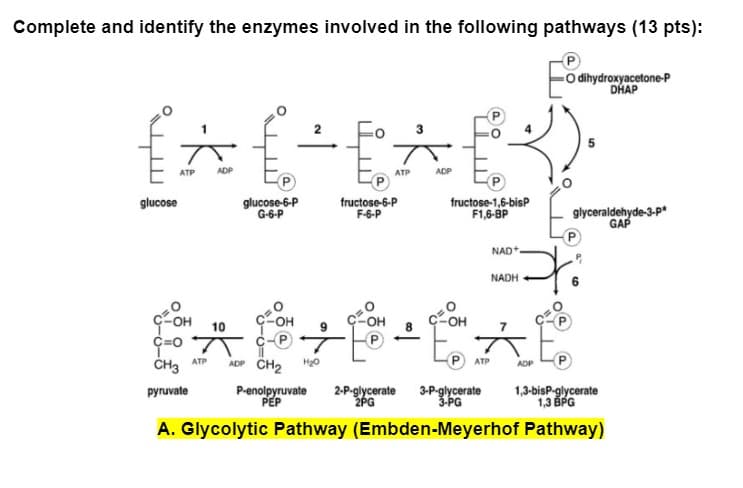
Complete and identify the enzymes involved in the following pathways (13 pts): -Odihydroxyacetone-P DHAP {^{-E^E). ATP ADP ATP ADP glucose glucose-6-P G-6-P fructose-6-P F-6-P fructose-1,6-bisP F1,6-BP NAD NADH glyceraldehyde-3-p* GAP pr E COH 10 ç=O CH3 ATP ADP CH₂ H₂O ATP ADP pyruvate P-enolpyruvate 2-P-glycerate 3-P-glycerate PEP 2PG 3-PG 1,3-bisP-glycerate 1,3 BPG A. Glycolytic Pathway (Embden-Meyerhof Pathway)
Q: A buffer system consists of A. a weak acid and its conjugate base. B. a weak acid and a proton…
A: A buffer is a aqueous solution which is used to resist the pH change upon addition of acid or base.…
Q: Prepare a 75 ml of a 10% (w/v) FeCl3 solution. How many grams of FeCl3 must be weighed out and…
A: Given Values: Volume of FeCl3 solution = 75 ml Concentration of the FeCl3 = 10% (w/v)
Q: 2. A 4-month-old child has signs of rickets. Digestive disorders were not noted. The child has had…
A: Calcitriol (1,25-DHCC) is the biologically active form of vitamin D. It regulates the plasma levels…
Q: Dissipation of the proton gradient is required for Formation of ATP from ADP+Pi…
A: As during electron transport chain, the proton gradient created by proton pumping is utilised to…
Q: Estimate the fragment sizes (bp) of the DNA bands from each sample lane
A: Introduction: In the gel electrophoresis, agarose derived from red seaweed acts as a sieve for…
Q: glycolysis step 7, ATP synthesis occurs through ______. The energy for this reaction comes _____…
A: Glycolysis is the process of oxidation of glucose into smaller molecules.it occurs in two phases as…
Q: Palmitoyl CoA is an inhibitor of acetyl CoA carboxylase. Group of answer choices True False
A: A thioester of acyl-CoA is palmitoyl-CoA. It's a "activated" version of palmitic acid that can be…
Q: What happens to a person's body when they consume dairy products when they are lactose intolerant?…
A: Introduction: Lactose is a disaccharide containing β-D-galactose and β-D-glucose in β (1,4) linkage.…
Q: H-C FADH₂ FAD H₂O COO™ 1 C-H COO™™ Fumarate ADP ATP COO™ HO–CH CH₂ COO™ Malate NAD COO™ CH₂ CH₂ COO™…
A: TThe citric acid cycle (CAC) is the key metabolic pathway responsible for the oxidative degradation…
Q: Identify the types of linkages in each of the three disaccharides. Only place three of the linkages.
A: Disaccharides are consist of two monosaccharides units linked by glycosidic bonds. The type of…
Q: Prepare a standard tube that contains 0.2 mL of 2.5 mM lysine. To this tube, add 0.1 mL of the…
A: Final concentration can be calculated from the formula C1V1=C2V2 Where C1= initial concentration V1=…
Q: Describe the hallmarks of the GLUT transporter – a passive glucose transporter, and the basis behind…
A: Glucose transporter 1 (or GLUT1) is involved in the facilitated transport of glucose across the…
Q: Please state if the statements are true or false. 1. An aplha 1,4 glycosidic linkage is considered…
A: The most common carbohydrates in diet are polysaccharides. They're polymeric carbohydrates with…
Q: Which of the following statements is TRUE about fatty acid activation before B -oxidation? The…
A: The fatty acids that are released during the digestion of triglycerides are broken down in by…
Q: Explain how enzymes work to speed up reactions
A: Enzymes are biological catalysts, function to speed up the reactions
Q: Draw a standard workflow on plant metabolite profiling using GC-MS.
A: GC- MS stands for Gas chromatography Mass spectrometry. GC-MS is used for drug development ,…
Q: Using good details, show how the function of two or three different nucleic acids is connected to…
A: Nucleic acids are chemical substances that occur naturally and act as the primary…
Q: Regarding the production of therapeutic proteins, which one of the following statements is not…
A: Post-translational modification refers to the covalent and generally enzymatic modification of…
Q: Find the connection among the words below and choose the letter of the word which is different. O A.…
A: Electrophoresis is run to separate the DNA, RNA, and protein molecules on the basis of their…
Q: In centrifugation, the components of a given mixture are subjected to CENTRIFUGAL force, which…
A: Cell organelles include mitochondria, nuclei, vacuoles, ribosomes, plastids, peroxisomes, and…
Q: Consider the carbohydrate maltose. a. How many molecules of acetyl CoA are formed from its complete…
A: Maltase is the enzyme that transforms maltose into glucose. During digestion, pancreatic or salivary…
Q: Give applications of enzyme kinetics
A: Enzyme kinetics: It is the quantitative study of enzyme catalysis. It measures reaction rates and…
Q: Which of the following is MOST soluble in polar solvents? A H3C LOH В с D В OD Oc О А H3C. HO л HC…
A: In the given image, structures A, B, and C represents fatty acids. A is a saturated fatty acid.…
Q: Describe briefly the hydrolysis reaction for carbohydrates as indicated to the picture A. What organ…
A: Insulin and glucagon are the hormones that control blood sugar levels. These hormones are of…
Q: What are the sequencing methods, strengths, and limitations used in 2022 for human genome?
A: The entire human genome has nucleic acid sequences which make up the genes for humans, these are…
Q: NH3 eo
A: The lipid molecules are amphipathic molecules which means they have polar and non-polar functional…
Q: write argumentative essay on, can bio plastics replace synthetic plastics ? if yes why so if no why…
A: There has been a surge in interest in biodegradable materials for agriculture, packaging, medicine,…
Q: In a Venn diagram compare the palmitic acid and the oleic acid.
A: Based on the structure provided (actual oleic acid acid is monounsaturated and 18 carbon long) the…
Q: QUESTION 2 CH3COO-C₂H5 + H₂O CH3COOH + C₂H5OH Ethyl acetate Acetate Ethanol Ethyl acetate, acetate…
A: Ethyl acetate, acetate and ethanol were added to water solution in concentrations of 0.3M and left…
Q: enzyme that converts succinate to fumarate, uses ____ as its coenzyme. FADH2 FAD…
A: Succinate to fumarate reaction- It occurs in the citric acid cycle
Q: Why is DNA so important
A: DNA is an abbreviation for deoxyribonucleic acid. It includes nucleotides, which are biological…
Q: A hamster receives a dose of 3 mg. of a compound and then 2/3 as much as the previous dose at the…
A: Regardless of the reason for administration, drug dosing must be safe and effective. Allometric…
Q: Which answer is correct (a or b) for the question about why your hands and feet get wrinkled after…
A: In a water molecule, two hydrogen atoms form single polar covalent bonds with an oxygen…
Q: Osmosis is water movement across a semipermeable membrane. Which of the following is true about…
A: Osmosis is the type of diffusion that involves the passive movement of water through the…
Q: The extracellular protein factor Decapentaplegic(Dpp) is critical for proper wing development in…
A:
Q: 2. (a) List the number of NADH and FADH2 equivalents formed and the corresponding number of ATP…
A: The fatty acid with 14 C is called myristic acid. Beta oxidation involves the sequential removal of…
Q: Give one condition for the denaturation step employed in blotting.
A: one condition for the denaturation step employed in blotting
Q: 1. For one mole of the fatty acid residue, determine the following:
A: The transferring of electrons from electrons Carrier and finally accepting by the Oxygen. The…
Q: 1. make your own version of Urea Cycle with diagram. 2. explain why these cycles are important for…
A: Ammonia is the end product of protein metabolism. The urea cycle helps to remove the ammonia formed…
Q: Which of the following is true about condensation reactions? A. Condensation reactions are…
A: Condensation reaction is the reaction of building complex molecule from small molecules with loss of…
Q: A polypeptide is digested with trypsin, and the resulting segments are sequenced:…
A: Introduction: A number of enzymes catalyze the breakdown of peptide bonds at a specific site in an…
Q: Find the connection among the words below and choose the letter of the word which is different. O A.…
A: Gel electrophoresis is the technique used to identify, quantify, and separate nucleic acids. It…
Q: Lipolysis involves the hydrolysis of glycolipids. O True False
A: Lipolysis is the metabolic process that hydrolyzes lipid triglycerides into glycerol and fatty…
Q: Which of the following is FALSE about photosynthesis? Group of answer choices: -Organisms that can…
A: Photosynthesis is a biochemical process by which green plants prepare carbohydrates. It evolves…
Q: All the reactions involved during gluconeogenesis occur in the cytosol except the one catalyzed by…
A: Including in non-photosynthetic living things, gluconeogenesis is a principal mechanism that results…
Q: Pyruvate carboxylase requires thiamine pyrophosphate as a coenzyme. True False
A: Coenzymes : Organic compounds required by the enzymes for their catalytic activity
Q: What is the pI, and how is it determined for amino acids that have noninionizable R groups?
A: pI is the isoelectric point at which no net charge on a molecule.
Q: The macromolecules that serve in the storage and transmission of genetic information are __________.…
A: Biomolecules are the organic compounds produced by living organisms. They are primarily made up of…
Q: 6. Explain briefly the importance of enzyme inhibition in biochemistry.
A: An enzyme is a protein that catalyzes biochemical reaction. It converts substrate into product. A…
Q: Which of the following amino acids has a hydrophobic R-group and would tend to be found on the…
A: Introduction: Amino acids are molecules that contain an amine group, carboxylic acid group, and a…
Step by step
Solved in 2 steps with 1 images

- Distinguishing the Mechanisms of Class I and Class I Aldolases Fructose bisphosphate aldolase in animal muscle is a class 1 aldolase, which forms a Schiff base intermediate between substrate (for example. fructose-1, 6-bisphosphate or dihydroxyacetone phosphate) and a lysine at the active site (see Figure I8.12). The chemical evidence for this intermediate conies from studies with aldolase and the reducing agent sodium borohydride, NaBH4. Incubation of the enzyme with dihydroxyacetone phosphate and NaBH4 inactivates the enzyme. Interestingly, no inactivation is observed if NabH4 is added to the enzyme in the absence of substrate. Write a mechanism that explains these observations and provides evidence for the formation of a Schiff base intermediate in the aldolase reaction.Choose the CORRECT sequence of glycine formation. transamination of 3-phosphoglycerate → hydrolysis of 3-phosphohydroxypyruvate → hydrolysis of 3-phosphoserine → transfer of the side-chain methylene group of cysteine to tetrahydrofolate → glycine transamination of 3-phosphohydroxypyruvate → oxidation of 3-phosphoglycerate → transfer of the side-chain amino group of serine to tetrahydrofolate → glycine oxidation of 3-phosphoglycerate → oxidation of 3-phosphohydroxypyruvate → hydrolysis of 3-phosphoserine → glycine transamination of 3-phosphohydroxypyruvate → hydrolysis of 3-phosphoserine → oxidation of 3-phosphoglycerate →transfer of the side-chain methylene group of serine to tetrahydrofolate → glycine oxidation of 3-phosphoglycerate → transamination of 3-phosphohydroxypyruvate → hydrolysis of 3-phosphoserine → transfer of the side-chain methylene group of serine to tetrahydrofolate → glycineDetermine if true or false 1. Isopentyl pyrophosphate (activated isoprene) undergoes cyclization during cholesterol biosynthesis 2. Malonyl CoA transacylase catalyzes the committed step in fatty acid biosynthesis. 3. Oxaloacetate carries acetyl CoA to the cytosol which is the site of fatty acid biosynthesis. 4. During fatty acid biosynthesis, propionyl CoA is the donor of the acetyl group. 5. During fatty acid biosynthesis, NADPH is used as the reducing equivalent in the process.
- The oxidation of glyceraldehyde 3-phosphate to 1,3-bisphosphoglycerate by glyceraldehyde 3-phosphate dehydrogenase has an unfavourable equilibrium constant (K'eq= 0.08; G′° = 6.3 kJ/mol), yet the flow at this point in the glycolytic pathway is smooth. How does the cell get out of the unfavourable equilibrium?Behenate (C22H44O2) can be obtained by chain elongation from palmitate. How many ATP equivalents are produced in the degradation of behenate to eleven acetyl-CoA?Consider docosanoic acid C12H43CO2H a. Label the alpha and beta Carbons. Show the beta-oxidation in an EXPANDED structure. b. Draw each acyl CoA derived from this fatty acid. c. How many acetyl Co A molecules are formed by complete beta-oxidation? d. How many cycles of beta-oxidation are needed for complete oxidation? e. How many molecules of ATP are formed from the complete catabolism of this fatty acid? Show the complete computation. f. How many moles of ATP per gram of fatty acid is formed from the complete catabolism of the given fatty acid? g. What is the molar mass of the given fatty acid? Solution: Show here the complete computations, [from a to e]
- The oxidation of glyceraldehyde 3-phosphate to 1,3-bisphosphoglycerate, catalyzed by glyceraldehyde 3- phosphate dehydrogenase, proceeds with an unfavorable equilibrium constant ( K'eq= 0.08; ΔG′° = 6.3 kJ/mol), yet the flow through this point in the glycolytic pathway proceeds smoothly. How does the cell overcome the unfavorable equilibrium?Select the following enzymes that utilize a mechanism where an enediol intermediate is formed. Check all that apply: a)phosphoglucoisomerase b)triose phosphate isomerase c)aldolase d) glyceraldehyde 3-P dehydrogenase e) hexokinaseIn the skeletal muscle, in anaerobic conditions, glyceraldehyde 3-phosphate is converted into pyruvate during the payoff phase of glycolysis; and this pyruvate is reduced into lactate during lactic fermentation. Part 1-Write the 11 balanced biochemical equations corresponding to all the reaction steps leading to the conversion of glyceraldehyde-3-phosphate into lactate through glycolysis followed by lactic fermentation. Part 2-Write the net equation of the whole transformation process (i.e. with glyceraldehyde-3-phosphate as the starting substrate; and lactate as the end product).
- Some bacteria catabolize glucose by the Entner–Doudoroff pathway, a variant of glycolysis in which glucose-6-phosphate is converted to 6 phosphogluconate (as in the pentose phosphate pathway) and then to 2 keto-3-deoxy-6-phosphogluconate (KDPG). Next, an aldolase acts on KDPG. (a) Draw the structures of the products of the KDPG aldolase reaction. (b) Describe how these reaction products are further metabolized by glycolytic enzymes. (c) What is the ATP yield when glucose is metabolized to pyruvate by the Entner–Doudoroff pathway? How does this compare to the ATP yield of glycolysis?Phosphoenolpyruvate carboxykinase (PEPCK, figure below)catalyzes first committed (rate-limiting) step in hepatic gluconeogenesis, namely the reversible decarboxylation of oxaloacetate to phosphoenolpyruvate (PEP) and carbon dioxide. This reaction can occur using either ATP or GTP as a source of the phosphate.Which following mutation can potentially improve the binding affinity of the substrate for this enzyme? a. LYS254ARG b. LYS254ASP c. ARG333GLU d. THR255GLYGalactose 1-phosphate: UDP-glucose uridyltransferase (GALT) is an enzyme important for the catabolism of galactose. Patients with GALT deficiency present within days of birth with feeding difficulty, fatigue, jaundice and liver problems, and failure to thrive. In addition, a buildup of galactose and its metabolites are present in the bloodstream. GALT catalyzes the following reaction in galactose catabolism. NOTE that GALT is reversible!! Classify this enzyme and explain why you classified it in this way.

