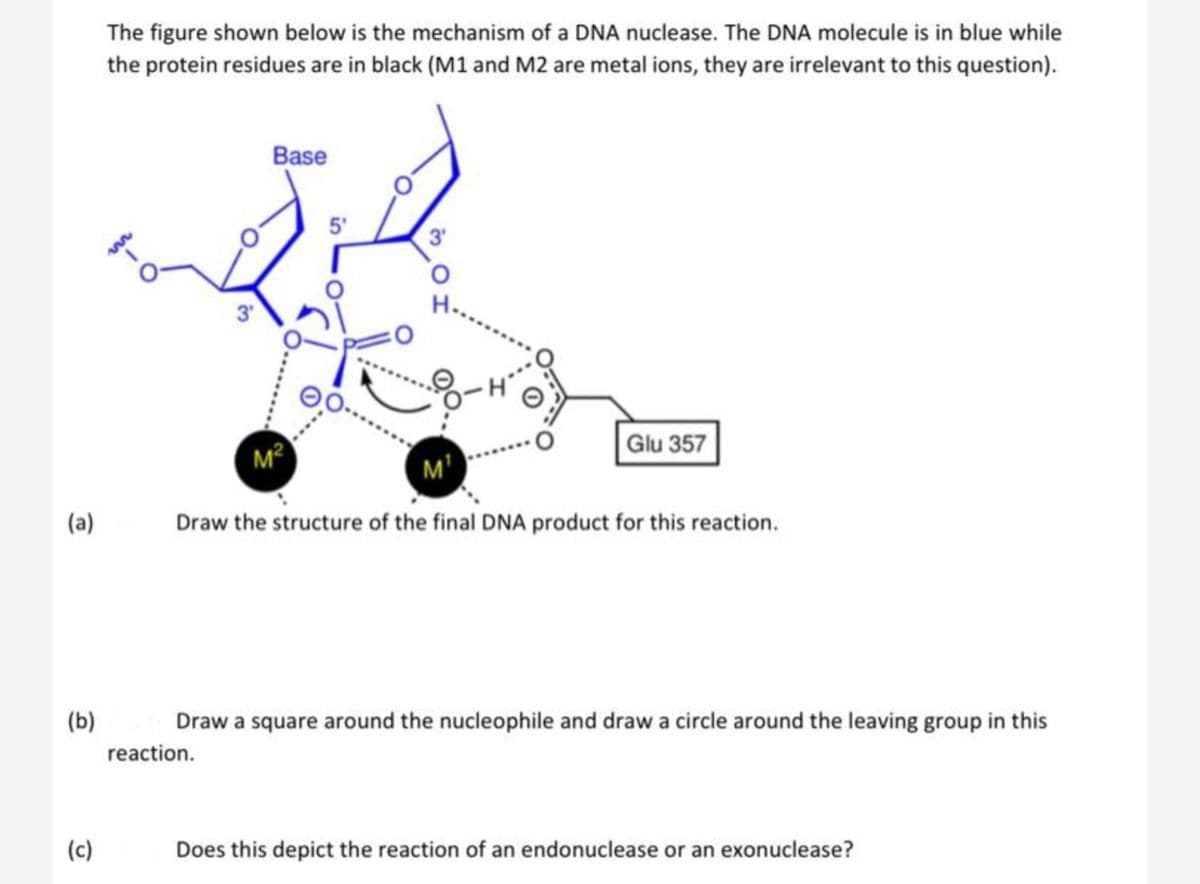
(a) (b) (c) The figure shown below is the mechanism of a DNA nuclease. The DNA molecule is in blue while the protein residues are in black (M1 and M2 are metal ions, they are irrelevant to this question). 3 Base reaction. M² 3' Glu 357 M¹ Draw the structure of the final DNA product for this reaction. Draw a square around the nucleophile and draw a circle around the leaving group in this Does this depict the reaction of an endonuclease or an exonuclease?
(a) (b) (c) The figure shown below is the mechanism of a DNA nuclease. The DNA molecule is in blue while the protein residues are in black (M1 and M2 are metal ions, they are irrelevant to this question). 3 Base reaction. M² 3' Glu 357 M¹ Draw the structure of the final DNA product for this reaction. Draw a square around the nucleophile and draw a circle around the leaving group in this Does this depict the reaction of an endonuclease or an exonuclease?
Human Heredity: Principles and Issues (MindTap Course List)
11th Edition
ISBN:9781305251052
Author:Michael Cummings
Publisher:Michael Cummings
Chapter8: The Structure, Replication, And Chromosomal Organization Of Dna
Section: Chapter Questions
Problem 12QP: DNA contains many hydrogen bonds. Are hydrogen bonds stronger or weaker than covalent bonds? What...
Related questions
Question
Transcribed Image Text:(a)
(b)
(c)
The figure shown below is the mechanism of a DNA nuclease. The DNA molecule is in blue while
the protein residues are in black (M1 and M2 are metal ions, they are irrelevant to this question).
3'
Base
reaction.
M²
5'
00
3'
Glu 357
M¹
Draw the structure of the final DNA product for this reaction.
Draw a square around the nucleophile and draw a circle around the leaving group in this
Does this depict the reaction of an endonuclease or an exonuclease?
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you

Human Heredity: Principles and Issues (MindTap Co…
Biology
ISBN:
9781305251052
Author:
Michael Cummings
Publisher:
Cengage Learning

Human Heredity: Principles and Issues (MindTap Co…
Biology
ISBN:
9781305251052
Author:
Michael Cummings
Publisher:
Cengage Learning