Consider a Newman projection of 2-methylbutane shown. Calculate the energy from the strain present in this conformation. CH3 CH3 kcal/mol H. 3 CH3 5. 6. wn Ener rteracton Type he CH he CC, ed CHH ed CHCH 7 +/- x 100 1, 4.
Consider a Newman projection of 2-methylbutane shown. Calculate the energy from the strain present in this conformation. CH3 CH3 kcal/mol H. 3 CH3 5. 6. wn Ener rteracton Type he CH he CC, ed CHH ed CHCH 7 +/- x 100 1, 4.
Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Andrei Straumanis
Chapter6: Alkanes & Alkenes
Section: Chapter Questions
Problem 1E
Related questions
Question
100%
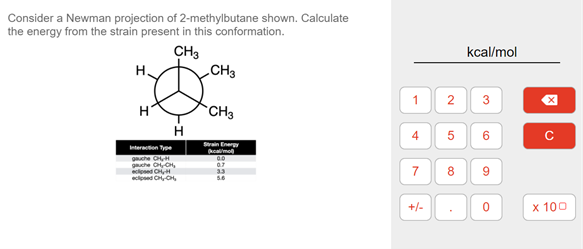
Transcribed Image Text:Consider a Newman projection of 2-methylbutane shown. Calculate
the energy from the strain present in this conformation.
CH3
CH3
kcal/mol
H.
1
H
CH3
6
Strain Enery
kcalmo
Interaction Type
guche H
guche CCH,
cosed CH,
cioned C C,
07
7
8
9
33
+/-
x 100
3.
2.
4.
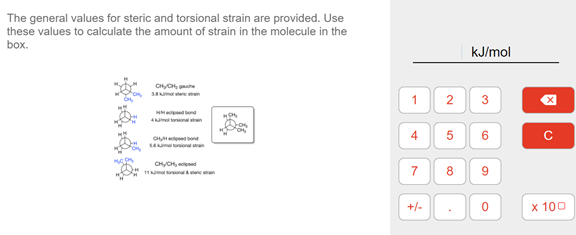
Transcribed Image Text:The general values for steric and torsional strain are provided. Use
these values to calculate the amount of strain in the molecule in the
box.
kJ/mol
1
3
H ond
4m toronan
6
C ond
SAma
CHyCH, d
11ma n
7
8
9.
+/-
х 100
2.
5.
4.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

Recommended textbooks for you

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning
