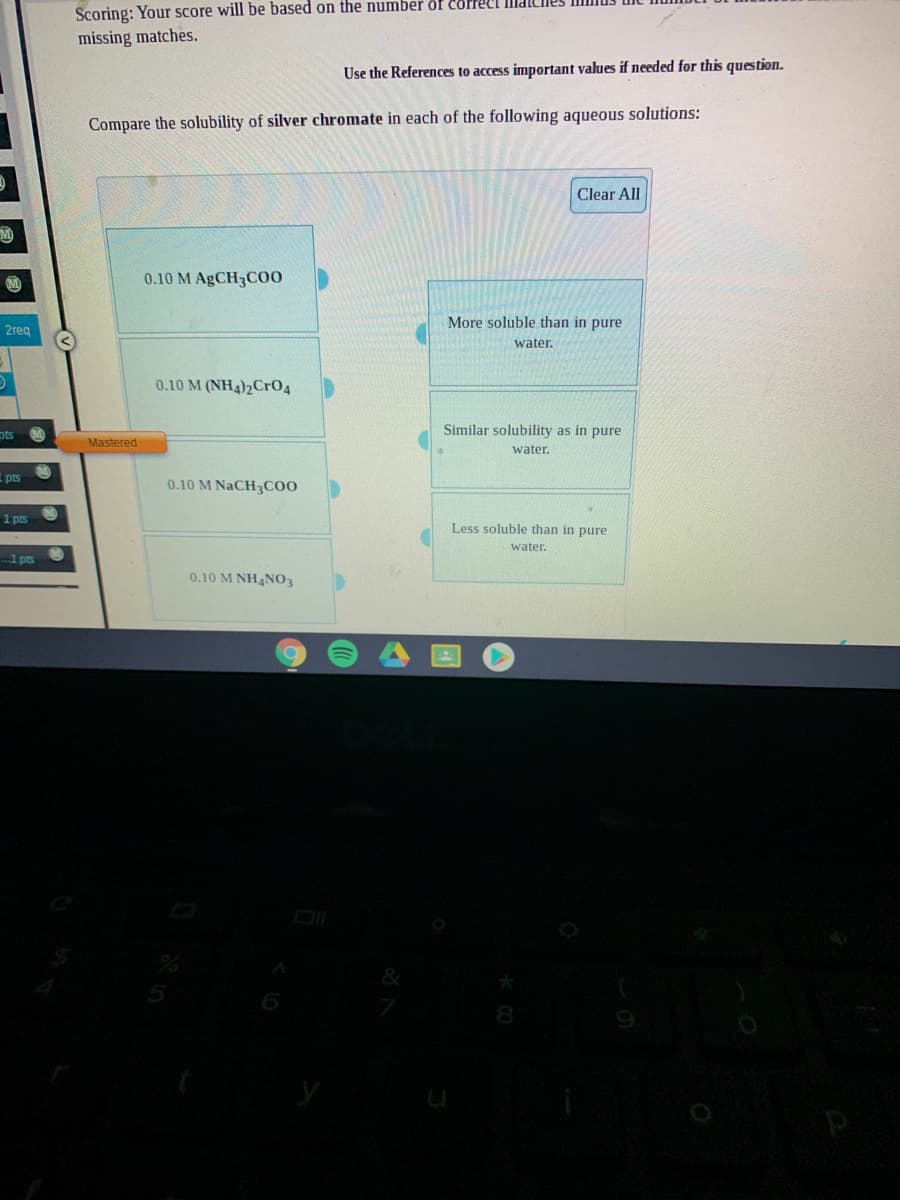
Compare the solubility of silver chromate in each of the following aqueous solutions: Clear All 0.10 M AgCH3CO0 More soluble than in pure water. 0.10 M (NH4)2Cr04 Similar solubility as in pure Mastered water. 0.10 M NaCH3C0O Less soluble than in pure water. 0.10 M NHNO3
Compare the solubility of silver chromate in each of the following aqueous solutions: Clear All 0.10 M AgCH3CO0 More soluble than in pure water. 0.10 M (NH4)2Cr04 Similar solubility as in pure Mastered water. 0.10 M NaCH3C0O Less soluble than in pure water. 0.10 M NHNO3
Chapter17: Complexation And Precipitation Reactions And Titrations
Section: Chapter Questions
Problem 17.15QAP
Related questions
Question
Q 7b.
Transcribed Image Text:Scoring: Your score will be based on the number of correel maICnes
missing matches.
Use the References to access important values if needed for this question.
Compare the solubility of silver chromate in each of the following aqueous solutions:
Clear All
M)
0.10 M AGCH3C00
More soluble than in pure
2reg
water.
0.10 M (NH4)2CrO4
ots
Similar solubility as in pure
Mastered
water.
pts
0.10 M NaCH3CO0
1 pts
Less soluble than in pure
water.
1 pts
0.10 M NH4NO3
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

