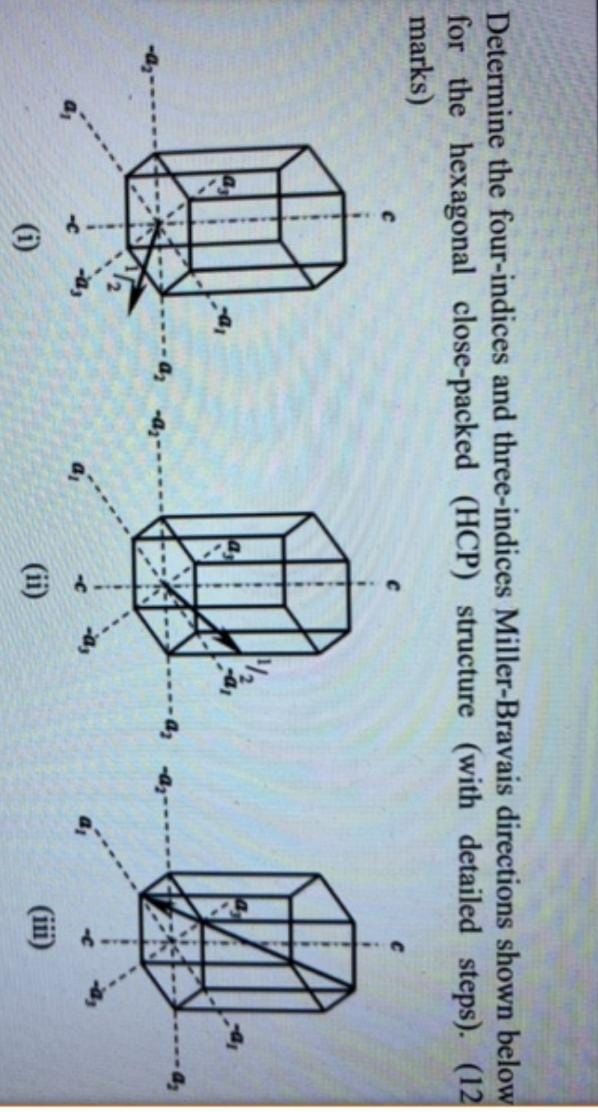
Determine the four-indices and three-indices Miller-Bravais directions shown below for the hexagonal close-packed (HCP) structure (with detailed steps). (12 marks) (i) (ii)
Q: Write the miller indices of the crsytallographic plane shown: 8.
A: For the given plane in the question, there are three steps for determination of the millers indices.…
Q: 3v point group is given as Co
A: In group theory, representation of higher dimension can be reduced in to representation of lower…
Q: edral site). Determine by how much the diameter of the C atom in y-Fe is overs ct to the gap of the…
A: An interstitial atom is one which occupies a site in crystal structure and normally not…
Q: A compound Y (150.0 g/mol) in a 1.00 cm quartz cell has an extinction coefficient (ε) of 23,150 at…
A: Answer = 509.12 Complete solution is given in next step.
Q: (a) Why the n to a' and a to a transitions are more prominent in UV- Visible spectroscopy? Also,…
A: (a) Why then to n and a to transitions are more prominent in UV Visible spectroscopy? Also, discuss…
Q: Before the endpoint, the Chloride ion which is in excess adsorbed on the surface of AgCl, imparting…
A: In a precipitation titration, analyte and titrant mixes and form an insoluble precipitate. Based on…
Q: Which row of the ICF table is defined by the balancedequation’s coefficients?
A: NOTE: It will be ICE i.e. initial-change-equilibrium table and not ICF.
Q: When the D line of sodium light impinges an air-diamond interface at an angle of incidence of 25.0°,…
A: The solution is given below -
Q: what the energy of neutron is suitable for crystal diffraction [the atomic spacing between two…
A: Given The atomic spacing between two bonded atoms is generally around
Q: Why magnetic moment of Fe(II) is higher than Mn(II). Discuss in terms of orbital contribution . (…
A:
Q: what will happen to the rate constant if the concentrat -fold.
A: As we know in elementary reaction order of reaction is sum of stoichometric coefficients of…
Q: stoichimetric
A: According to the question, we need to determine the stoichiometric ratio of Fe3O4 in Fe2O3. Let the…
Q: important is it to embed the 'emitter recoilness emission of a Y-ray. How The Mossbauer effect…
A: Emitter nucleus is embedded in a bulky, rigid crystal lattice to ensure that energy don't get lost…
Q: a. На Hb splitting pattern integration b.
A: Splitting pattern in NMR- Count how many identical hydrogen atom present in the adjacent and…
Q: 12. (a). In principle, a material sample can be analyzed to its functional group content using Raman…
A: Answar- a) 1) What are the Basic Principles of Raman Spectroscopy. It is the shift in…
Q: 5. Surface excess of the compounds that have surface activity is: 7,-0 b) l'₁ 0 d) r>0 only above…
A: The solution is given below -
Q: The polarity of silica gel (stationary phase) is low. Select one: O True O False
A: In chromatography, we use a stationary phase, and one mobile phase. The stationary phase must be…
Q: Examples Example (1);- The following data obtained from decomposit of H2 02 in aquees Solution:…
A: Please find the graph attached here with the solution .
Q: explain why it would not be possible to determine the orders with respect to both iodide and…
A: To explain: why it would not be possible to determine the orders with respect to both iodide and…
Q: Given the following data for Lisinopril, pick the best answer based on Lipinski rules of five:…
A: Lipinski rules of five: The properties of the oral drug are, Permeable or poor absorption Log P…
Q: Why are n-π* transitions generally of low intensities/low absorptions?
A: The n to π* transition corresponds to the excitation of an electron from one of the unshared pair to…
Q: 3) Figure below shows an x-ray diffraction pattem for single crystal scandium nitride (ScN), having…
A:
Q: give handwritten explanation and answer- Considering the patterns you have identified, estimate the…
A: Here we have to explain the melting point of elements Cadmium, Vanadium and Cobalt-
Q: ronydride principie blnd metals Ih of the M-H-BH M. B-H monodentate bidentate tridentate Detemine…
A: Symmetry is very important in chemistry researches and group theory is the tool that is used to…
Q: What is piezoelectricity? short discription
A: The pizoelectricity concept was put forward by Paul-Jacques Curie and his younger brother Pierre,…
Q: Explain the effect of donor concentration (doping level) and temperature on electron mobility in…
A: Please find your solution below : The electron mobility tells that how quickly and electron can move…
Q: Stainless steel typically contains 1% carbon, 1-5% manganese, 0.05% phosphorus, 1-3% silicon, 5-10%…
A: Stainless-steel is a commonly used alloy of iron, carbon and some other metals. Each element have to…
Q: KUESTION: 1 Silicon at T=300K contains a acceptor impurity concentration at Na=1016 cm-3. Determine…
A: Given:- Let the donor concentration = ND cm-3 Acceptor concentration NA =…
Q: 1. Using the data below calculate the solvation energy of the complex. Give your answer in kJ mol-1…
A: Given that, for a complex Molecular system Energy (Hartrees) Complex in gas phase -769.759370…
Q: re 1 shows an x-ray diffraction pattern for a-iron (BcC) taken using a di hochromatic x-radiation…
A:
Q: Give the chemical symbol for the clement with the ground-state clectron conliguration (Ar4s3d".…
A: 1) Chemical symbol for the element with electronic configuration [Ar]3d24s2 is Titanium.…
Q: We Want Spectroscopic, IR bands,, HNMR bands
A: The structure of the unknown compounds can be readily determined by analyzing the spectroscopical…
Q: hedict the poduct that will be frimed by the neachon below. Colzlag) + Mn (5)>
A: Here aqueous cobalt iodide CoI2 and solid manganese Mn have been reacted. We have to predict the…
Q: why is lupac for Hg2Cl2 Mercury (I) Chloride, instead of Mercury (II) Chloride?
A: Given, Molecules is Hg2Cl2 Why is IUPAC for Hg2Cl2 Mercury (I) Chloride, instead of Mercury (II)…
Q: Name of Experiment: Determination of the formula and stability constant of a silver complex. Q1:…
A: Name of Experiment- Determination of formula and stability constant of Silver complex. 1) Purpose…
Q: d) By using the data provided below, calculate the PillingBedworth ratio for the nickel (Ni) –…
A: D) Given: Atomic mass of nickel = 58.7 g.mol-1 Density of nickel = 8.91 Mg.m-3 Molecular mass of…
Q: Explain 'why glass cells are not used in, UV-spectroscopy.
A: To explain: Why glass cells are not used in UV-spectroscopy.
Q: Consider the mineral dissolution reactions below: FeSmackinawite -> Fe2+ + S2- CuScovellite ->…
A: Given: ΔGo of Fe+2 = -78.8 kJ/mol ΔGo of S-2 = 85.7 kJ/mol ΔGo of FeS = -100.4 kJ/mol…
Q: Consider the following series of poly(phenylene oxide) polymers A–D, and their glass transition…
A: The estimation of Tg depends on the versatility of the polymer chain - the more stable the chain,…
Q: The Butler Volmer (BV) rate equation should be introduced in electrochemistry because: The Nernst…
A: Electrochemistry is branch of chemistry in which we deal with both oxidation and reduction reaction…
Q: Define the following:(i) Schottky defect (ii) Frenkel defects (iii) F-centre
A: The answer for part (i) is given below. Kindly repost the other parts as separate one.
Q: How is the MnO4- ion different from the Cu2+ metal ion in regards to UV-Vis?
A: UV-Vis Spectroscopy is a technique that is used to analyze how much light a chemical substance…
please very soon
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

- In a vacuum chamber filled with argon gas at 100 mTorr, a plasma is ignited resulting to 1% ionization. This is already a "hot plasma" by lab processing standards. Calculate the number density of plasma electrons (should be about the same as the argon ion density due to quasineutrality) in this 1% ionized plasma.In a vacuum chamber filled with argon gas at 100mTorr, a plasma is ignited resulting to 1% ionization. This is already a "hot plasma" by lab processing standards. Calculate the number density of plasma electrons (should be about the same as the argon ion density due to quasineutrality) in this 1% ionized plasma.I) Diamond crystallises in a face-centred cubic structure with a = 3.5625 Å. Calculate the d-spacing and peak position of the first allowed peak (i.e. the peak with the lowest hkl value that meets the selection rules) using a wavelength of 0.71073 Å. II) A solvothermal synthesis gave rise to a mixture of two crystalline phases: one phase consists of 50-65 μm octahedral crystallites while the other phase consists of 150-180 μm hexagonal plates. Identify a separation technique that would likely be effective in separating these materials.
- atomic radius is 0.1430 nm calculate the planer density of (211) plane at BCC structureTrue or False 1. When reacting a metal that can be included in tetracycline coordination, it should give a the drug a 3D arrangement rather than a planar because of the delocalization of the electrons on the ring. 2. Macrolides such as erythromycin are used as alternative for patients with allergy on beta-lactam antibiotics 3. Concomitant administration of cephalosporins and macrolides can prolong the QT elongation in electroencephalogram 4. When administered orally, azithromycin has a long duration of action 5. Chloramphenicol are known to cause a Type B adverse drug reaction known as Cross-allergic reactionsSpectrum (a) in the figure to the right is the UV-vis absorption spectrum of CdTe nanoparticles. What is the bandgap of these particles in eV? (TIP: You don’t need to square the absorbance values.)
- 1. "Band structure" of "Hatom chain" and "H mole chain" are drawn as "graphs" and "intermediate process". Describe it in detail.Determine the linear and planar densities of the (111) direction of zinc (HC structure).A device to understand how iron behaves at high temperatureswe want to design. This device (with 1% accuracy) allows the change in volume (heating) of a 1cm 3 iron cube.the resulting polymorphic transformation) must be able to feel. HMK structure in iron 911 0 C and latticeparameter 0.2863 nm. At 913 0 C, it has YMK structure and its lattice parameter is 0.3591 nm. Of measuring instrumentDetermine the accuracy tolerance it needs (i.e. at least how many cm 3 to detect a changemust). It is necessary to use at least 6 digits after the comma in this question. Hint: YMK first andFor HMK, the volume of an atom is found and then, using the number of atoms in each structure, the ironThe question can be started by finding the different volumes.
- In a vacuum chamber filled with argon gas at 100mTorr, a plasma is ignited resulting to 1% ionization. This is already a "hot plasma" by lab processing standards. Calculate the number density of plasma electrons (should be about the same as the argon ion density due to quasineutrality) in this 1% ionized plasma. (You can find all relevant constants in the net).We have a pink crystals of an ionic compound of an f- block element (M). The crystals house the octahydrate [ M(H2O)8]3+ and an anion which is not chloride and does not luminesce under UV. (A) First, draw and label a plausible solid-state geometry for the hydrated cation. (B)Provide ALL potential identities for the f- block element, M, based on the information supplied above, defending your choices and discussing the applicable chemical concepts. (C)What are the potential solid state MCI3 geometries and coordination numbers for ALL of the components proposed in section (B)? Explain briefly why, focusing on the key chemical principles. (D) In ion - exchange chromatography, which of the trivalent f-block ions indicated in component (B) would you anticipate to elute first? Explain your response, emphasising the important chemical concepts.I need backgorund information of ferrocene and it's components also who found it. Thank you so much!

