In the POF, molecule, the P atom is the central atom. Draw a Lewis diagram of POF, for which all formal charges are equal to zero. How many double bonds are there in the structure that you have drawn? number of double bonds = Draw a Lewis diagram in which the octet rule is satisfied on all atoms. What are the formal charges on the following atoms in the structure that you have drawn? P Based on formal charge, which is the best Lewis structure for the molecule?
In the POF, molecule, the P atom is the central atom. Draw a Lewis diagram of POF, for which all formal charges are equal to zero. How many double bonds are there in the structure that you have drawn? number of double bonds = Draw a Lewis diagram in which the octet rule is satisfied on all atoms. What are the formal charges on the following atoms in the structure that you have drawn? P Based on formal charge, which is the best Lewis structure for the molecule?
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter3: Atomic Shells And Classical Models Of Chemical Bonding
Section: Chapter Questions
Problem 94AP: The molecular ion S3N3 has the cyclic structure All SN bonds are equivalent. (a) Give six...
Related questions
Question
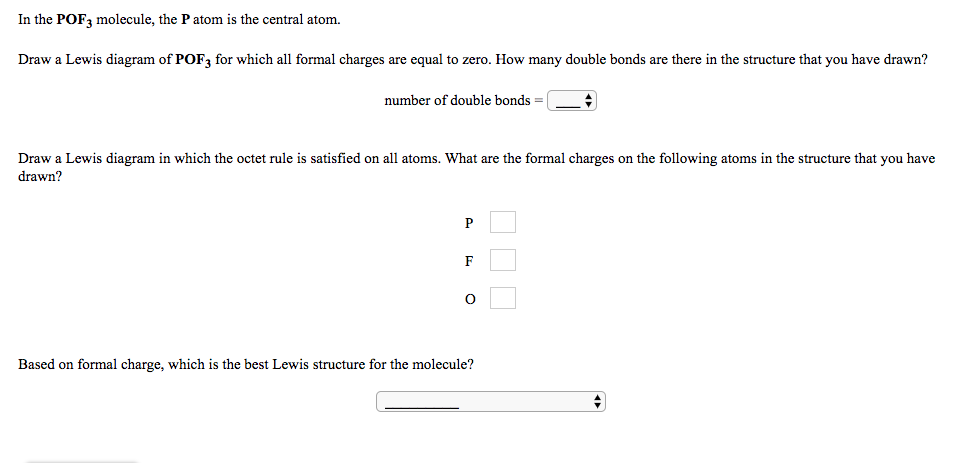
Transcribed Image Text:In the POF, molecule, the P atom is the central atom.
Draw a Lewis diagram of POF, for which all formal charges are equal to zero. How many double bonds are there in the structure that you have drawn?
number of double bonds =
Draw a Lewis diagram in which the octet rule is satisfied on all atoms. What are the formal charges on the following atoms in the structure that you have
drawn?
P
Based on formal charge, which is the best Lewis structure for the molecule?
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning



Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning