Consider the table. Metal Tm (K) Li 454 371 336 Rb 312 Cs 302 Na K A.Svap Using the data, calculate ASfus and = AHfus (kJ/mol) Tb (K) AHvap (kJ/mol) 1615 1156 1033 956 942 AS fus= 2.99 2.60 2.33 2.34 2.10 ASvap I for Li. 134.7 89.6 77.1 69 66 K-m J K-me
Consider the table. Metal Tm (K) Li 454 371 336 Rb 312 Cs 302 Na K A.Svap Using the data, calculate ASfus and = AHfus (kJ/mol) Tb (K) AHvap (kJ/mol) 1615 1156 1033 956 942 AS fus= 2.99 2.60 2.33 2.34 2.10 ASvap I for Li. 134.7 89.6 77.1 69 66 K-m J K-me
World of Chemistry, 3rd edition
3rd Edition
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Chapter10: Energy
Section: Chapter Questions
Problem 7STP
Related questions
Question
Please answer 3 simple q.this
![What concentration of SO3 is in equilibrium with Ag,SO, (s) and 1.90 x 10-³ M Ag+?
The Ksp of Ag₂SO3 can be found
this table.
[so-] =
1.52 X10-5
Incorrect
I
M](/v2/_next/image?url=https%3A%2F%2Fcontent.bartleby.com%2Fqna-images%2Fquestion%2Ff6b731bf-263a-45d3-a264-a5330485f8e3%2F55b7d636-56a6-4cc7-907e-4a3795efcf64%2Fhcf8yme_processed.jpeg&w=3840&q=75)
Transcribed Image Text:What concentration of SO3 is in equilibrium with Ag,SO, (s) and 1.90 x 10-³ M Ag+?
The Ksp of Ag₂SO3 can be found
this table.
[so-] =
1.52 X10-5
Incorrect
I
M
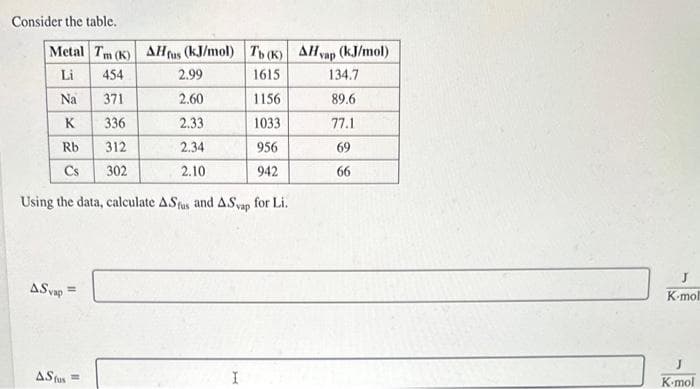
Transcribed Image Text:Consider the table.
Metal Tm (K) AH fus (kJ/mol)
Li
454
Na
371
K
336
Rb
312
Cs 302
Using the data, calculate ASfus and
ASvap
=
2.99
2.60
2.33
2.34
2.10
AS fus=
Tb (K) AHvap (kJ/mol)
1615
1156
1033
956
942
ASvap
I
for Li.
134.7
89.6
77.1
69
66
J
K-moll
K-mol
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning