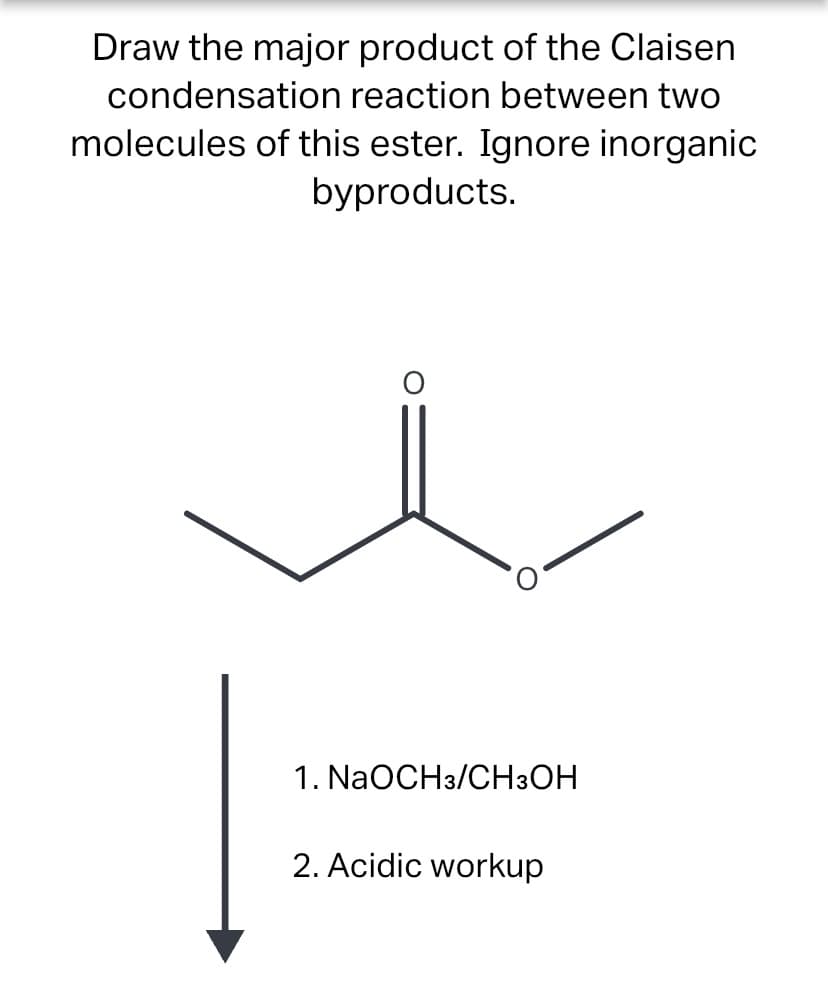
Draw the major product of the Claisen condensation reaction between two molecules of this ester. Ignore inorganic byproducts. 1. NaOCH3/CH3OH 2. Acidic workup
Q: N. O2-azabicyclo[2.2.2]oct-2-ene 1-azabicyclo[2.2.2]oct-2-ene O 1-azabicyclo[2.4,2]oct-2-ene
A: The given compound is a bicyclic compound. The root name of the bicyclic compound is given on the…
Q: water 97℃ Boiling point of solution 99℃ Mass of flask 118.2g Mass of distilled water w/…
A: Given, Boiling point of distilled water 97℃ Boiling point of solution 99℃ Mass…
Q: UESTION 9 Which of the following reactions gives the elimination products shown? Mark all that are…
A: Given that all the possible products are elimination products.
Q: Make a comparative table, highlighting similarities and differences, between the materials: a.…
A: Polyamide has a much greater capacity than silica gel and cellulose and is complementary to PC in…
Q: For the following endothermic reaction system at equilibrium: Fe203(s) + 3H2(g) * 2Fe(s) + 3H20(g)…
A: Le Chatelier's principle: This principle is used to predict the effect of a change in conditions on…
Q: Question 1: In each of the compounds below, circle all of the chiral carbons, if any. он Br Br OH…
A: Chiral Carbon => Carbon having 4 different substituents or groups attached. Chiral Carbon denoted…
Q: malate + NAD+ oxaloacetate + NADH + H+ If the pH = 7 and [malate] = 0.0010 M, [oxaloacetate] =…
A: For the given reactions the standard reduction potentials are taken from electrochemical series data…
Q: HC=CCH,CH, CH,CH=CHCH, CH3CH2CH2CH3 С. d. HCI, H2O, H2S e. CH3CH2CH3, CICH2CH2OH, CH3CH2OH
A:
Q: A 0.01kg plant extract in 2dm' of water was shaken with 0.1dm' ether. Upon checking, 1.7x10 kg of…
A: Mass of plant extract = 0.01 kg = 10 g
Q: 1.1 Fill in the missing (correct) words only: (a) In general, as and as increases. increases,…
A: Hii there, As there are multiple question posted. we are answering first question. If you need…
Q: What is the equilibrium concentration of Br2 if [HBr]=0.35M, [H2] =0.22M at equilibrium? K=62.5 H2 +…
A: Given that - Equilibrium constant , K = 62.5 Hydrogen bromide concentration, [HBr] = 0.35 M…
Q: One Erlenmeyer contains 50 mL of 0.1 M Na2CO3 solution, the other Erlenmeyer contains 50 ml of 0.1 M…
A: Here we have to determine the volume of HCl solution of 0.1M needed to react with 50 ml of 0f 0.1 M…
Q: Give some advantages of steam distillation over simple distillation.
A: Steam distillation is a process used for separating and purifying isolate temperature-sensitive…
Q: the isotope product?
A:
Q: Calculate the concentrations of all ions after 50.0 mL of a 0.100 M AgNO3 solution is mixed with…
A: Given : Concentration of AgNO3 = 0.100 M Volume of AgNO3 solution = 50 ml Concentration of Na2SO4…
Q: Complete the Table Type of Radioactive Particle Change in Change in Protection Decay Emitted Mass…
A: here, we have to find the symbols, change in atomic numbers, change in mass numbers, and the…
Q: Consider the reaction, HQ = H'(aq) + Q(aq). At 350.0 K, the AS°rxn D -81.0 J/mol·K and the AH°rxn =…
A: we need to find the value of pKa
Q: All of the following are true of SN2 reactions except: The rate of the SN2 reaction of a substrate…
A: Sn2 reaction is bimolecular nucleophilic substitution reaction.
Q: B The density of comm ercial concentrated HNO3 is 1-41 a /mL. The concentrated acid contains 69%…
A: Molarity :- The number of moles of solute dissolved in one litre of solution is defined as molarity…
Q: From zoom lecture on green house gases Use information from the zoom lecture to briefly explain why…
A:
Q: calculate the % yield of the preparation of benzocaine: mass of 4-aminobenzoic acid = 2.75g molar…
A: The percentage yield is given as the ratio of practical yield and that of theoretical yield, whole…
Q: Select all of the technologies that úse infrared light. ear thermometer O global positioning O TV…
A: Given: Devices which uses infrared light
Q: Given Ammonium, NH4*'. but the center element has to be The most electronegative element is Draw the…
A: a. Now we know that as we go from left to right in a periodic table the electronegativity increases…
Q: Alabandite is made of manganese (II) sulfide (MnS). The mineral adopts the FCC structure. The length…
A:
Q: A 6.91 mol sample of ethylene is burned in a bomb calorimeter that has heat capacity of 348.0 J C-1.…
A:
Q: Why did Rutherford think that there was a nucleus in the middle of the atom surrounded by space?…
A:
Q: Give the clear handwritten answer...
A: GIVEN:-
Q: Draw the structures and give the systematic names of the products below. (Show alcoLate For each) 1)…
A: Grignard reagent when reacts with formaldehyde gives primary alcohol...
Q: C-0, C-C, spC-H). ted to them (eg Mass spectrum (electron ionization) of dibenzalacetone Mark the…
A: Peaks at # 3020 cm-1 is due to C-H stretching of aromatic compound. # 1710 cm-1 due to CO bond…
Q: A 1,2-methyl shift occurs when which of the following iodides is heated in ethanol? O…
A: Solution - The correct option is -(b) (CH3)3CCHICH2CH3
Q: Question 2: In each of the compounds below, find the stereocenter, label the four groups from 1 to 4…
A: The R-S configuration is given to a stereocenter using Cahn-Ingold-Prelog (CIP) rules. The…
Q: Draw the structures and give the systematic names of the products below. (Show alcoLate For each) 1)…
A: When alkyl or aryl halide react with magnesium in presence of ether then grignard reagent is formed.…
Q: rate
A:
Q: Write the structural formulas corresponding to the IUPAC name: 4-isobutyl-2,5-dimethylheptane
A: The structure of an organic compound is written in the following steps: Determine the number of…
Q: Given that water is the most abundant chemical compound in living organisms, the way in which other…
A:
Q: Given the molecular structure of Caprazamycin B (CPB, figure 1), answer the following questions.…
A: 1.1 Atoms forming double bonds have sp2 hybrisation. Since there are 8 double bonds present ( CO2H…
Q: how many molecules are in 53.3 moles of co2
A: Mole concept: The number of molecules or atoms present in the one mole of the substance is equal to…
Q: CH2-CH3 СНЗСН2-S-S-CH2CH2CH3 CH3 CH3-C-CH2- SH | CH3
A: Given:
Q: Write a bond-line formula for each of the
A: Answer: Bond line notation is a shorthand way of writing organic structural formulas: The carbon…
Q: Propane (C3H8) is often used to heat homes. The combustion of propane follows the following…
A: Answer: 172.64 grams
Q: Match the correct formula to the correct term. Molarity A. grams solute /grams solution* 100 Percent…
A: Match the following--
Q: Determine the enthalpy change, AH°, for the following reaction; N2(g) + 2H2(g) → N2H4() Given the…
A: Determine the enthalpy change of the following reaction ---
Q: The thermochemical equation which is associated with AH°f, the standard enthalpy of formation for…
A:
Q: Feb 2021 solubility in water? A. Na+ and Clions are favorable sites for H-bonding to form. . The…
A: Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: Provide the correct IUPAC name for the skeletal (line-bond) structure shown here. Br
A:
Q: Which of these pertains to the process by which elements are formed within stars? A. Star formation…
A: As per bartleby guidelines I answered only first question so please don't mind.Thanks in advance.
Q: 6. Calculate AHReaction for the Reaction: N2H4 + 2N2O5 2HNO3 + 2NO2 + 2 NH oubave ro Aon AH = -303…
A:
Q: 2. Sodium nitrate decomposes into sodium nitrite and oxygen gas with heat. When 0.123 g of sodium…
A:
Q: For the oxidation of ammonia: 4NH3 + 3O2 arrow forward 2N2 + 6H2O it was found that the rate of…
A: Given: Rate of formation of N2 = 0.23 M/s
Q: 15. 1) Make a table showing the physical properties (molecular mass, m.p or b.p., solubility,…
A:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Show the arrow pushing mechanism for getting tropylium ion from the benzylic carbocationDefine a two-step method consisting of Grignard addition followedby dehydration, or a one-step Wittig reaction ?Why is the reaction of the type shown below usually done? a.To make an aldehyde or ketone less water soluble b.To make the molecule more reactive c.To protect a ketone or aldehyde carbonyl d.To increase the oxygen content e.To make the alpha hydrogens more acidic
- Please complete the following reaction showing all arrow pushing mechanisms. What is the name of this reaction?Please propose a synthesis of the target molecule using as many steps or reagents and answer these questions. 1. Why use chemoselectivity as the functional group and not another? 2. Why regioselectivity? 3. Why stereoselectivity? 4. What are the changes in the oxidation state?During benzoic acid synthesis, the Girgnard reagent is formed when this atom ______ inserts itself causing the initial type of carbon ____ to be changed to this final type of carbon ____. Please help fill in the blanks above.
- In a few sentences, briefly explain the difference between acid catalyzed and base promoted enolation of aldehydes and ketones.Write structures for the carbonyl electrophile and enolate nucleophile that react to give the aldol below. You do not have to consider stereochemistry. Draw the enolate nucleophile in its carbanion form. Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner. Separate multiple reactants using the + sign from the drop-down menu.Perform a retrosynthetic analysis and suggest a synthesis of the target molecule (on the left) from the given starting material (on the right). (any reagents maybe used
- Why is a protecting group used in this reaction? Ethylene glycol can be recycled which would decrease the E-Factor of this reaction. explain why this is based on the reaction scheme above.Show the complete and detailed reaction mechanism (oxidation reaction) of 3-bromobenzyl alcohol in the presence of 3-phenylpropanol. (show movement of electrons, charges, arrows, reagents, biproducts etc.)Please help with the attached OChem reaction. Fill in the correct reagents, provide the stepwise mechanism for both step 1 and 2, and include the correct use of arrows showing the flow of electrons. Thanks!

