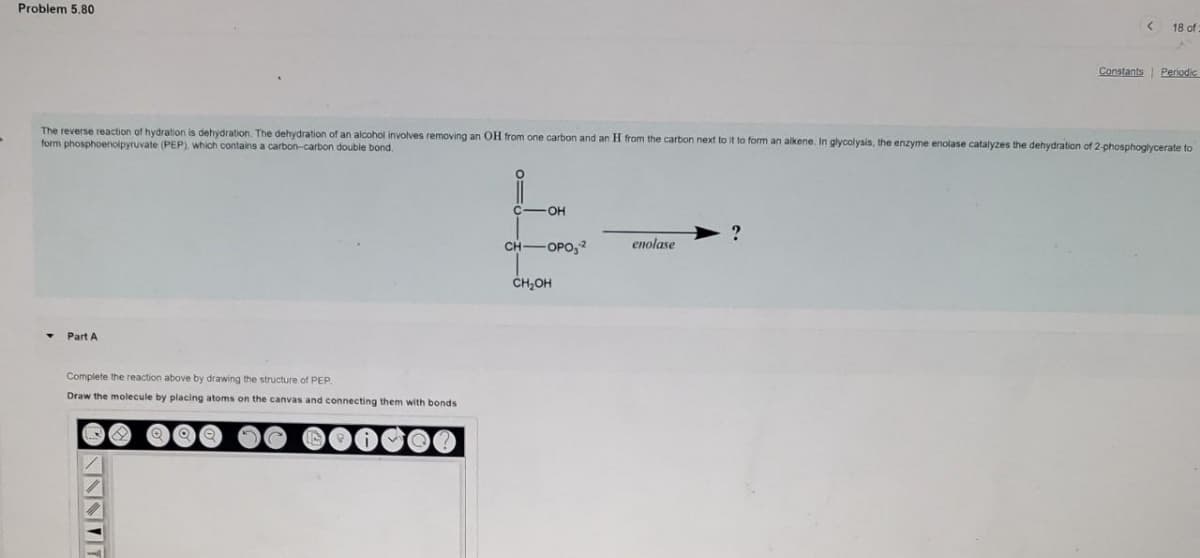
Complete the reaction above by drawing the structure of PEP. Draw the molecule by placing atoms on the canvas and connecting
Q: Metabolic pathway characteristics: a. Give one example of an amphibolic pathway. b. What…
A: Metabolic pathways are a series of process which includes chemical reactions occurring in a cell.…
Q: a series of tubes contains 1 mL of diiluent. 0.5 mL of serum is added to tube 1 and then mixed. 0.2…
A: Recall that : Equation of dilution: M1V1 = M2V2 where: M1 is the molar concentration of the stock…
Q: Why are single strand binding proteins (ssb proteins) needed in DNA replication? (note that you need…
A: DNA consists of two strands of polynucleotide that are linked to each other via hydrogen bonding…
Q: In the peptide, SWEETLY, bond via its side chain. (Give the full name of the amino acids) can still…
A: Recall that: for a given peptide amino acid sequences are written with N-terminal amino acid on the…
Q: The active site of chymotrypsin indicates that the following residues are needed: a. Asp and…
A: Chymotrypsin is a digestive enzyme that helps to digest proteins in our food. Chymotrypsin cleaves…
Q: What do x-helices and B-sheets have in common?
A: Alpha helix and beta sheets are two types of secondary structure present in the protein structure.…
Q: AMP is an activator of fructose 1,6-bisphosphatase (FBPase-1) True False
A: Glycolysis is the process by which one molecule of 6 carbon glucose is broken down into 2 molecules…
Q: Elaborate on the industrial processes of water purification and wastewater treatment. In your…
A: Introduction Water pollution is process by which water get polluted for various human activities.…
Q: Use the data below to answer the following question: How much more energy is stored in a gram of fat…
A: A heterogeneous class of substances with comparatively similar physical characteristics are referred…
Q: 6.) Calculate the concentration (N) of arsenic acid (H3A504) in a solution if 25mL of that solution…
A: Given that, Volume of 0.1894 molar KOH required to neutralize anseric acid is 35.21 mL. Volume of…
Q: Question 9 Which of the following fatty acid does have greater solubility in wat O 16:0 O 18:0 O…
A: Fatty acids are a class of lipids that are nonpolar and are insoluble in water. The naturally…
Q: Identify the components of animal fatty acid synthase (FAS). acyl-CoA dehydrogenase B-ketoacyl…
A: Enzymes are highly specialized proteins that have extraordinary catalytic power, greater than that…
Q: 5. Oxaloacetate, glycerol, and lactate can be used in gluconeogenesis. T F
A: Sometimes organisms need a method to synthesize glucose from non-carbohydrates precursors. The…
Q: More energy comes out of glucose degradation if pyruvate proceeds to the mitochondria for oxidative…
A: Degradation of glucose is termed as glycolysis which is a catabolic pathway in which 6 carbon…
Q: How can a noncompetitive inhibitor affect enzyme activity without binding to the active site? a. It…
A: The substance that decreases the activity of enzyme is called enzyme inhibitor. There are two types:…
Q: of the gluconeogenesis wing is not true? A. Phosphofructokinase-1 (PFK-1) and…
A: Glycolysis - is a process in which one mole of glucose is partially oxidized into two moles of…
Q: DNA: Explain the Meselson-Stahl Experiment.
A: The mode of replication in DNA, in which a parental duplex DNA gives rise to two identical daughter…
Q: This question is about "ANSERINE" not just "SERINE
A: Anserine is a dipeptide composed of beta alanine and methyl histidine. It is found in brain of birds…
Q: You measure Protein A and Protein B to have a Kd of 10-8 M. At what concentration of Protein B will…
A: Here we have 2 proteins, 'A' and 'B' . The reversible equilibrium between these 2 proteins is shown…
Q: ĭ HO O-C-R' O=O=O=O -O-C-R" -O-C-R"" A -P=00=0 HII -N-C-R" OH E O-sugar O || -0-C-R" -NH3+ -(CH₂)12…
A: Lipids are a chemically diverse group of biomolecules that have two things in common: low…
Q: The active site _______________. a. is the compound that an enzyme reacts with during the chemical…
A: The active site is that region of an enzyme where substrate molecules bind. The binding of substrate…
Q: Suppose that the thermal denaturation experiment on the protein with the Met to Lys mutation took…
A: pKa is the pH at which a weak acid is 50% dissociated. Changes in pH affect the ionisation of…
Q: Explain the cell response to FED, and then FASTED states, by glucose transporter (GLUT)-4. (answer…
A: Carbohydrate from food is converted to glucose by digestive enzymes which then enters the…
Q: What is the difference between starch and cellulose that makes one digestible and the other not…
A: Chemically carbohydrates are polyhydroxy aldehydes or ketones. They have the general formula :…
Q: The side chain of asparagine contains: OA) a hydroxyl group OB) an amine group OC) a carboxyl group…
A: Proteins are composed of amino acids, which are bound together by peptide linkage. Amino acids…
Q: Discuss the factors affecting enzyme activity
A: Enzymes are high molecular weight protein molecules that catalyse biochemical reactions. The…
Q: what element is this? after MDA reacts with something Ph CH3 N CH3
A: MDA is malondialdehyde . It is a product of lipid peroxidation in corals. Determination of this MDA…
Q: 2. Biuret Test Color of 3% CuSO4 solution: Sample casein alanine albumin gelatin distilled water 3.…
A: Proteins are large molecules made up of amino acid residues linked via a peptide bond. Amino Acids…
Q: What charged groups are present in leucine at a pH = 7? OA) 1× NH3¹ B) 1 x COO OC) 1× NH3¹ and 1 ×…
A: Amino acids have a central carbon atom known as α-carbon atom. Each amino acid has an amino group,…
Q: Give the isoelectric points of following 2 tripeptides DRI and RID.
A: pI (isoelectric point) of an amino acid is the PH at which the amino acid carries zero net charge. A…
Q: Calculate the net charge on the following tripeptides at pH 5.0: (a) Leu-His-Asp [0] (b)…
A: Recall that: Amino acid sequences are written with N-terminal amino acid on the left and C-terminal…
Q: How is aquaporin synthesised? List step by step. How is this protein made and targeted to its final…
A: Aquaporins (AQP) are protein channels that function in the transfer of water at very high rates…
Q: HO 0 0 NH₂ НО.
A: The fundamental building blocks of proteins are called amino acids, which are made up of one…
Q: Choose the best answer The active site of an enzyme is the place where the following happens:…
A: A catalyst is a component that speeds up a chemical reaction without really being a reactant.…
Q: What are the roles of sigma factor, rho factor, and an intrinsic terminator in transcription in…
A: Introduction: Transcription is the synthesis of single-stranded messenger RNA (mRNA) from the…
Q: Why carbon C-2 and C-5 are mark? why not just ona carbon is marked since just one carbon was marked…
A: Gluconeogenesis is the synthesis of glucose molecules from non-carbohydrate sources such as…
Q: 4. A single base addition and a single base deletion approximately 15 bases apart in the mRNA…
A: As per the central dogma of molecular biology, the genetic information stored in the DNA is copied…
Q: d) Alcohols usually don't behave as a weak acids, but tyrosine does. Explain why Y ionizes, while S…
A: Note: Hi! Thank you for the question. We are authorized to answer one question at a time. Since you…
Q: 12. The graph shows values of arterial plasma bicarbonate concentration and pH for different persons…
A: Metabolic alkalosis is caused by acid base imbalance in the blood. It is characterised by increase…
Q: If a dialysis tube that is permeable to water but not sucrose contains a 40% sucrose solution that…
A: Water travels across a semi-permeable membrane from the side of the membrane with a lower…
Q: . What is the effect of boiling in the test tube with starch solution and saliva?
A: “Since you have asked multiple questions, we will solve the first question for you. If youwant any…
Q: 15. Cellular proteins are oftentimes post-translationally modified. Choose one of the following…
A: Post translational modification are the modifications done in the protein by covalently attaching…
Q: HN 2 H₂N-C C 6 3 N C 5 C following nitrogenous bases is represented by this structure? 7 ZI 8 CH
A: The nucleic acids are constituted of nucleotides. The nucleotides consist of a base (adenine,…
Q: The following are true of the mitochondrial structure I. The inner mitochondrial membrane is…
A: Mitochondria are power house of cell, known to synthesize ATP. They are membrane bound organelles…
Q: (a) (b) (c) The figure shown below is the mechanism of a DNA nuclease. The DNA molecule is in blue…
A: The given figure shows the mechanism of a DNA nuclease. Nucleases breaks the phosphodiester bonds of…
Q: II. Questions 1. What is the positive result to the following tests? a. Ninhydrin Test: b. Biuret's…
A: Ninhydrin test : The ninhydrin test is a chemical test which is used to check whether a given…
Q: The brown discoloration of apples and potatoes is due to an enzyme catalyzed chemical reaction.…
A: Cut apples and potatoes turn brown or discolored on exposure to air. This is due to conversion of a…
Q: Compare and contrast the principle behind DNA migration in agarose gel electrophoresis from that of…
A: Electrophoresis is a technique that involves the migration of charged molecules under the influence…
Q: Which one of the following statements about PEP (phosphoenolpyruvate) synthesis is correct? Pyruvate…
A: PEP, also known as 2-phosphoenolpyruvate, is a crucial biochemical intermediary. It participates in…
Q: All enzymes have an optimal temperature and pH environment. Choose how the following changes might…
A: Enzymes are high molecular-weight protein molecules that catalyse biochemical reactions. The…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- Problem 1: For your new year's resolution, you resolve to lose 5lbs of body fat through physical exercise. If you work out hard, you can generate 200 W of mechanical power and be 20% efficient at it. How long will you have to exercise at this rate to burn the desired amount of fat? If you chose to diet instead and cut your food intake of 2400 kcal/day to 1600 kcal/day, how long will it take you to lose the desired amount of weight?Problem 2. When alcohol is consumed in excessive quantities, the resulting levels of NADH cause metabolic abnormalities, and one of which is high levels of fatty acid synthesis. Fatty acid synthesis, also a cytoplasmic process, uses acetyl-CoA as a substrate and NADPH as a reducing agent. Based on the above, we can see how acetate is converted to acetyl-CoA in the mitochondria, but fatty acid synthesis takes place in the cytosol. Complete the analysis by accounting for high acetyl-CoA concentration in the cytosol. Study the summary of reactions of citrate metabolism. E. How would increasing the rate of the reaction catalyzed by malate dehydrogenase decrease the concentration of oxaloacetic acid (OAA) in the cytosol? F. How would decreasing the concentration of OAA increase the concentration of acetyl-CoA in the cytosol by Le Chatelier’s principle?Problem 3. Excess alcohol consumption can result in fatty liver disease. Liver cells synthesize triacylglycerides from fatty acids formed by fatty acid synthesis. Excess triacylglycerides can accumulate in the liver compromising normal liver function. D.Explain how stimulation of the rate of fatty acid synthesis changes the rate of fatty acid oxidation based on allosteric regulation of the rate-limiting step of beta-oxidation.
- Problem #1: Indicate the total number of ATPs produced in each of the following oxidation reactions from 5 moles of glucose. A. pyruvate to Acetyl CoA B. glucose to Acetyl CoA C. Citric acid cycleProblems 14 and 15: some of the exponents are unclear. Here they are: 14. Calculate vi and the degree of inhibition caused by a competitive inhibitor under the following conditions:(a) [S]=2x10-3 Mand[I]=2x10-3 M(b) [S]=4x10-4 Mand[I]=2x10-3 M (c) [S]=7.5x10-3 Mand[I]=10-5 MAssume that Km = 2 x 10-3 M, Ki = 1.5 x 10-4 M and Vmax = 270 nmoles x liter-1 x min-1.The degree of inhibition is the percent of the uninhibited velocity reached in the presence of the inhibitor. 15. (a) What concentration of competitive inhibitor is required to yield 75% inhibition at a substrate concentration of 1.5 x 10-3 M if Km =2.9x10-4 M and Ki =2x10-5 M? (b)Towhatconcentration must the substrate be increased to reestablish the velocity at theoriginal uninhibited value?Task №1. The disease is described in which the activity of phosphofructokinase of adipose tissue isn't regulated by citrate. How can the lipid metabolism in adipose tissue be changed in such genetic defect?
- Problem 3. Excess alcohol consumption can result in fatty liver disease. Liver cells synthesize triacylglycerides from fatty acids formed by fatty acid synthesis. Excess triacylglycerides can accumulate in the liver compromising normal liver function. A.Explain how palmitoyl-CoA is related to fatty acyl CoASH esters (acyl-CoA) B.Explain how increasing the rate of triglyceride formation changes the concentration of acyl-CoA in the cytosol. C.Explain how the rate of triglyceride formation stimulates the rate of fatty acid synthesis based on the concentration of palmitoyl-CoA in the cytosolPROBLEM 3-1 Sentence InterpretationWhen scientists submit papers for publication, they often dread the re-sponse of reviewers. Here are four sentences that could have been writ-ten in different structural arrangements by reviewers to deliver the same news. Which statement is the one most likely resulting in the paper being accepted, and which is most likely the one resulting in rejection? Explain why.1. Overall, although this manuscript is of interest for structural biologists, a more detailed analysis of ABC should be provided. 2. Although a more detailed analysis of ABC should be provided, this manuscript is of interest for structural biologists.3. This manuscript is of interest for structural biologists, but a more detailed analysis of ABC should be provided.4. A more detailed analysis of ABC should be provided, but overall, this manuscript is of interest for structural biologists. PROBLEM 3-2 Word Placement and Flow Rewrite the following paragraph. Place words such that the reader…Task 1. How many ATP is produced in entire oxidation of 1 acetoacetate molecule? Will you get more or less ATPs oxidizing β-hydroxybutyrate?
- Problem # 3: Calculate the number of moles of ATP produced from the complete oxidation of 900 g glucose in the cells. Show your computation/solution.TASK 1: Prepare a 0.020 M Cupric Sulfate Solution.Capacity of volumetric flask provided: 50 mLCalculation of mass required: *(MM = 249.68 g/mol) Show how you will prepare this. TASK 2: Preparation of a Dilutions of 0.020 M Cupric SulfateDilution 1 - Prepare a 1:2 dilution of 0.020 M Cupric Sulfate for a final volume of 200 mL.How much stock is required to make this solution?Exercise 5-9 Phenylalanine deaminase Phenylalanine deaminase positive bacteria can remove the amine functional group from the amino acid and the amine group is released as ammonia waste. Phenylpyruvic acid is also produced by this reaction. What can phenylalanine deaminase positive bacteria do with phenylpyruvic acid?



