Consider the molecule and its rotational energy diagram. Note that the tert-butyl (tBu) group s the largest group. Br Molecule for Q14 Rotational Coordinate a. Complete the Newman projection to show the molecule in its lowest energy conformation (D). F b. Complete the Newman projection to show the molecule in its highest energy conformation (A). F c. Which group is eclipsed with the tBu group at position E in the energy diagram? d. What is the dihedral angle for the Br-C-C plane and the tBu-C-C plane at position F in the energy diagram?
Consider the molecule and its rotational energy diagram. Note that the tert-butyl (tBu) group s the largest group. Br Molecule for Q14 Rotational Coordinate a. Complete the Newman projection to show the molecule in its lowest energy conformation (D). F b. Complete the Newman projection to show the molecule in its highest energy conformation (A). F c. Which group is eclipsed with the tBu group at position E in the energy diagram? d. What is the dihedral angle for the Br-C-C plane and the tBu-C-C plane at position F in the energy diagram?
Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Andrei Straumanis
Chapter17: Conjugation And Molecular Orbital (mo) Theory
Section: Chapter Questions
Problem 19CTQ
Related questions
Question
give clear handwritten answer asap!
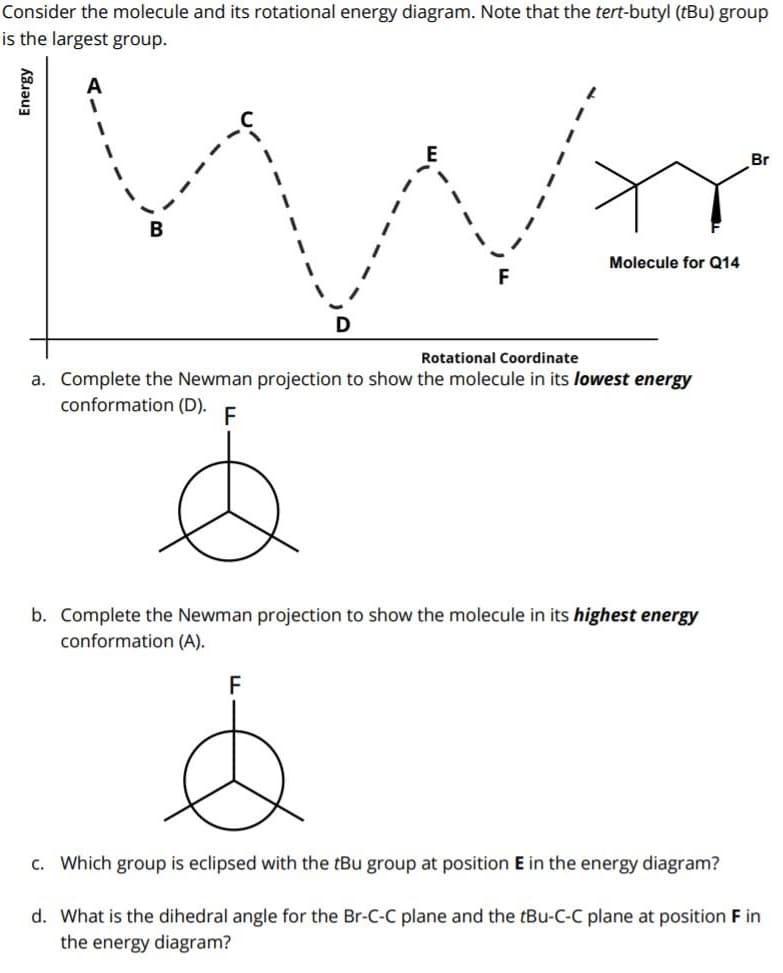
Transcribed Image Text:Consider the molecule and its rotational energy diagram. Note that the tert-butyl (tBu) group
is the largest group.
A
Br
B
Molecule for Q14
Rotational Coordinate
a. Complete the Newman projection to show the molecule in its lowest energy
conformation (D).
F
b. Complete the Newman projection to show the molecule in its highest energy
conformation (A).
F
c. Which group is eclipsed with the tBu group at position E in the energy diagram?
d. What is the dihedral angle for the Br-C-C plane and the tBu-C-C plane at position F in
the energy diagram?
Energy
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,
