Intensity (counts)- CRYSTALLINE STRUCTURE MORPHOLOGY COMPOSITION TOPOGRAPHY 32000 NIK Cr 0.01 Energy (keV) Figure 5.32. EDS spectrum of a high-temperature superalloy. COMPOSITION MORPHOLOGY TOPOGRAPHY CRYSTALLINE STRUCTURE 0 Tags #192 R Helys 10.00
Intensity (counts)- CRYSTALLINE STRUCTURE MORPHOLOGY COMPOSITION TOPOGRAPHY 32000 NIK Cr 0.01 Energy (keV) Figure 5.32. EDS spectrum of a high-temperature superalloy. COMPOSITION MORPHOLOGY TOPOGRAPHY CRYSTALLINE STRUCTURE 0 Tags #192 R Helys 10.00
Physical Chemistry
2nd Edition
ISBN:9781133958437
Author:Ball, David W. (david Warren), BAER, Tomas
Publisher:Ball, David W. (david Warren), BAER, Tomas
Chapter21: The Solid State: Crystals
Section: Chapter Questions
Problem 21.3E
Related questions
Question
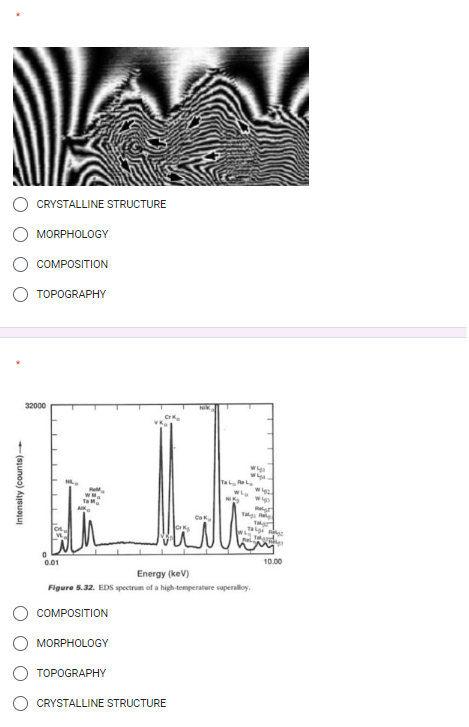
Transcribed Image Text:Intensity (counts)-
CRYSTALLINE STRUCTURE
MORPHOLOGY
COMPOSITION
TOPOGRAPHY
32000
NIK
Cr
0.01
Energy (keV)
Figure 5.32. EDS spectrum of a high-temperature superalloy.
COMPOSITION
MORPHOLOGY
TOPOGRAPHY
CRYSTALLINE STRUCTURE
0
Tags
#192
R
Helys
10.00
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Recommended textbooks for you

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,