Content I:sduy ga Holiday Inn Youngstown S... them. s-1.Q.2021.04.20q.doc Page 1 of 5 Pages in Total 4/2 MacBook Air 08 DD F7 F4 F5 F6 64 F10 2$ & V * 5.
Q: Convert a speed of 251 cm/s to units of inches per minute. Also, show the unit analysis by dragging…
A: We have to convert speed in inch/meter.
Q: Chenustry II CHiM4.001 O https://blackboard.liu.edu/webapps/assessment/take/take.jsp?course…
A: Alcohols can be defined as the otganic compoud in which carbon atom is attached to hydroxyl group.
Q: apha Dorley,- Learn…
A: The given reaction is : For the first order reaction the rate constant, k is given by :
Q: II Rb Courses- Blackboard Learn > General Psychology - Fall 2021 A ALEKS - Griffin Barden - Learn…
A: Given that : The heat energy absorbed by the chemical reaction = 350 kJ The heat energy flowed in…
Q: A liquid flows initially into a vessel initially empty to a steady rate of 70 cm³ s'. The pictorial…
A: Since you have asked multiple questions, we will solve first three sub-parts for you. If you want…
Q: Mg Metal MgO Trial 1 Trial 2 Trial 1 Trial 2 Volume HCI 100 mL 100ML 0.2550g 58001 O.0276 moL Mass…
A: Given: Mg Metal MgO Volume of HCl 100 mL 100 mL Mass of solid Added 0.2550 g 1.008 g…
Q: Two carts mounted on an air track are moving toward one another. Cart 1 has a speed of 1.1 m/s and a…
A: Given, mass of cart 1 =0.45 kg speed of cart 1 = 1.1 m/s mass of cart 2 = 0.75 kg
Q: ces Experimental data for the density of n-pentane liquid for high pressures, at 50°C, are listed as…
A:
Q: Thermal Equilibrium Aluminum : 2.5 9 ) Cp= 0.897 J /gc mass T; = 500°C 300 g %3D water : Cp 4.189…
A: The detailed solution of your question given below-- Since, we know that the law of conservation of…
Q: Help
A: Given data, 0.150 M NaOH (M1) Volume of HCl (V2) =50 mL Molarity of HCl (M2) =0.120 M
Q: 2. A trench mortar fires a shell at angle of 30° with the horizontal with an initial velocity of 500…
A:
Q: Weicome students Mancopa X O Quiz 11 (CHEM101) Chem101 i app.101edu.co Question 1 of 29 Convert 2.70…
A: So we have given pressure= 2.7 atm We will convert it into torr.
Q: Consider the problem below: (Equation 1) H2(g) + F2(g) ---> 2 HF(e) AH = -542.2 kJ/mol %3D (Equation…
A:
Q: 20.77°C -300mL 600mL - 200mL 500m 400mL 300mL 200mL 100mL 100mL SOLIDS SOLUTIONS LIQUIDS SOLUTIONS…
A:
Q: Q4-A vertical cylinder tank with diameter of 12m and depth of 4m is filled to the top with water at…
A: Given thatDiameter of vertical cylinder=12 mDepth of vertical cylinder=4mInitial…
Q: 12. y= xVa²- x² + a²sin1÷, where a is a parameter
A:
Q: When the velocity of enzyme is equal to 80% Vmax, Km is equal to A [S] 1/2 [S] B 1/3 [S] C 1/4 [S]
A: Km is the concentration of substrate at which the rate of the reaction(v) is the half of the Vmax ,…
Q: A waste treatment pond is 50 m long and 25 m wide, and has an average depth of 2 m. The density of…
A: Given that: Length of the pond (l) = 50 m = 164.04…
Q: It's possible that poor software project management was to blame for the failure of the baggage…
A: Ans. Failure of the baggage system at Denver International Airport: The Denver International…
Q: E Give Up? Resources O Hint Match each variable to the corresponding scenario. Steve buys a…
A: For each scenario there is a change in variable which is identified in step two.
Q: The following data are for the system 3A(g) + B(g) ⇌ 2C(g) Find PB and PC after 1 min
A: When two reactive compounds are mixed, then they react according to the stoichiometry of the…
Q: 5) In some system with a constant composition, U is a function of V and T. Write the differential…
A: Internal energy is a function of the volume and the temperature. In a closed system of constant…
Q: Content A ALEKS - DOMINGO BAYONITO -> A…
A: Mole can be defined as a standard unit for measuring large quantities of very small entities such as…
Q: Time Poential 200 Tere 1a700 mn minl 1000000 2 0.007700 3 0.015400 0.023100 0.030800 60.038500 7…
A: Peak 3 is corresponding to acetic acid. Peak 2 is corresponding to ether group. Peak 1 is…
Q: L- Learn…
A: Step1: The given values for mercury(I) chloride (Hg2Cl2) solution are as follows: Here "mM" is…
Q: A drug is infused continuously intravenously (IV) for treatment of a particular disease. The volume…
A: The rate of the reaction is referred to the change in the molar concentration in the distinct…
Q: Graph the data below to determine the appropiate regression. Use your regression equation to…
A:
Q: Nodes A) 1s B) 2s C) 3s D) 2p E) 3p
A: Nodes are the region where the electron density of orbital is zero. In the above figure, there are 2…
Q: Staple your graph to the data sheet. Complete the following table: T-1(K-1) In(VP) 353 aw3 =0.00297…
A: From the given data first we will find out the ln(VP) values for 1 mole. Then we will draw the…
Q: Help
A: Balancing of reaction – A chemical reaction involves reactants and products. According to law of…
Q: solve for CS (show work please!) (22.05g) CS (28.49-100) + (50g)(4.184 J/g°C) (28.49- 25.1) =0
A: The given problem is as following:- (22.05g) Cs (28.49 -100) + (50 g) (4.184 J/goC) (28.49 - 25.1)…
Q: It is desired to obtain a functional relationship between the mass density p of air and the altitude…
A: is the given relation. Take log on either side ln This is a linear relation with slope =k2 and y…
Q: Water flows through a pipe with an inside diameter of 10mm at the rate of 1.2 m^3/min. Compute for…
A: Velocity of water= volume flow rate ÷ area of cross section
Q: Do expect to get similar data from visual and VS/Avis data collection? Explain
A: A question based on UV and visible spectrum that is to be accomplished.
Q: - Rule violated: 2p 4个4 f t1t4t Rule violated: - 2p- 3s 3p
A: Since you have asked a question with multiple sub-parts, we will solve first three sub-parts for…
Q: -NH NO2 -NH2 NO2 + HCI 12 10 11 NO2 NO2 C,HgN Mw: 107.1560 CSH3CIN2O4 Mw: 202.5500 C13H1|N3O4 Mw:…
A:
Q: 4. Several process variables streams are shown below. Compute the indicated quantities in terms of…
A:
Q: A…
A: According to the ideal gas equation - PV = wRT/M Here - P = Pressure V = Volume = 10 L w = mass of…
Q: The following data are for the system 3A(g) + B(g) = 2C(g) Time (min)0 PA (atm) 0.850…
A: Given a reversible reaction: 3A(g) + B(g) <-----> 2C
Q: I'm having trouble with the major reactions to these three-could you please help me?
A: 1. When carbonyl reacts with amine they form an enamine. 2. Carbonyl reacts with phosphonium yilde…
Q: Th Course: CHEM262-SEC01 Org: x O OWL Exam U Noor Abid - 32843659 s x b Answered: Place the…
A: When germinal di carboxylic acid on heating wil undergo decarboxylation and produce CO2
Q: E. Measuring the Density of Antifreeze 23. 20.0 °C Temperature of antifreeze Trial 3 Trial 2 Trial 1…
A: Mass of Antifreeze=(Mass of flask+anti freeze - mass of flask) Trial 1 Mass of anti…
Q: 100kg/hr water is heated from 25 C to 200 C. Steam enters at 500kPa and 973.15K and leaves at 300kPa…
A:
Q: Convert a speed of 973 cm/s to units of inches per minute. Also, show the unit analysis by dragging…
A: The conversion units required are 1S = 160 min 1cm = 12.54 in
Q: 16 liters. The drug is excreted (removed) through the kidneys at rate ke , proportional to the…
A: Solution -
Q: Q. 9 is the one I need help with
A:
Q: can you show the work for problem numbe
A: XZ A where X = atom Z = atomic number = no.of protons (p)…
fill in the structure with any assumed c & h atoms. how do you know if or if not they belong there?
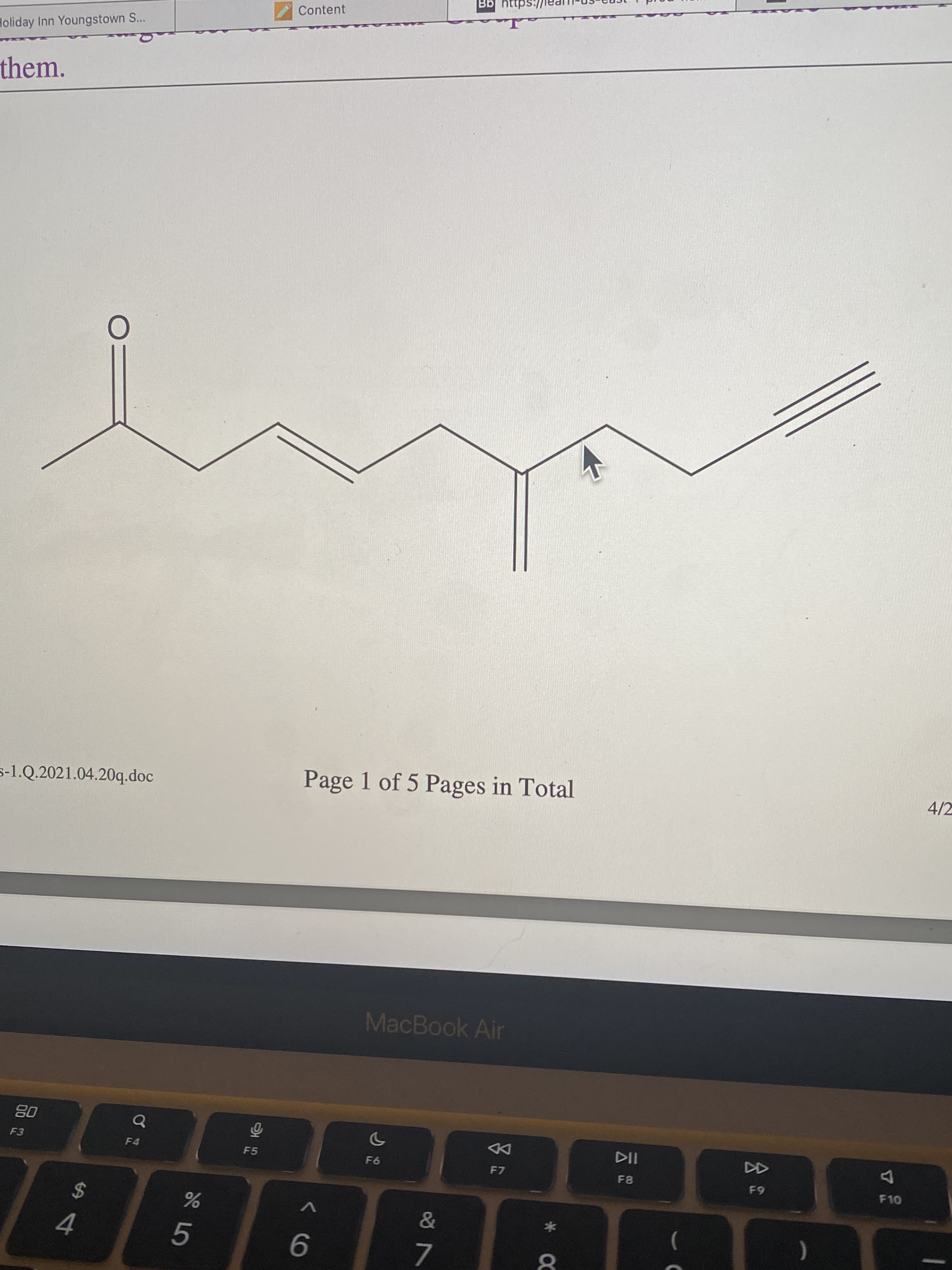
Step by step
Solved in 2 steps with 1 images

- PV = nRT. The pressure is 0.9912atm. V = 50.5 mL. T = 21.5°C and n = 0.00200mol. Calculate R from this data in Latm/molK.A drug is infused continuously intravenously (IV) for treatment of a particular disease. The volume of the body fluid V = 16 liters. The drug is excreted (removed) through the kidneys at rate ke , proportional to the concentration of the drug in the body fluids. The drug is ineffective and not used by the body if its concentration is below Cmin and becomes poisonous if the concentration goes over Cmax . It is metabolized at a rate km as the concentration is >=Cmin. Let the parameter values Cmin= 4 mg/l, Cmax =40 mg/l, ke = 0.2/hr and km = 0.3/hr.a. How much is the maximum possible rate of infusion of the drug?b. How long the drug would continue to be effective if the infusion has been stopped as the concentration reached at the maximum level?Provide the remaining data missing
- If may calculations po, please show all steps leading to the final answer po. Here’s the link in accordance with the topic po: https://drive.google.com/file/d/1_FnDtXCrFKSol3RNWIG_9tNQ7IxgxD6t/view?usp=drivesdkForgot to put the units they are asking for on the previous submission. Am I doing this correctly? A gaseous sample of molecules, with a molar mass of 87.9 g/mol, is contained in a cylinder at a temperature of 362. K and pressure of 101.9 kPa. The collision cross section of a molecule is 0.73 nm2 The collision freqency, z, of the gas under these conditions is ____________ s-1.How to set up for problem 2
- What is the Molecular weight of a certain volatile liquid which is placed in 225 g flask which has total capacity volume of 252.17 mL. After the liquid was heated to 96.7 oC the gass volatilized and the weight of the flask measure 227.33g. What is the could be the MW of the volatile sample?Show solution : if 12m³ of nitrogen at constant temp of 30°c and 12kpa abs is permitted to expand to 30m³, what is the resulting pressure? Ans. 50 kpahttps://m.youtube.com/watch?v=vM1SP346XBc&list=PLeJOSNLNZfHubfLdq0kOayASeUllMOGn4&index=4 I watched this the lecture video over and over and I am allowed to work with someone but I am having trouble with part B and I provided the YouTube link of the data or video attached to this lab
- Basis: https://youtu.be/Y4NMpO1xI8UA monolayer of CO molecules is adsorbed on the surface of 1.00 g of an Fe/AI2O3 catalyst at 77 K, the boiling point of liquid nitrogen. Upon warming, the carbon monoxide occupies 4.25 cm3 at O oC and 1.00 bar. What is the surface area of the catalyst?One of the highest concentrations of peroxyacyl nitrates has been found in the Los Angeles (LA) basin at 30 ppbppb, reaching its peak at mid-afternoon. Most of the pollution in the LA basin is created in an area of only 3000 km2km2 and the pollution depth below the inversion layer is normally about 500 mm. This concentration was therefore found in 1500 km3km3 of air above Los Angeles. Peroxyacyl nitrates contain two relatively weak bonds that can be broken by low-energy photons. The weakest is the N−ON−O bond in the nitrate functional group of the molecule with a standard bond-dissociation energy of 201 kJ mol−1. What is the longest wavelength of a photon, in nanometres, that has enough energy to cause the photodissociation of the weakest bond. Assume all energy of the photon is efficiently used for the photodissociation.

