Do you expect benzaldehyde or stilbene to travel farther on a TLC plate? Please explain, being sure to mention important intermolecular interactions.
Do you expect benzaldehyde or stilbene to travel farther on a TLC plate? Please explain, being sure to mention important intermolecular interactions.
Chapter89: Thin-layer Chromatography
Section: Chapter Questions
Problem 1P
Related questions
Question
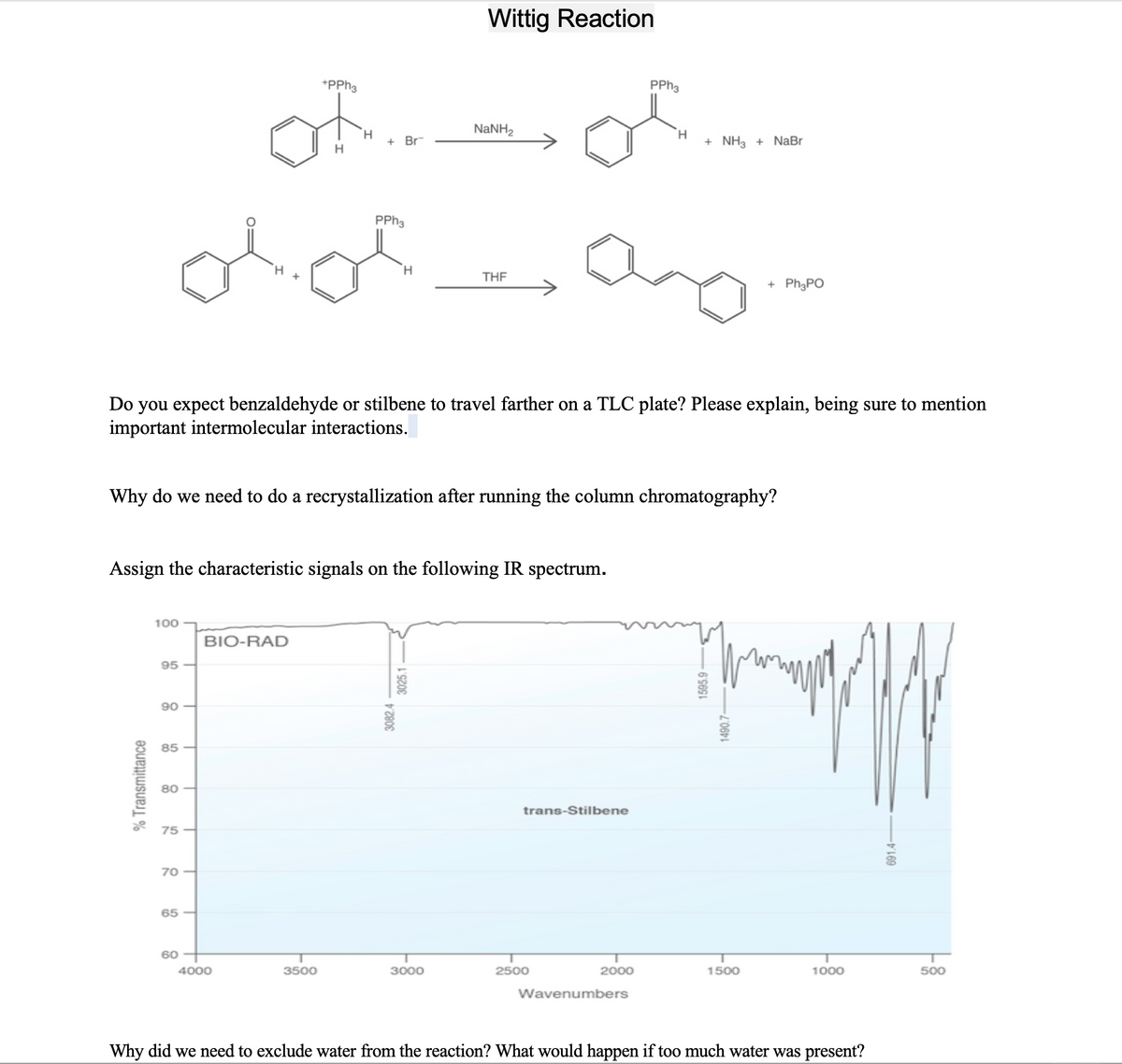
Transcribed Image Text:Wittig Reaction
*PPH3
PPH3
NaNH2
H.
+ Br
H.
+ NH3 + NaBr
PPH3
H.
THE
+ PH3PO
Do you expect benzaldehyde or stilbene to travel farther on a TLC plate? Please explain, being sure to mention
important intermolecular interactions.
Why do we need to do a recrystallization after running the column chromatography?
Assign the characteristic signals on the following IR spectrum.
100
BIO-RAD
95 -
90
85
80
trans-Stilbene
75
70
65
60
4000
3500
3000
2500
2000
1500
1000
500
Wavenumbers
Why did we need to exclude water from the reaction? What would happen if too much water was present?
% Transmittance
3082.4
3025.1
1595.9
691.4
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning


EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning
