Data Sheet with Sample Results Moles of acetic acid contained in 50.0 mL of Mass of sodium acetate needed to equalize the a 0.10 M sample solution: _ moles of acetic acid in sample solution: Buffer pH Study Buffer Sample initial pH: 4.75 pH after addition of 0.100 M HC1 pH after addition of 0.100 M NaOH Total volume added pH Total volume added pH 1.00 mL 4.70 1.00 mL 4.80 2.00 mL 4.65 2.00 mL 4.85 3.00 mL 4.60 3.00 mL 4.90 4.00 mL 4.54 4.00 mL 4.97 5.00 mL 4.48 5.00 mL 5.05 Diluted Buffer Sample initial pH: 4.80 pH after addition of 0.100 M HC1 pH after addition of 0.100 M NaOH Total volume added pH Total volume added pH 1.00 mL 4.50 1.00 mL 5.00 2.00 mL 4.20 2.00 mL 5.25 3.00 mL 3.35 3.00 mL 5.70 4.00 mL 2.50 4.00 mL 9.50 5.00 mL 2.20 5.00 mL 11.67 Initial pH of unbuffered HCl: 3.00 pH after addition of 1.0 mL NAOH: 10.50
Data Sheet with Sample Results Moles of acetic acid contained in 50.0 mL of Mass of sodium acetate needed to equalize the a 0.10 M sample solution: _ moles of acetic acid in sample solution: Buffer pH Study Buffer Sample initial pH: 4.75 pH after addition of 0.100 M HC1 pH after addition of 0.100 M NaOH Total volume added pH Total volume added pH 1.00 mL 4.70 1.00 mL 4.80 2.00 mL 4.65 2.00 mL 4.85 3.00 mL 4.60 3.00 mL 4.90 4.00 mL 4.54 4.00 mL 4.97 5.00 mL 4.48 5.00 mL 5.05 Diluted Buffer Sample initial pH: 4.80 pH after addition of 0.100 M HC1 pH after addition of 0.100 M NaOH Total volume added pH Total volume added pH 1.00 mL 4.50 1.00 mL 5.00 2.00 mL 4.20 2.00 mL 5.25 3.00 mL 3.35 3.00 mL 5.70 4.00 mL 2.50 4.00 mL 9.50 5.00 mL 2.20 5.00 mL 11.67 Initial pH of unbuffered HCl: 3.00 pH after addition of 1.0 mL NAOH: 10.50
Chemical Principles in the Laboratory
11th Edition
ISBN:9781305264434
Author:Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Chapter24: The Standardization Of A Basic Solution And The Determination Of The Molar Mass Of An Acid
Section: Chapter Questions
Problem 3ASA: A 0.3012g sample of an unknown monoprotic acid requires 24.13mL of 0.0944MNaOH for neutralization to...
Related questions
Question
100%
I need help to calculate this data please
thanks a lot
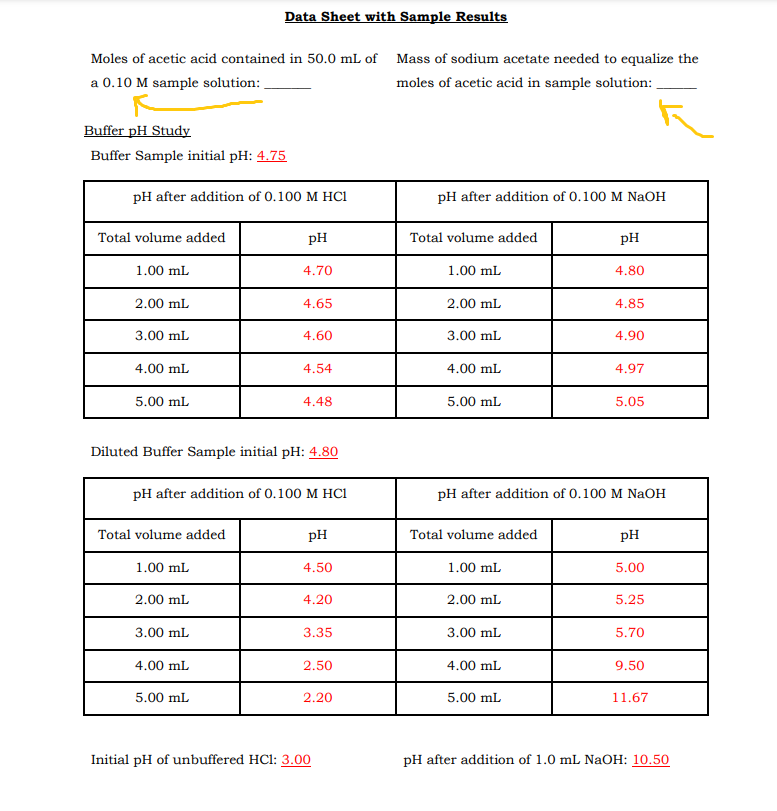
Transcribed Image Text:Data Sheet with Sample Results
Moles of acetic acid contained in 50.0 mL of Mass of sodium acetate needed to equalize the
a 0.10 M sample solution:
moles of acetic acid in sample solution:
Buffer pH Study
Buffer Sample initial pH: 4.75
pH after addition of 0.100 M HC1
pH after addition of 0.100 M NaOH
Total volume added
pH
Total volume added
pH
1.00 mL
4.70
1.00 mL
4.80
2.00 mL
4.65
2.00 mL
4.85
3.00 mL
4.60
3.00 mL
4.90
4.00 mL
4,54
4.00 mL
4.97
5.00 mL
4.48
5.00 mL
5.05
Diluted Buffer Sample initial pH: 4.80
pH after addition of 0.100 M HC1
pH after addition of 0.100 M NaOH
Total volume added
pH
Total volume added
pH
1.00 mL
4.50
1.00 mL
5.00
2.00 mL
4.20
2.00 mL
5.25
3.00 mL
3.35
3.00 mL
5.70
4.00 mL
2.50
4.00 mL
9.50
5.00 mL
2.20
5.00 mL
11.67
Initial pH of unbuffered HCl: 3.00
pH after addition of 1.0 mL NAOH: 10.50
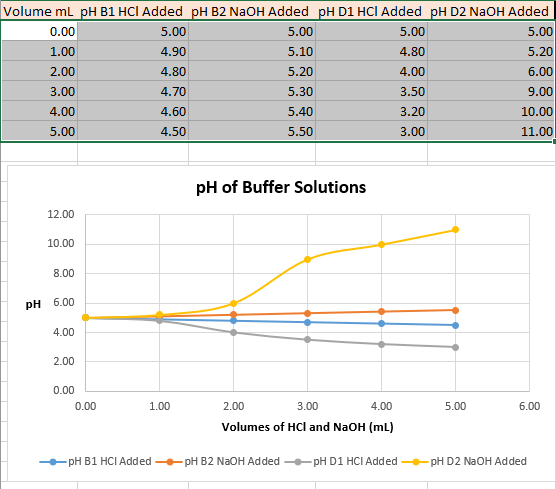
Transcribed Image Text:Volume ml pH B1 HCI Added pH B2 NaOH Added pH D1 HCI Added pH D2 NaOH Added
0.00
5.00
5.00
5.00
5.00
1.00
4.90
5.10
4.80
5.20
2.00
4.80
5.20
4.00
6.00
3.00
4.70
5.30
3.50
9.00
4.00
4.60
5.40
3.20
10.00
5.00
4.50
5.50
3.00
11.00
pH of Buffer Solutions
12.00
10.00
8.00
pH 6.00
4.00
2.00
0.00
0.00
1.00
2.00
3.00
4.00
5.00
6.00
Volumes of HCl and NaOH (ml)
-pH B1 HCI Added pH B2 N2OH Added
pH D1 HCI Added
pH D2 NaOH Added
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 5 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemical Principles in the Laboratory
Chemistry
ISBN:
9781305264434
Author:
Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:
Brooks Cole



Chemical Principles in the Laboratory
Chemistry
ISBN:
9781305264434
Author:
Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:
Brooks Cole

