Concentration of ATP: 5.0*10-3 Concentration of ADP: 5.0*10-4 Concentration of Phosphate (Pi): 1.0*10-3 ∆G ̊= -31 kJ/mol at 298 K for the hydrolysis of ATP. Calculate the Q and ∆G for ATP hydrolysis.
Concentration of ATP: 5.0*10-3 Concentration of ADP: 5.0*10-4 Concentration of Phosphate (Pi): 1.0*10-3 ∆G ̊= -31 kJ/mol at 298 K for the hydrolysis of ATP. Calculate the Q and ∆G for ATP hydrolysis.
Chapter29: The Organic Chemistry Of Metabolic Pathways
Section29.SE: Something Extra
Problem 36AP
Related questions
Question
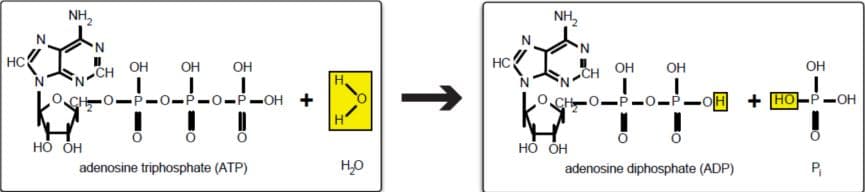
Concentration of ATP: 5.0*10-3
Concentration of ADP: 5.0*10-4
Concentration of Phosphate (Pi): 1.0*10-3
∆G ̊= -31 kJ/mol at 298 K for the hydrolysis of ATP.
Calculate the Q and ∆G for ATP hydrolysis.
Transcribed Image Text:NH,
NH,
HC
OH
OH
HC
он
OH
OH
OH
-CH
CH
-P-OH +
-P-0-
H + HO-P -OH
но он
но он
adenosine triphosphate (ATP)
H,0
adenosine diphosphate (ADP)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Recommended textbooks for you


General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:
9781305081079
Author:
STOKER, H. Stephen (howard Stephen)
Publisher:
Cengage Learning,


General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:
9781305081079
Author:
STOKER, H. Stephen (howard Stephen)
Publisher:
Cengage Learning,

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning


Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning