A voltaic cell utilizes the following reaction: 2Fe+(ag) + H2 (g)→2Fe2+(ag) +2H+(aq). Part A What is the emf of this cell under standard conditions? Express your answer using three significant figures. E° = 0.771 V Submit Previous Answers Correct Part B What is the emf for this cell when (Fe+1 = 3.90 M, P, = 0.99 atm, [Fe?+1 =7.0×104M, and the pH in both compartments is 3.80? Express your answer using two significant figures. E = Submit Request Answer
A voltaic cell utilizes the following reaction: 2Fe+(ag) + H2 (g)→2Fe2+(ag) +2H+(aq). Part A What is the emf of this cell under standard conditions? Express your answer using three significant figures. E° = 0.771 V Submit Previous Answers Correct Part B What is the emf for this cell when (Fe+1 = 3.90 M, P, = 0.99 atm, [Fe?+1 =7.0×104M, and the pH in both compartments is 3.80? Express your answer using two significant figures. E = Submit Request Answer
Chemistry: An Atoms First Approach
2nd Edition
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Steven S. Zumdahl, Susan A. Zumdahl
Chapter17: Electrochemistry
Section: Chapter Questions
Problem 135CWP: Consider a galvanic cell based on the following half-reactions: a. What is the expected cell...
Related questions
Question
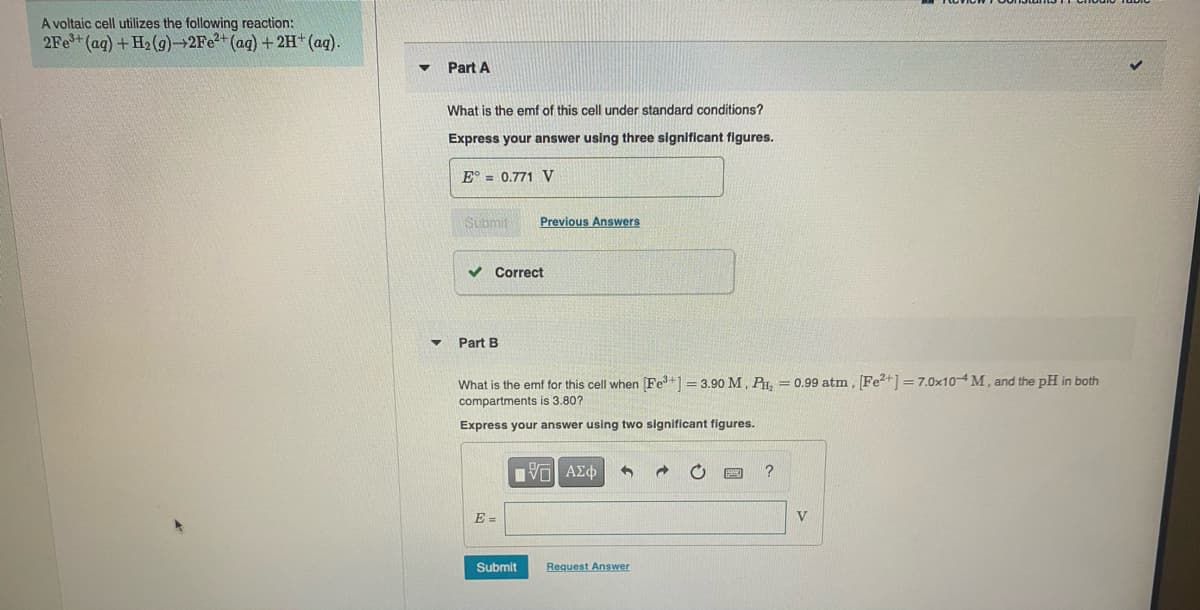
Transcribed Image Text:A voltaic cell utilizes the following reaction:
2Fe+(ag) + H2 (g)→2Fe2+(ag) +2H+(aq).
Part A
What is the emf of this cell under standard conditions?
Express your answer using three significant figures.
E° = 0.771 V
Submit
Previous Answers
Correct
Part B
What is the emf for this cell when (Fe+1 = 3.90 M, P, = 0.99 atm, [Fe?+1 =7.0×104M, and the pH in both
compartments is 3.80?
Express your answer using two significant figures.
E =
Submit
Request Answer
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning