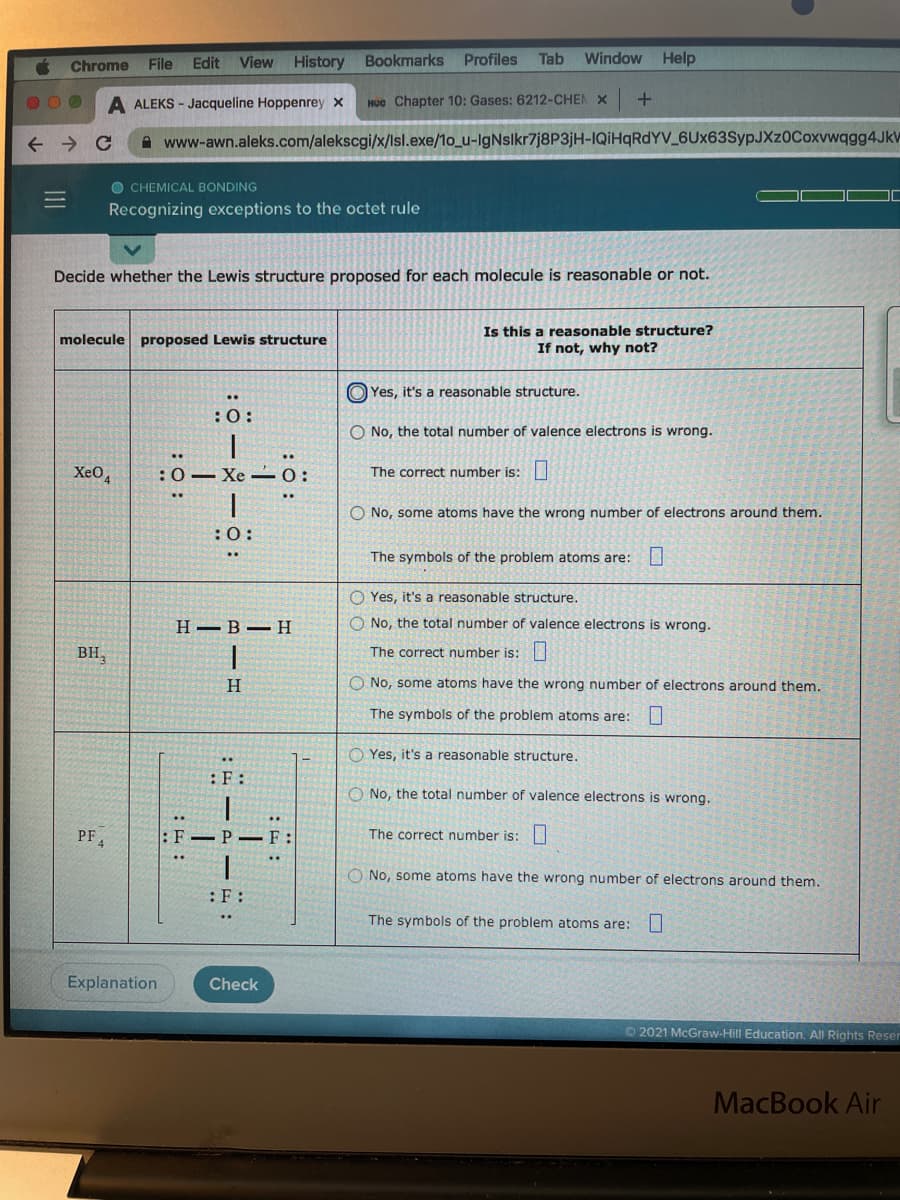
Decide whether the Lewis structure proposed for each molecule is reasonable or not. Is this a reasonable structure? If not, why not? molecule proposed Lewis structure O Yes, it's a reasonable structure. :0: O No, the total number of valence electrons is wrong. Xe0, :0- Xe 0: The correct number is: .. .. O No, some atoms have the wrong number of electrons around them. :0: The symbols of the problem atoms are: U O Yes, it's a reasonable structure. Н— В — Н O No, the total number of valence electrons is wrong. BH, The correct number is: H O No, some atoms have the wrong number of electrons around them. The symbols of the problem atoms are: O Yes, it's a reasonable structure. :F: O No, the total number of valence electrons is wrong. PF :F P F: The correct number is: No, some atoms have the wrong number of electrons around them. :F : The symbols of the problem atoms are: L :ö:
Decide whether the Lewis structure proposed for each molecule is reasonable or not. Is this a reasonable structure? If not, why not? molecule proposed Lewis structure O Yes, it's a reasonable structure. :0: O No, the total number of valence electrons is wrong. Xe0, :0- Xe 0: The correct number is: .. .. O No, some atoms have the wrong number of electrons around them. :0: The symbols of the problem atoms are: U O Yes, it's a reasonable structure. Н— В — Н O No, the total number of valence electrons is wrong. BH, The correct number is: H O No, some atoms have the wrong number of electrons around them. The symbols of the problem atoms are: O Yes, it's a reasonable structure. :F: O No, the total number of valence electrons is wrong. PF :F P F: The correct number is: No, some atoms have the wrong number of electrons around them. :F : The symbols of the problem atoms are: L :ö:
Chemistry for Engineering Students
4th Edition
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Lawrence S. Brown, Tom Holme
Chapter5: Gases
Section: Chapter Questions
Problem 5.97PAE: 97 Homes in rural areas where natural gas service is not available often rely on propane to fuel...
Related questions
Question
Transcribed Image Text:Chrome
File
Edit
View History Bookmarks
Profiles
Tab
Window
Help
A ALEKS - Jacqueline Hoppenrey x
Hoc Chapter 10: Gases: 6212-CHEN X
A www-awn.aleks.com/alekscgi/x/lsl.exe/1o_u-IgNslkr7j8P3jH-IQİHQRDYV_6Ux63SypJXz0Coxvwqgg4JkV
O CHEMICAL BONDING
Recognizing exceptions to the octet rule
Decide whether the Lewis structure proposed for each molecule is reasonable or not.
Is this a reasonable structure?
If not, why not?
molecule proposed Lewis structure
O Yes, it's a reasonable structure.
O No, the total number of valence electrons is wrong.
Xe0,
:0- Xe -0:
The correct number is:
O No, some atoms have the wrong number of electrons around them.
:0:
The symbols of the problem atoms are:
O Yes, it's a reasonable structure.
H -B-H
O No, the total number of valence electrons is wrong.
BH,
The correct number is:
H.
O No, some atoms have the wrong number of electrons around them.
The symbols of the problem atoms are:
O Yes, it's a reasonable structure.
O No, the total number of valence electrons is wrong.
PF4
:F–P – F:
The correct number is:
O No, some atoms have the wrong number of electrons around them.
:F:
The symbols of the problem atoms are:
Explanation
Check
O2021 McGraw-Hill Education. All Rights Reser
MacBook Air
111
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax