12) carbon monoxide
13) potassium oxide
14) antimony tribromide
15) zinc phosphate
16) copper (II) bicarbonate
17) dinitrogen tetroxide
18) manganese (IV) carbonate
19) lead (IV) nitride
20) pentacarbon decahydride
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning


Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning


World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co