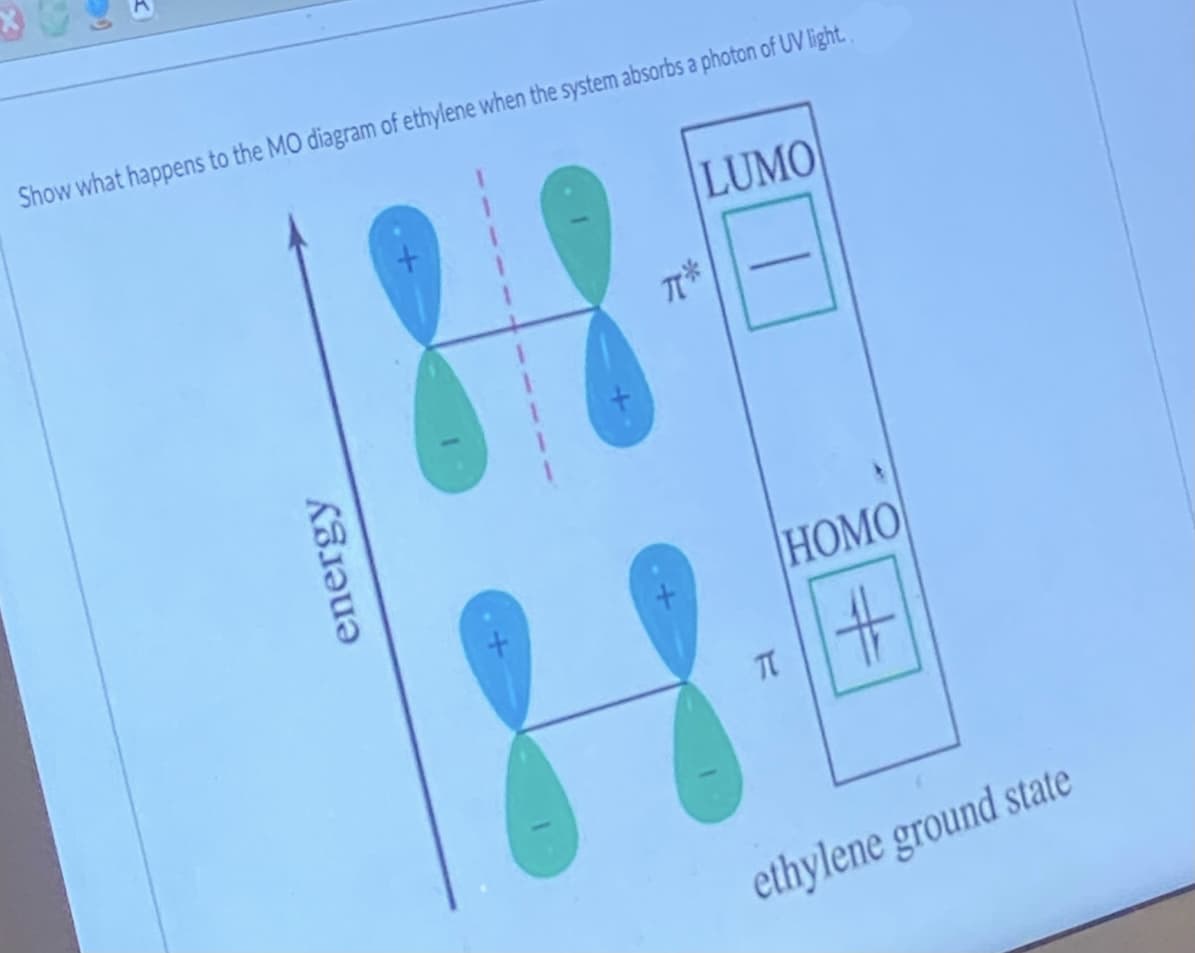
Show what happens to the MO diagram of ethylene when the system absorbs a photon of UV light. 11 18 LUMO HOMO %23 TC ethylene ground state energy
Q: How do you account for the fact that the dipole moments of fluoromethane and chloromethane are…
A: INTRODUCTION: Dipole moment is defined as the product of charge and the distance of separation…
Q: gu configura an S configuratic has an S configuration, r hat are the configurations around the CH3 d…
A: We have to tell that what E/Z configuration given compounds are exhibiting
Q: Four different molecules are shown below, and the highest priority functional group in each molecule…
A: The data given is,
Q: Trans 1,2-diphenylethylene. Find the canonical or resonant forms. Indicate your order of…
A: please find the answer attached
Q: 6. Set up the secular determinant for cyclobutadiene using Huckel approximation (a) solve for the…
A:
Q: xylenes (dimethylbenzenes) ortho, meta and para
A: The answer is as follows:
Q: ect the best depiction for the four pi-molecular orbitals of cyclobutadiene showing correct relative…
A: In this question, we will draw orbital energy diagram for give incomplete picture ( means without…
Q: Calculate the largest A using the appropriate rules. for the n-n' transition for the following…
A: If a molecule has a more resonating structure, it will have less energy and the structure will be…
Q: Use the inscribed polygon method to show the pattern of molecularorbitals in cyclooctatetraene.…
A: When a compound is aromatic, it gains extra stability. Aromaticity was determined by many methods.…
Q: a) C7H1402= 2(7)-14+2 CH CH, CHS CH CH2 CH CH 2H 3H 4H 2H 3H (overlapping quartet & triplet) PPM CHz…
A: NMR-spectroscopy is a technique to determine the structure of the organic molecules. The H1-NMR…
Q: • Q: Calculate the wavelength of the radiation that will be absorbed in promoting an electron from…
A: For butadiene X= +1.618 , + 0.618 X= - 1.618, -0.618
Q: 9. Use Frost's Circle to draw the electronic configuration of the ground state of the…
A:
Q: Consider the molecule and its rotational energy diagram. Note that the tert-butyl (tBu) group s the…
A:
Q: 1. Label the following rings as aromatic, anti-aromatic, or non-aromatic. Briefly indicate your…
A: a. 1. non-aromatic 2. non-aromatic 3. aromatic 4. non-aromatic 5. aromatic
Q: 1. Find out the correct order of increasing wavelength in UV-vis spectroscopy for the molecules and…
A:
Q: Determine the reactant that completes the following Diels-Alder rxn. II IV b.V Oc." Od.' a.
A: This is an example of Diels-Alder reaction (4+2 cycloaddition). Here benzyne acts as dienophile. we…
Q: Write the splitting pattern of hydrogens in vinyl chloride. Hb Hc Ha (A) Ha = doublet, Hb = doublet,…
A: splitting pattern of ha =2n+1 2n’+2 (double of dounles )
Q: Construct an MO energy diagram for the cycloheptatrienyl cation and describe its ground-state…
A: The MO-diagram for the cycloheptatrienyl cation is shown below.
Q: 9. Rank the following groups in order of decreasing the priority. (а) —СНз, —СН2CН3, —СH2СH2CН3, —…
A:
Q: CH3 CH3 9-BBN H3C. (1 еq.) "В 'B: 1 9-borab 3. + 3.
A: For 1 ) position 1 and 3 are respectively R, R configuration and For 2) position 1 and 3 are…
Q: 2 The dipoie moment of 1,2 -dibromobenzene (Structure 1jund 1,3 dibromubenene [ structure 2] is…
A: note: As per our company guidelines, we are supposed to answer only 1st question at a time. Kindly…
Q: What types of orbitals overlap to provide stability to the tert-butyl carbocation ((Ch3) 3C +) by…
A: Carbocation is sp2 hybridised. And in hyperconjugation carbocation sp2 orbital interacts with…
Q: why is the bond dissociation energy of tert-butyl(95kcal/mol) lower than propane (99kcal/mol)?
A:
Q: Rank the compounds in order of increasing symmetry: (a) OF2, (b) CF4, (c) NF3.
A: The lewis structures of the given molecules are,
Q: 10.1. Show, by construction of both a TS orbital array and an orbital symmetry correlation diagram,…
A:
Q: Pick the gauche Newman projection with the highest stability? H CH CH3 HA H, CHCHS H. HH H. CH3 CH3…
A: Correct answer is (II). Gauche Newman projection II is most stable among gauche forms. Reason: Angle…
Q: FTHANE 20 REPREMNTATION Click to rotate along the C-C bond by intervals of 60°. MODEL COMFORMATION…
A: The different conformation of ethane can be drawn as follows:
Q: Assume that the conjugated system of 1,3,5-hexatriene behaves as a particle-in-a box model.…
A: The HOMO of a compound is the highest occupied orbital and LUMO is the lowest unoccupied orbital.…
Q: A molecule with the molecular formula of C4H6 produces the IR spectrum shown below. Draw a structure…
A: IR spectroscopy is an important tool for detecting the functional group in an unknown organic…
Q: 3. Draw the splitting diagram for the indicated hydrogen and determine the expected splitting…
A: Well, in order to draw splitting pattern we need to look the neighbouring protons which are…
Q: For the following molecula: CH₂ Н Br CH3 A) Select its diastereomer: Н. -OH H -H CH3 CH3 -OH -Br B)…
A:
Q: The index of Hydrogen deficiency indicates the degree of unsatuiation of the molecule which help in…
A: Degree of unsaturation for hydrocarbons is calculated using formula : U = ( Hydrogen in alkane -…
Q: 12. Which of the following structures has the S configuration? H CH,CH, CH,CH, CH3 "OH H,CH,C Br CH3…
A: First of all wedge dash is converted to fischer projection and then Configuration is predicted .
Q: 9. Use the Cahn-Ingold-Prelog rules to choose the correct priority sequence for the assignment of…
A: Cahn-Ingold-Prelog rule :- In this rule, the R or S configuration of optically active…
Q: 2. Assign the R and S configuration of the following molecules. В. С. А. CH3 H OCH3 H3C CH2CH3 H HS…
A: R/S nomenclature is based on CIP (Cahn-Ingold-Prelog) rule which states that higher is the atomic…
Q: 4. Conjugated Compounds: The definition, instrumental analysis, synthesis and the application The…
A: Conjugation Compounds with more than one double or triple bond that are present in alternative…
Q: the (c) Provide following : HB Hc splitting patterns for НА (i) Ý HB На (üi) X' `Hc
A:
Q: How can the two isomers having molecular formula C2H6O be distinguishedby IR spectroscopy?
A: Isomers are defined as the chemical compounds that have same chemical formula but have different…
Q: Usimg SPARTAN® Molecular Modeling draw and design the transition state and do the appropriate…
A: Diels Alder reaction involves electron rich diene and electron poor dienophile that form cyclo…
Q: 9. Use the Cahn-Ingold-Prelog rules to choose the correct priority sequence for the assignment of…
A: Always heteroatoms (compare to C, H) will be preceded in priority.
Q: What multiplicity will the CH3 of the following molecule have? CH3 a) Singulete b) Doublet c)…
A: NMR SPECTROSCOPY: Nuclear Magnetic Resonance spectroscopy is used to find out the presence of carbon…
Q: "The IR absorption due to the stretn Stretching of which of these causon- hydrogen bonds occue at…
A:
Q: Which of the following compounds exhibits the largest carbonyl stretching frequency? В. С. D. Е. H.…
A: Stretching frequency depends upon the strength of bond . If the bond is stromger then stretching…
Q: What are the E/Z configurations of the following compounds? -ОН CH₂Br H3CO HO CH=CHNH2 CC13 HO…
A: Assign priority to the groups attached to each doubly bonded carbon atom according to the CIP…
Q: 11) which molecule below has S-configuration? A B D
A: We'll answer the first question since the exact one wasn't specified. Please submit a new question…
Q: Why does "Zincblende" have a F(-4)3m space group designation? F stands for face centered and the…
A: One way of representing point groups, plane groups and space groups is Hermann-Mauguin notation. The…
Q: Calculate the degrees of unsaturation for a compound of molecular formula CgH&BRNO,. 6. 0 5 3.
A: Degrees of unsaturation is also known as Index of Hydrogen Deficiency (IHD), is a count of how many…
Q: Select the correct arrangement for the following processes from the low to high energy: O 1-…
A:
Q: b) The B3LYP/6-31G(d) energy-minimized structure of 3-(4'-nitrophenyl)furan is shown below on an…
A: There are two factors that govern the stability of biaryl systems: Electronic repulsion between…
Q: The index of Hydrogen deficiency indicates the degree of unsatuiation of the molecule which help in…
A:
10.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Draw the potential energy profile of 3-ethyl-2-methylpentane as a function of the dihedral angle. (Conformational analysis is done with respect to the bond joining C2 and C3.)Answer the following questions for 1,3,5- hexatriene, the conjugated triene containing six carbons.Which MOs are bonding? Which are antibonding?Answer the following questions for the MOs of 1,3,5-hexatriene: a. Which are bonding MOs, and which are antibonding MOs? b. Which MOs are the HOMO and the LUMO in the ground state? c. Which MOs are the HOMO and the LUMO in the excited state? d. Which MOs are symmetric, and which are antisymmetric? e. What is the relationship between HOMO and LUMO and symmetric and antisymmetric MOs?
- Answer the following questions for the MOs of 1,3-butadiene: a. Which are pie bonding MOs, and which are p* antibonding MOs? b. Which MOs are symmetric, and which are antisymmetric? c. Which MO is the HOMO and which is the LUMO in the ground state? d. Which MO is the HOMO and which is the LUMO in the excited state? e. What is the relationship between the HOMO and the LUMO and symmetric and antisymmetric orbitals?1.The [CoCl4]2- anion is intensely blue colored and [CoCl6]4- is a lightly colored pink. explain breifly 2.Why is the method of descending symmetry needed to derive an mo diagram for acetylene (ethyne), but not needed for diazene((NH)2)?Which compoundshave zero dipole moments? cyclohexene or 1,2-dichlorocyclohexene
- Answer the following questions for the molecular orbitals (MOs) of 1,3,5,7-octatetraene: a. How many MOs does the compound have? b. Which are the bonding MOs, and which are the antibonding MOs? c. Which MOs are symmetric, and which are antisymmetric? d. Which MO is the HOMO and which is the LUMO in the ground state? e. Which MO is the HOMO and which is the LUMO in the excited state? f. What is the relationship between HOMO and LUMO and symmetric and antisymmetric orbitals? g. How many nodes does the highest-energy MO of 1,3,5,7-octatetraene have between the nuclei?Does the MO energy diagram of cyclooctatetraeneConsider 1-bromopropane, CH3CH2CH2Br. (a) Draw a Newman projection for the conformation in which CH3 and -Br are anti (dihedral angle 180°). (b) Draw Newman projections for the conformations in which - CH3 and -Br are gauche (dihedral angles 60° and 300°). (c) Which of these is the lowest energy conformation? (d) Which of these conformations, if any, are related by reflection?
- Does chloromethane contain a plane of symmetry? Is it a chiral?What is the degree of unsaturation of C8H10ClNO? a. 4 b. 5 c. 6 d. 7Draw the energy diagram for cyclooctatetraene dianion , C8H8 (2-) , molecular orbitals. The polygon rule is helpful. Label each MO as bonding , antibonding, or nonbonding, and add the nonbonding line. Identify the LOMO, HOMO, LUMO and HUMO orbitals. Is this dianion aromatic , non aromatic or anti aromatic? Thank you!



