Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Andrei Straumanis
Chapter2: Lewis Structures
Section: Chapter Questions
Problem 12CTQ
Related questions
Question
Transcribed Image Text:101 Chem101
b My Questions | bartleby
x +
->
app.101edu.co
M
Apps
G
M Gmail
YouTube
Maps
a AMAZON
Translate
O Gflights
USCIS
ъ ВАТERBLY
C CHEGG > KATAPULK CUBA
SUPERMARKET23
Essay Writing Ser...
G calculator - Googl...
Reading List
Question 26 of 45
Submit
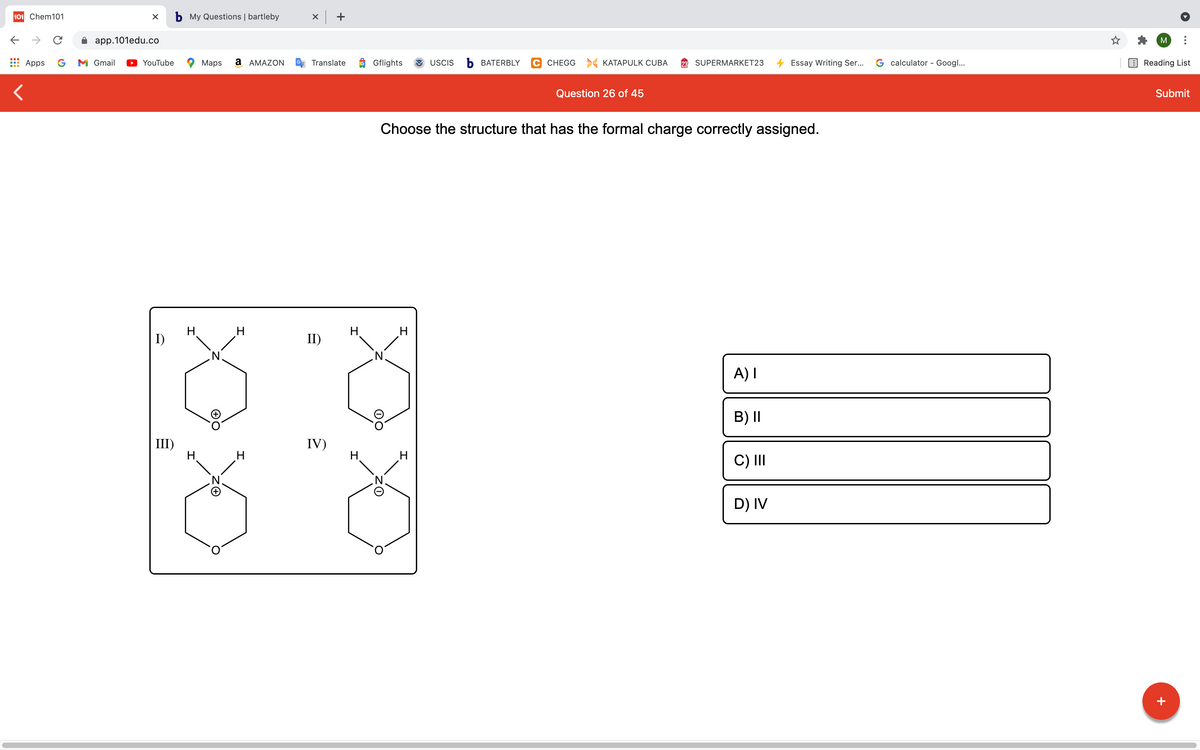
Choose the structure that has the formal charge correctly assigned.
H.
I)
II)
H
A) I
B) I|
III)
H.
IV)
H.
C) II
D) IV
+
エ
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning
