Does the equilibrium favor the reactant or the products? Choose one: products neither reactants There is no way to tell Where is the equilibrium position on the graph? Choose one: At the lowest point in the curve At the highest point in the curve on the left (reactant side) Halfway between the reactants and products At the highest point in the curve on the right (product side)
Does the equilibrium favor the reactant or the products? Choose one: products neither reactants There is no way to tell Where is the equilibrium position on the graph? Choose one: At the lowest point in the curve At the highest point in the curve on the left (reactant side) Halfway between the reactants and products At the highest point in the curve on the right (product side)
Chemistry by OpenStax (2015-05-04)
1st Edition
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Chapter3: Composition Of Substances And Solutions
Section: Chapter Questions
Problem 72E: What mass of HCl is contained in 45.0 mL of an aqueous HCl solution that has a density of 1.19 g...
Related questions
Question
Consider the gas-phase reaction of hydrogen and iodine at 298.15 K and the graph showing the free energy of the H2-I2-HI mixture.
H2(g)+I2(g)⇌2HI(g)
Does the equilibrium favor the reactant or the products?
Choose one:
products
neither
reactants
There is no way to tell
Where is the equilibrium position on the graph?
Choose one:
At the lowest point in the curve
At the highest point in the curve on the left (reactant side)
Halfway between the reactants and products
At the highest point in the curve on the right (product side)
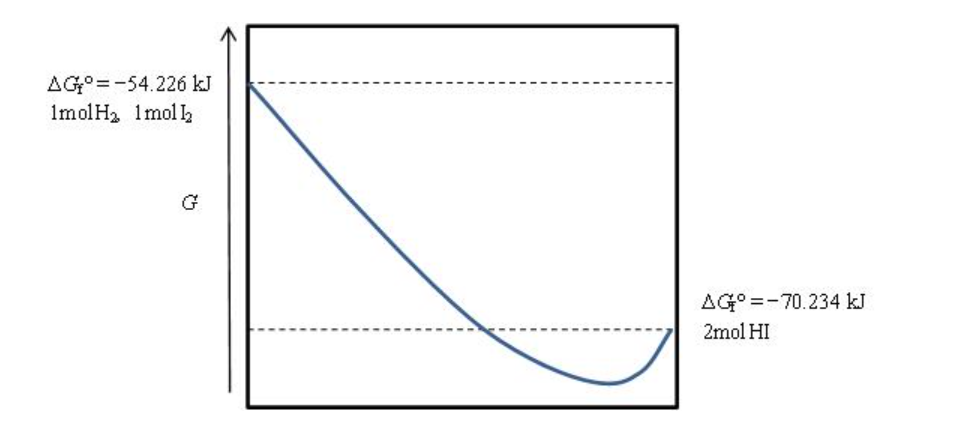
Transcribed Image Text:AG°=-54.226 kJ
ImolH2 1mol 2
G
AG° =-70.234 kJ
2mol HI
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning