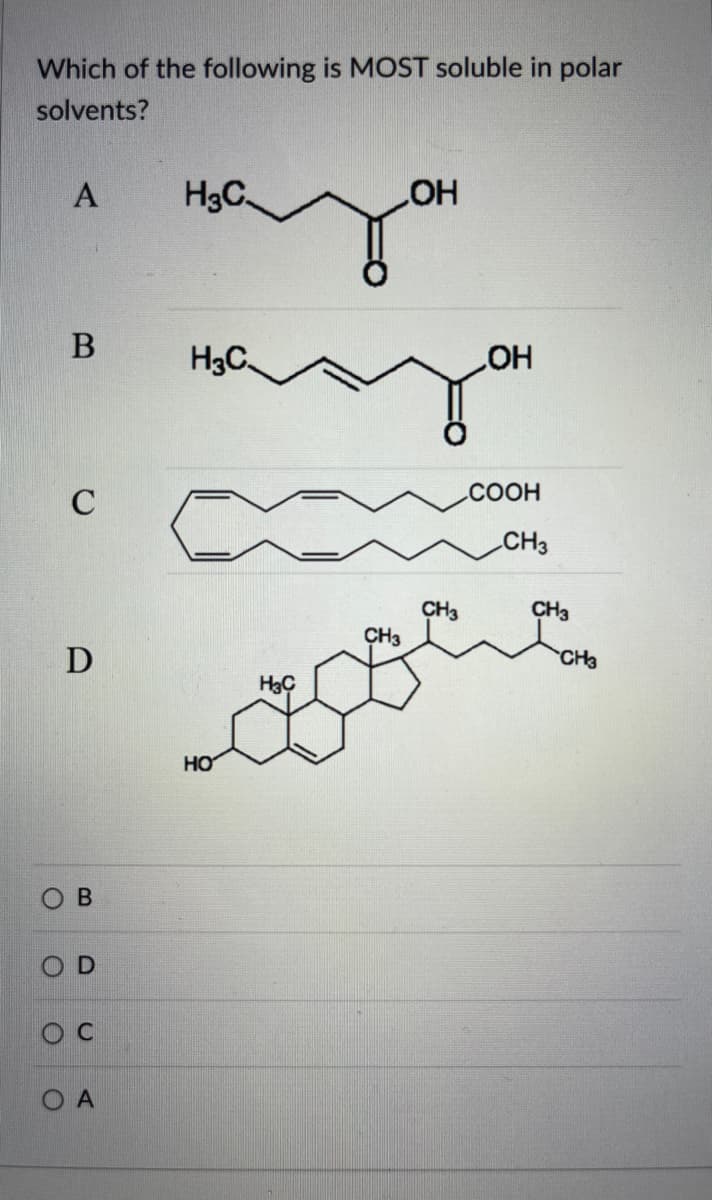
Which of the following is MOST soluble in polar solvents? A H3C LOH В с D В OD Oc О А H3C. HO л HC CH3 CH3 ОН COOH CH3 CH3 CH3
Q: Determine the class of lipid (see attached image for choices) shown by the following structures and…
A: Lipids are a class of biomolecules that are nonpolar in nature. These are hydrophobic in nature and…
Q: Consider the complete oxidation of one mole of simple TAG containing lignoceric acid residues…
A: During the digestion of a triglyceride the fatty acids that are released are broken down in a series…
Q: What happens to a person's body when they consume dairy products when they are lactose intolerant?…
A: Introduction: Lactose is a disaccharide containing β-D-galactose and β-D-glucose in β (1,4) linkage.…
Q: HO-CH CH II CH Q-CH2CH2N(CH3)3 --=0 동_들 2-0- -CH-CH2 C=0
A: The given structure has a polar head group, and two hydrophobic groups present in it. It means the…
Q: KM is determined by measuring the reaction velocity of two enzymes (X and Y) at different…
A: Enzymes are usually protein molecules which catalyzes a biochemical reaction by decreasing its…
Q: Compare and identify condensation (dehydration) and hydrolysis reactions
A: There are various types of reactions that occur inside the cell such as hydrolysis, dehydration,…
Q: Discuss briefly but concisely the importance of the following metabolic pathway. H.B-oxidation of…
A: Introduction: Fatty acids from the diet or through degradation of triglycerides stored in the…
Q: Identify what type of micropipette should be used to accurately transfer the volume in the ITEM 10…
A: micropipeptes are used to dispense liquids in microliters volumes.
Q: Which of the following is not involved in cyclic cyclic photophosphorylation? cytochrome bf complex…
A: Cyclic cyclic photo phosphorylation occurs in thylakoid membrane where the conversion of ADP to ATP…
Q: D.3 Loading Samples and Running the Gel Use special gel loading tips or a micro-syringe to load…
A: Generally when performing western blotting the volume loaded in the well is 20 uL. Since your…
Q: Prepare a 75 ml of a 10% (w/v) FeCl3 solution. How many grams of FeCl3 must be weighed out and…
A: Given Values: Volume of FeCl3 solution = 75 ml Concentration of the FeCl3 = 10% (w/v)
Q: What TYPE of inhibition is observed in the following plot: 1/v₂ Intersect at Y axis 1/Km Competitive…
A: Enzymes can be inhibited , when the substrate and inhibitor compete for the binding to the same…
Q: raw a schematic diagram on glycogenolysis by showing the structural formula of its substrate. Show…
A: Glycogenolysis is the process in which stored glycogen is broken down into glucose.
Q: 8. Which of the following is/are TRUE about bioassay? 1. It is a dose-response type of research. II.…
A: As a result of comparing a substance's effect on a test organism with a standard preparation, a…
Q: what is the regulatory step during cholesterol biosynthesis?
A: Cholesterol is found in both membranes and lipoproteins. Cholesterol synthesis is an…
Q: Statement A hexose sugar Insoluble in water Formed by condensation reactions Contain the elements,…
A: The biological macromolecules nucleic acids, proteins carbohydrates and lipids form the building…
Q: QUESTION 2 The following carbohydrate tests have been performed on a sample containing an unknown…
A: Sugars are tasted sweet in taste. Simple sugars are classified as monosaccharides. These substances…
Q: QUESTION 2 ADP ATP AG"= 1.75 kJ/mol AG"=-18.9 kJ/mol 1,3-Bisphosphoglycerate → 3-Phosphoglycerate…
A: Free energy change for biochemical reaction is given by ΔG°' = -R T ln Keq , where Keq is the…
Q: Identify (1) the group where the lipid belongs, and (2) determine whether the lipid is saponifiable…
A: A lipid is a material that is water insoluble but soluble in solvents like alcohol, and chloroform.…
Q: "Using the concepts and techniques you have learned from the two labster simulations, provide…
A: Monogenic disorders are those disorders that occur due to a single non-functional gene in our body.…
Q: D arrangement of localized regions of proteins A. PRIMARY B. SECONDARY
A: The three-dimensional arrangements of atoms in an amino acid chain, that folded up into specific…
Q: What are the applications of human genome into health, physiology, and biochemistry?
A: The Human Genome Project is a large-scale scientific project aimed at decoding the chemical makeup…
Q: Which of the following is the common carbohydrate in all types of gangliosides? O D-galactose…
A: Glycolipids are lipid molecules containing carbohydrate residues linked to a hydrophobic lipid…
Q: Which of the following are true regarding parasites in the phylum Apicomplexa? a) they are…
A: Apicomplexan parasites are the large phylum of intracellular parasites characterized by the apical…
Q: The successful sequencing of the human genome The human genome holds an extraordinary amount of…
A: The Human Genome project or HGP is a huge project which was aimed to map and sequence the entire…
Q: Which of the following statements is TRUE about cholesterol biosynthesis? Group of answer choices…
A: Cholesterol biosynthesis pathway involves the conversion of the Acetyl CoA to the Acetoacetyl CoA…
Q: Explain how osmosis works including the concept of concentration gradient
A: Osmosis is a vital process in many biological systems. For example, it is responsible for the…
Q: 3. How is cytochrome c related to apoptosis?
A: In multicellular organisms programmed cell death is refer to as apoptosis. It is a method by which…
Q: Which of the following amino acids undergoes phosphorylation? Histidine Glutamic Acid…
A: Post-translational modification of proteins refers to the covalent enzymatic modification of…
Q: Triglycerides are comprised of glycerol plus ____ fatty acids, and are ____ compared to fatty…
A: Fatty acids are lipids composed of long-chain hydrocarbons ending by a carboxylic acid functional…
Q: All the reactions involved during gluconeogenesis occur in the cytosol except the one catalyzed by…
A: Gluconeogenesis is the process of synthesis of glucose from non Carbohydrate sources like aminoacids…
Q: 22. The blood group by the MN system depends on the combi- nation of the codominant genes CM and CN.…
A: For the blood grouping by MN system, According to hardy Weinberg equation, a gene that exists as two…
Q: A child receiving adequate nutrition and vitamin D, shows signs of rickets. The blood calcium…
A: Calcium are minerals that are essential for human health. They are involved in many physiological…
Q: deduce the DNA sequence based on the following electrophoreograms.
A: The electrophoretic technique is used to separate proteins, DNA, and RNA based on their size and…
Q: QUESTION 2 CH3COO-C₂H5 + H₂O CH3COOH + C₂H5OH Ethyl acetate Acetate Ethanol Ethyl acetate, acetate…
A: Ethyl acetate, acetate and ethanol were added to water solution in concentrations of 0.3M and left…
Q: A class of animals called ruminant animals that includes grassers (e.g., cows, zebras, giraffes,…
A: Introduction: An adaptation is something that is concerned with the structure or behavior of an…
Q: Which of the following statements is FALSE? Group of answer choices: - Plastoquinone has a higher…
A: Photosynthesis is the process by which plants utilise light energy from the sun to prepare food and…
Q: what will happen if there is an accumulation of acetyl CoA in the liver mitochondria?
A: Acetyl coenzyme A is a thioester between the acyl group carrier, acetic acid, and a thiol that acts…
Q: HO HO [Select] terpene triacylglycerol steroid glycolipid ОН но HN НО
A: There are different types of lipids known as phosphoglycerolipids, sphingolipids,…
Q: xplain briefly but concisely 3. Mammalian liver can carry out gluconeogenesis using oxaloacetate as…
A: Gluconeogenesisis the process that requires energy. It is not a simply the reversal of process of…
Q: A researcher has measured the initial rate of an enzyme-catalyzed reaction as a function of…
A: Km and Vmax are important features of an enzyme thatvcan determine the efficiency of the enzyme.…
Q: Η O ΝΗΣ NH NH ΝΗΣ HO NH
A: Cation exchange chromatography is a method of separation of biomolecules (proteins/peptides) based…
Q: Make a mind map for cellular respiration
A: Cellular respiration is a process which involve metabolic reactions taking place in cells. These…
Q: Calculate the unknown concentration of the PROTEIN C with an absorbance value of A412 given the…
A: Protein concentration can be determined with the help of spectroscopy. Spectrophotometer is an…
Q: Pyruvate carboxylase requires thiamine pyrophosphate as a coenzyme. True False
A: Coenzymes : Organic compounds required by the enzymes for their catalytic activity
Q: Using the free energy values of glycolysis and pyruvate transformation to ethanol calculate free…
A: To find the standard free energy value (∆G'0) of a reaction that is the sum of 2 independent…
Q: An experiment was carried out to measure the reaction rate of hydrolysis of acetylcholme (substrate)…
A: We need to draw the Lineweaver Burk (LB) plot to find out the answers. LB plot has 1/[S] as X-axis…
Q: how the function of two or three different lipids is connected to their structure. be meaningful; ●…
A: Introduction: Macromolecules are large molecules within the cells. It is made from thousands of…
Q: The Km of liver glucokinase is _____ than typical blood glucose concentrations. At typical blood…
A: Glucokinase, gluco is glucose and kinase is an enzyme which is known to phosphorylate a substrate.…
Q: Please state if the statements are true or false. 1. A pyranose is a sugar in Haworth projection…
A: Pyranoses are sugar molecules having six members. Cytosolic pyruvate kinase catalyzes…
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

- A cation has a(n) ________ charge.a. neutralb. positivec. alternatingd. negativeThe turn tonicity Refers to the strength of solutions. True or false?You take a bottle of soft drink out of your refrigerator. Thecontents are liquid and stay liquid, even when you shakethem. Presently, you remove the cap and the liquid freezessolid. Offer a possible explanation for this observation.
- Rx Ephedrine sulfate (fz. pt = -0.13°C). 2%Sodium chloride MW 58.5Purified water qs ad. 30 mL How much sodium chloride should be used to make this eye solution isotonic with tears? the answer is 224mgA patient needs 1mg of a medication. The Australian Injectables Drug Handbook states that each gram of this medication is to be mixed with 10mL of water over 3-5 minutes via IV bolus. After preparing the medication, how many mL per minute would you administer if you do it over 3 minutes? Please do it in dimensional analysis or something please show work. I'm confusedYour supervisor gave you a 5% saline (NaCl) solution. You need to make 100ml of 1.0% saline solution. How much 5% saline would you take to complete the task?
- The UV-Vis Spectroscopy technique is based on: A. Rotational excitation B. Nuclear excitation C. Electronic excitation D. Vibrational excitation E. None of the above. Which of the following is not a deprotonated substance: a) KOH b) LiOH c) HA d) BA 1–fluid ounce (29.573 mL) solution contains 4.5 grains (291.60 mg) of silver nitrate. How much sodium nitrate must be added to this solution to make it isotonic with nasal fluid?
- Which of the following positively-charged ions is about 10,000 times more concentrated outside the cell than inside (useful in muscle contraction, and in signal transduction of neurons)? bicarbonate chloride sodium calcium E. magnesiumHow many milligrams of sodium chloride and lincomycin are required to prepare 100 mL of a 1% lincomycin isotonic with the blood? Please do it soonGuanosine (C10H13N5O5) in solution has a maximum absorbance at a wavelength of 275 nm. The molar extinction co-efficient at this wavelength is 84M−1cm−1and the path length is 24.7 cm. Through the use of a spectrophotometer, it is found that the that A275= 1.48. What is the concentration of the guanosine solution in grams/litre? Molecular weights (g/mol): C-12, H-1, N-14, O-16 Select one: A. 0.201869 g/L B. 0.435188 g/L C. 0.059919 g/L D. 0.294046 g/L E. 0.000713 g/L









